
You float at the rim of an enormous spiraling canyon, its walls curving away in both directions like the interior of a living amphitheater — the oral groove of a single *Paramecium caudatum*, a structure no wider in reality than a few dozen micrometers yet rendered here as a vast descending ramp of biological architecture. The pellicle beneath you catches the phase-contrast illumination as a faintly metallic, striated membrane, its longitudinal protein ridges running in parallel bands across the ectoplasm, the entire surface shimmering in stark chiaroscuro — brilliant silver-white edged with ghostly diffraction halos against a deep charcoal void. Along the groove walls, thousands of compound ciliary organelles — membranelles and cirri packed in overlapping rows — beat in thundering metachronal waves that roll like slow luminous breakers down toward the cytostomal pit below, a pulsing aperture of absolute blackness where membrane trembles with the continuous business of phagocytotic engulfment. Rod-shaped bacteria tumble helplessly in the induced vortex current, glowing faintly gold-white where refracted light passes through their lipid membranes, spiraling inward and clustering as the groove narrows and flow accelerates — each one destined to be sealed inside a food vacuole and digested over the next several minutes by lysosomal enzymes. Above and behind you, the cell's body wall arches like frosted glass, and through it the macronucleus glows in warm amber — a massive kidney-shaped organelle that controls the vegetative metabolism of this single cell, a sovereign world operating entirely on chemistry, pressure, and the ceaseless mechanical labor of ten thousand coordinated cilia beating at up to forty strokes per second.

Looking up from your position in the depths, you are suspended in what feels like a still, faintly viscous olive broth, staring into a living chimney of *Euglena viridis* cells ascending in loose spiral formation toward the warmly glowing water surface far above — each 60-µm spindle body packed with coiling chloroplast ribbons that absorb the downwelling light and re-emit it as a saturated electric emerald, the collective effect transforming the entire water column into a slow-rotating pillar of green fire. Near the anterior tip of every ascending cell, a pinpoint stigma eyespot — a terracotta-orange fleck of carotenoid pigment shielding a photoreceptive swelling at the base of the flagellum — acts as a directional light sensor, steering each organism upward through positive phototaxis as it drives itself with an undulating anterior flagellum through a medium where inertia is negligible and viscous drag governs every motion. The pellicle, a corset of interlocking proteinaceous strips spiraling beneath the cell membrane, holds each body in its characteristic tapered shape while allowing the subtle euglenoid wriggling — metaboly — that ripples through stationary cells as a slow muscular contraction, the surface catching ambient light as faint iridescent sheen. The lime haze suffusing the water is not an artifact but a consequence: chlorophyll molecules fluorescing weakly under filtered daylight, the collective biochemical exhaust of thousands of simultaneous photosynthetic reactions occurring within cells close enough to touch, their interior lamellae quietly splitting water and fixing carbon in the warm light your upward gaze cannot quite reach.

You are pressed against a mosaic of fractured diatom frustules — silica relics whose hexagonal pore arrays and delicate striae refract light into scattered jewels across the dark sediment below — as the advancing pseudopod of *Amoeba proteus* looms overhead like a glacier calving in living glass. The hyaline leading cap is optically pristine ectoplasm, a dome of biological glass whose curvature bends ambient light into warm caustic arcs that sweep across the substrate ahead, while inside the transparent ectoplasmic tube the endoplasm surges forward as a dense, granular river of liquid bronze — food vacuoles the color of smoked honey, mitochondria like scattered copper shards, refractile granules tumbling in the cytoplasmic stream as cytoplasmic streaming, driven by actomyosin networks, propels this single cell forward at rates of 1–10 µm per minute. At the sol-gel boundary where flowing endoplasm converts in real time to the rigid structural wall of the ectoplasm, iridescent interference fringes shimmer in soft lavender and pale gold — a living phase transition made visible, the cytoplasm cycling between liquid sol and solid gel states as the pseudopod simultaneously builds its own walls and advances through them. The entire interior of this one cell is a cathedral of scale: you, the size of a human red blood cell, are dwarfed by a single pseudopodial extension whose mass recedes behind you into a trembling amber mountain of cytoplasm, its far contours dissolving into the warm brown-green haze of pondwater dense with dissolved organics and bacteria drifting in Brownian suspension.
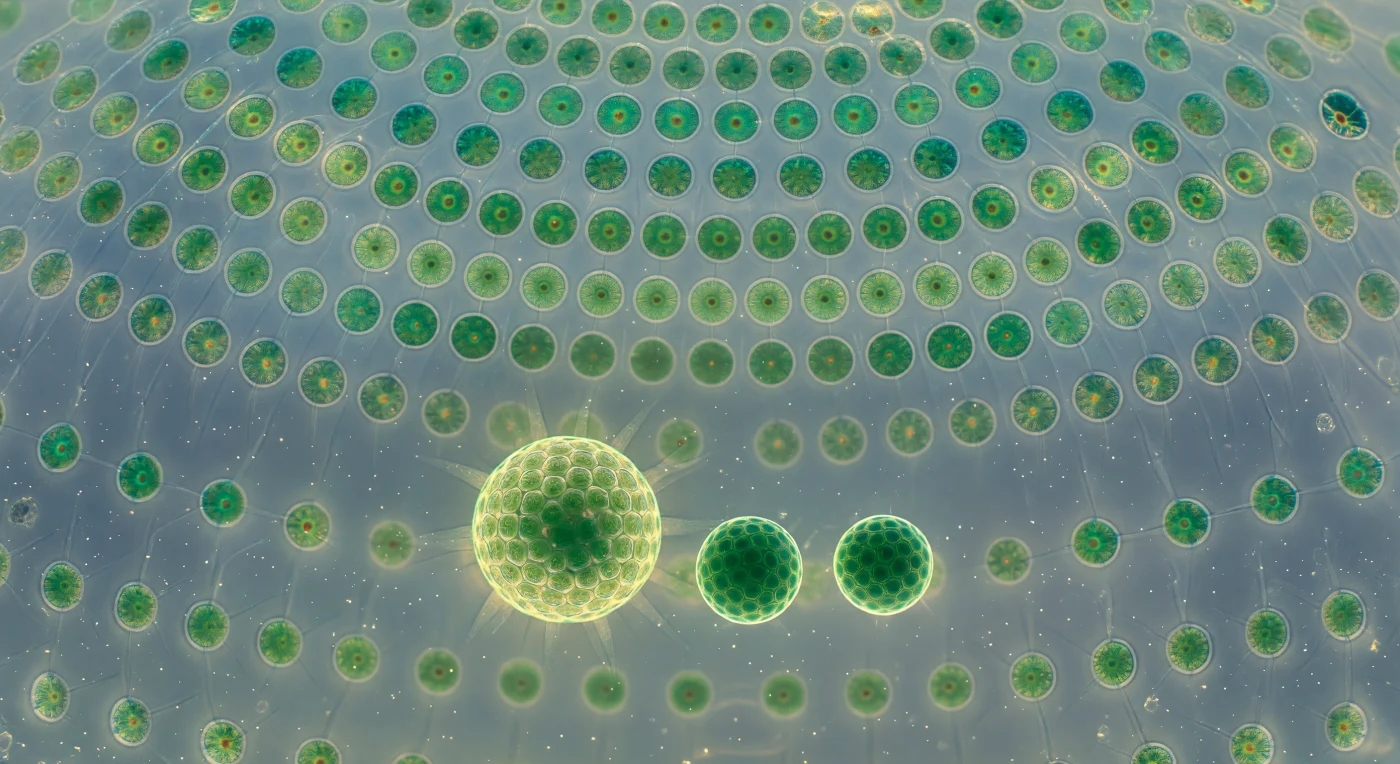
You stand at the hollow center of a living sphere barely half a millimeter wide, yet the scale it commands is monumental — the curving dome overhead resolved into a geodesic mosaic of thousands of somatic cells, each a ten-micron biflagellate organism locked into the transparent glycoprotein matrix like an emerald pressed into pale resin, connected to its neighbors by hair-fine cytoplasmic strands that catch the diffuse aquatic light as faint silver threads, stitching the entire colonial organism into a single coordinated architecture. This is *Volvox globator*, a green alga that occupies the threshold between unicellularity and true multicellularity: its roughly two thousand somatic cells are terminally differentiated — they can never divide — and exist solely to row the colony through the water column using paired flagella whose metachronal beat ripples continuously across the dome's inner face like wind crossing luminous moss, the collective motion rotating the whole sphere as it climbs toward light. Suspended in the clear fluid of the interior, three daughter colonies drift like planets in a glass reliquary — each one an inversion embryo that formed inside the parent through a developmental process called palintomy, dividing rapidly without growth and then turning itself inside out through a choreographed rupture, so that the flagella emerge facing outward — the largest daughter already shimmering with its own coordinated cilia-light, the smallest still a dense jade ball of cells waiting for its flagella to wake. The fluid between them trembles with Brownian motion, carries dissolved oxygen gradients shaped by photosynthesis, and holds the chemical signals that coordinate reproduction across the colony; the entire structure, from dome to daughters to the aqueous medium binding them, is a single organism in negotiation with itself.

The darkness here is not metaphorical — it is the true optical zero of open ocean water at night, a medium that presses with the slight viscous drag of a fluid too dense to ignore, carrying its cargo of dissolved salts and slow-sinking flakes of marine snow past your bacterium-scaled body. Then the first Noctiluca scintillans cell detonates its bioluminescent discharge twenty micrometers away: a cold, precise burst at 490 nanometers that lasts a tenth of a second and in that interval reveals a gelatinous sphere roughly a millimeter across — vast from where you float — its plasma membrane bending its own emitted blue-green light into a brief aureole while luciferin-laden vesicles at the cytoplasmic periphery dim from blue-white to teal as the chemical substrate exhausts itself. The flash is confiscated rather than extinguished, and the darkness that replaces it is total, but before any adjustment is possible a second cell fires three body-lengths away, then a third behind it, the chain reaction propagating through mechanical pressure waves in the water as each new discharge prints a cerulean afterimage: a crescent of transparent cell wall, the shadow of a food vacuole containing a half-digested diatom, a trailing tentacle catching cold light like a wet filament. Marine snow drifts into one burst and scatters it into a foggy halo, and the cumulative strobing turns the water into something like a storm seen from inside a thundercloud — each interval of absolute blackness already collapsing under the weight of the next discharge, overlapping rings of cold fire propagating outward through oceanic infinity.

You hover directly above the living drain of a single-celled giant, staring down into the spiraling oral disc of a *Stentor coeruleus* — a blue-green whirlpool roughly half a millimeter tall, yet structurally as intricate as any coral reef or rain forest canopy. The rim of the disc is ringed with compound membranelles, each a fused paddle of hundreds of cilia sweeping in tight coordination at twenty to forty beats per second, generating the slow clockwise gyre that continuously funnels bacterial rods and algal cells down into the cytostomal pit below, a dark gravitational center from which nothing returns. Through the semi-transparent pellicle the macronucleus descends like a string of river pearls — its beaded lobes the control architecture for the thousands of genes the organism must run simultaneously, a single nucleus shattered into redundant nodes threading through cobalt endoplasm dense with food vacuoles at every stage of digestion. The parallel striations of stentorin pigment granules run longitudinally across the cortex in alternating Prussian blue and pale aquamarine bands, a biological textile so precisely repeated it reads as woven fabric rather than chemistry. Around the outer disc, diatom frustules and smaller protists orbit just beyond the pull of the current like debris at the edge of a gravity well, tracing arcs through amber-hazed water that is not empty but thick with dissolved organics, Brownian noise, and the slow chemical conversations of a living freshwater world.
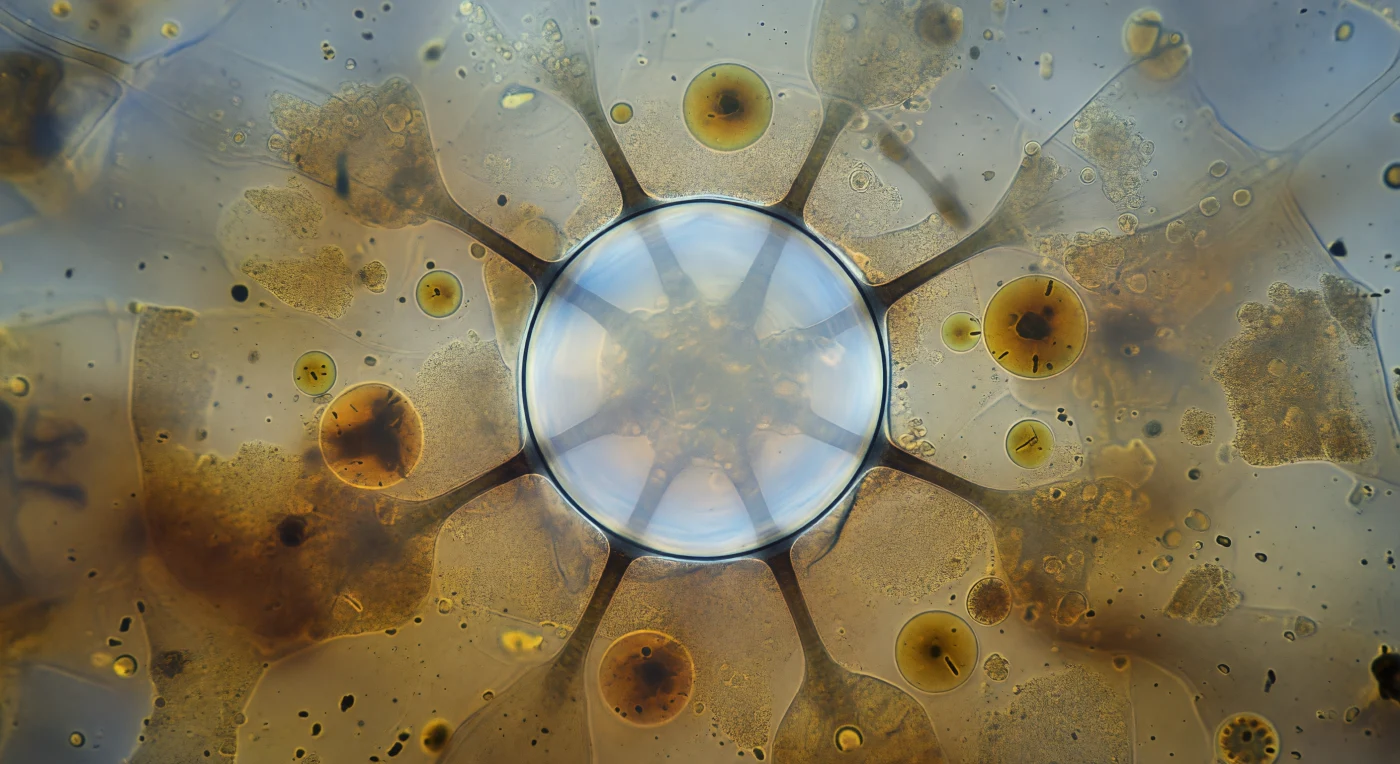
You are suspended within the cytoplasm of a living Paramecium multimicronucleatum, pressed face-to-face with a contractile vacuole thirty micrometres across — a sphere of pure osmotic tension that fills the foreground like a cathedral dome of living glass, its membrane so vanishingly thin it registers only as a hairline of silver-cold pressure, its interior glowing with the pale blue-white luminescence of water gathered over hundreds of seconds of continuous pumping against the cell's own osmotic gradient. Six nephridial tubules radiate outward from its equator as dark spokes dissolving into a dense amber haze of food vacuoles and tumbling granules, each tubule a membranous canal that has spent the entire fill cycle harvesting cytosolic water from the surrounding endoplasm through active proton-pump-driven mechanisms — a remarkable feat of hydraulic engineering in an organism with no kidneys, no excretory organs, no multicellular infrastructure of any kind. The surrounding cytoplasm presses inward from every direction as a slow granular tide of mitochondria, lysosome-laden food vacuoles ranging from five to fifteen micrometres, and dense near-black granules drifting on invisible currents of cytoplasmic streaming, the entire crowded interior existing at Reynolds numbers so vanishingly small that inertia is meaningless and every motion is governed entirely by viscosity and diffusion. The vacuole hangs at its absolute volumetric limit, membrane curvature at maximum, the entire system one millisecond from total systolic collapse — a violent expulsion through the pore complex at the cell surface that will eject the full contents outward in under 100 milliseconds, the cell's only defense against the osmotic tide endlessly trying to swell it to rupture.
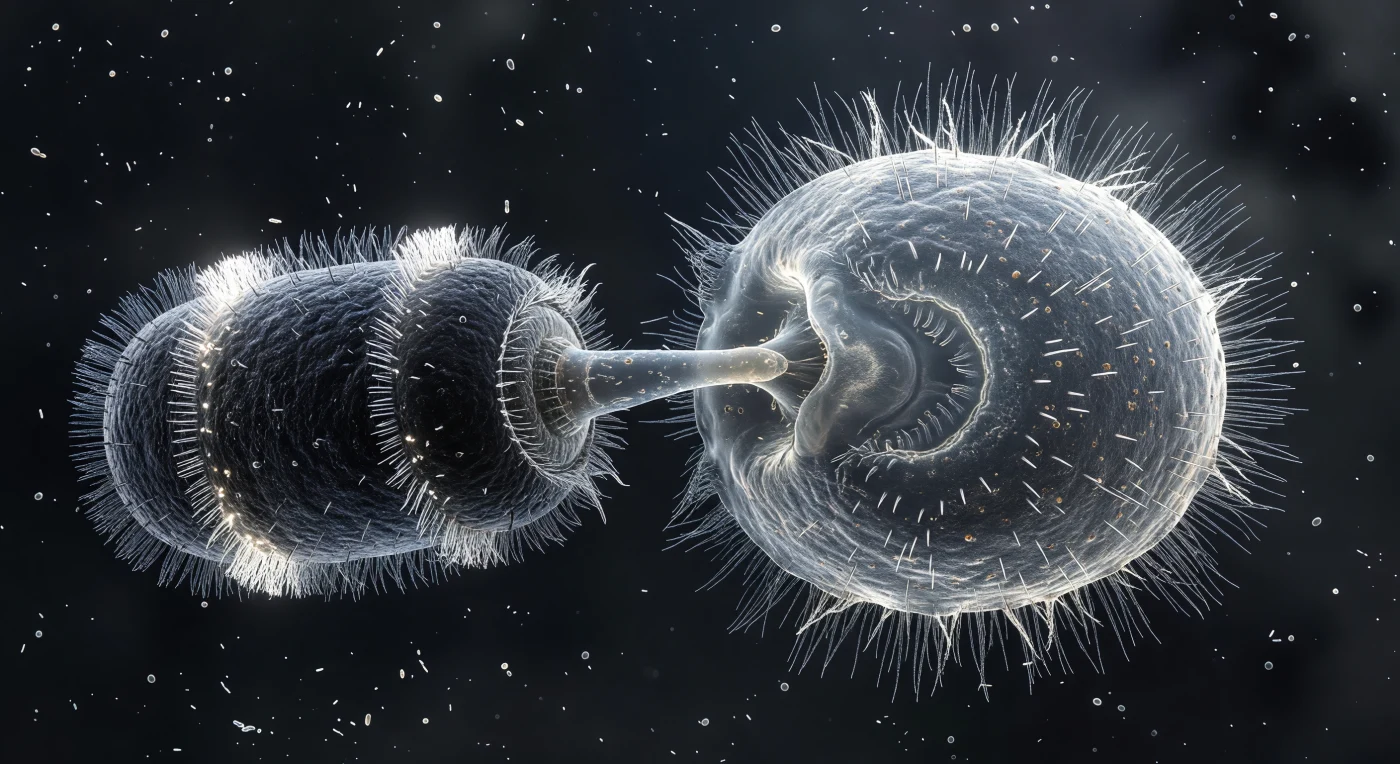
At the moment frozen here, a compact barrel-shaped predator occupies the left of the scene, its dark cylindrical body encircled by two luminous coronets of beating cilia — the Didinium nasutum in full attack posture, its muscular proboscis pressed against the flanking membrane of a Paramecium three times its size. The point of contact has become a crushed, glowing concavity where the Paramecium's pellicle — that semi-rigid alveolar cortex normally holding the cell's football form — is caving inward under a focused mechanical thrust that neither organism can reverse. Across the entire Paramecium surface, thousands of trichocyst filaments have discharged in the same instant, each a protein thread extruded at explosive speed from a cortical capsule, the collective result a radiant corona of spun-glass strands extending deep into the bacterial haze — a last-resort chemical and physical defense now clearly arriving too late, as the proboscis tip has already committed the pellicle to irreversible deformation. Inside the Paramecium's translucent body, food vacuoles at varying stages of digestion glow amber and brown against the hyaline ectoplasm, and the cilia ringing the cell have shattered from their normal metachronal wave into a disordered defensive bristle, the coordinated swimming machinery overwhelmed by a distress signal propagating through the cortex faster than the beat cycle can compensate. The surrounding medium reads as a near-black continuum alive with drifting bacterial rods and cocci at varying focal depths, a reminder that this arena of predation operates entirely within the viscous, inertia-free physics of low Reynolds number flow, where force is transmitted directly and instantly through the aqueous film, and where the compressed geometry of two colliding cells at this scale carries the visual and physical weight of a collision between much larger worlds.

You hover above a silty seafloor like a ghost the size of a bacterium, the entire scene suffused with a sourceless blue-green transmitted light that turns the sediment into an ochre desert of mineral boulders and organic debris too vast to step over. Before you, the calcite architecture of a living foraminifera rises as an overwhelmingly white and luminous spiral of globular chambers — each surface smooth and chalk-brilliant, each suture a precisely carved groove, the whole structure built by a single-celled organism that secretes calcium carbonate walls and adds new chambers across its lifetime in a logarithmic sequence guided by no blueprint but chemistry and membrane geometry. Radiating outward from its aperture in every direction across that sediment surface is the reticulopod network: a web of anastomosing, ghost-thin cytoplasmic strands nearly invisible against the blue-green haze, betraying their existence only through the amber streams of organelle granules flowing bidirectionally along them — inward convoys of captured nutrition, outward flows of exploratory cytoplasm, the whole traffic system driven by myosin motors walking actin tracks within filaments barely wider than a few hundred nanometers. To your left, a diatom frustule — its silica walls perforated by pore arrays that scatter faint prismatic light — is gripped by converging strands and drawn imperceptibly inward, prey caught in a trap whose geometry is legible only by watching what moves through it, a living net spun not from silk but from the organism's own extended body.

You are suspended in murky freshwater just below the sediment surface, gazing upward into a space dominated almost entirely by the curved amber ceiling of an *Arcella vulgaris* test — a secreted chitinous dome perhaps 100 to 200 micrometers across, its semi-transparent walls transmitting light as a warm honey-gold stain, thickening toward the equatorial rim into deep amber-brown like centuries-old lacquered resin. At the precise center of this vaulted ceiling, the aperture opens downward as a dark circular portal ringed by a sharp chitinous lip, and from it descend four lobopodia — thick, blunt-tipped cylinders of hyaline ectoplasm, utterly transparent yet structurally substantial, extending with the glacial slowness of pseudopodial flow at the low Reynolds numbers that govern all motion at this scale, where viscosity overwhelms inertia and every movement is a negotiation with the fluid itself. These extensions of living cytoplasm carry faint internal granules drifting in the streaming endoplasm, and where they catch transmitted light they glow a cold blue-white against the dome's warmth, each one a probe reaching into the particulate world below — a world of drifting bacterial rods, fragments of decaying plant material, and colloidal organic matter that renders the water itself luminous and dense with chemical information. The entire scene conveys the reality that this single-celled organism, armored in its self-built organic shell, is actively engaging its environment through coordinated cytoplasmic extensions while anchored safely within its amber vault.

The view fills entirely with living green — you are pressed against the outer wall of a single *Paramecium bursaria*, the cell's transparent pellicle curving away in every direction like the hull of an illuminated lantern, its fine longitudinal ridges catching transmitted light as bright silver seams in the pale amber membrane. Beyond that thin architectural boundary, hundreds of *Chlorella* endosymbionts crowd the cytoplasm in an almost unbroken mosaic, each a compact chlorophyll-loaded disc roughly four micrometres across, their boundaries nearly touching so that the interior reads as a continuous forest canopy — a dense, layered ceiling of saturated green pressing outward to the cell's margins and receding inward through progressive shadow and softening focus. These are not passive passengers: *P. bursaria* maintains its symbionts through a suppression of the normal lysosomal digestion that would destroy ingested algae, while the *Chlorella* repay the arrangement with photosynthetic sugars, making the whole organism a self-contained greenhouse that migrates toward light as a single behavioral unit. Deep within the green mass, the macronucleus floats as a kidney-shaped clearing of pale grey-lavender, its smooth boundaries luminous against the particulate darkness around it — the polyploid somatic nucleus that governs the cell's daily housekeeping while a much smaller micronucleus, invisible in this crowd, holds the germline in reserve. At the outermost margin, the cilia fringe the pellicle as a continuous silver halo of backlit filaments, each individual hair beyond clean resolution but the whole array shimmering with the coordinated metachronal wave that drives this green, inhabited vessel through its aqueous world.
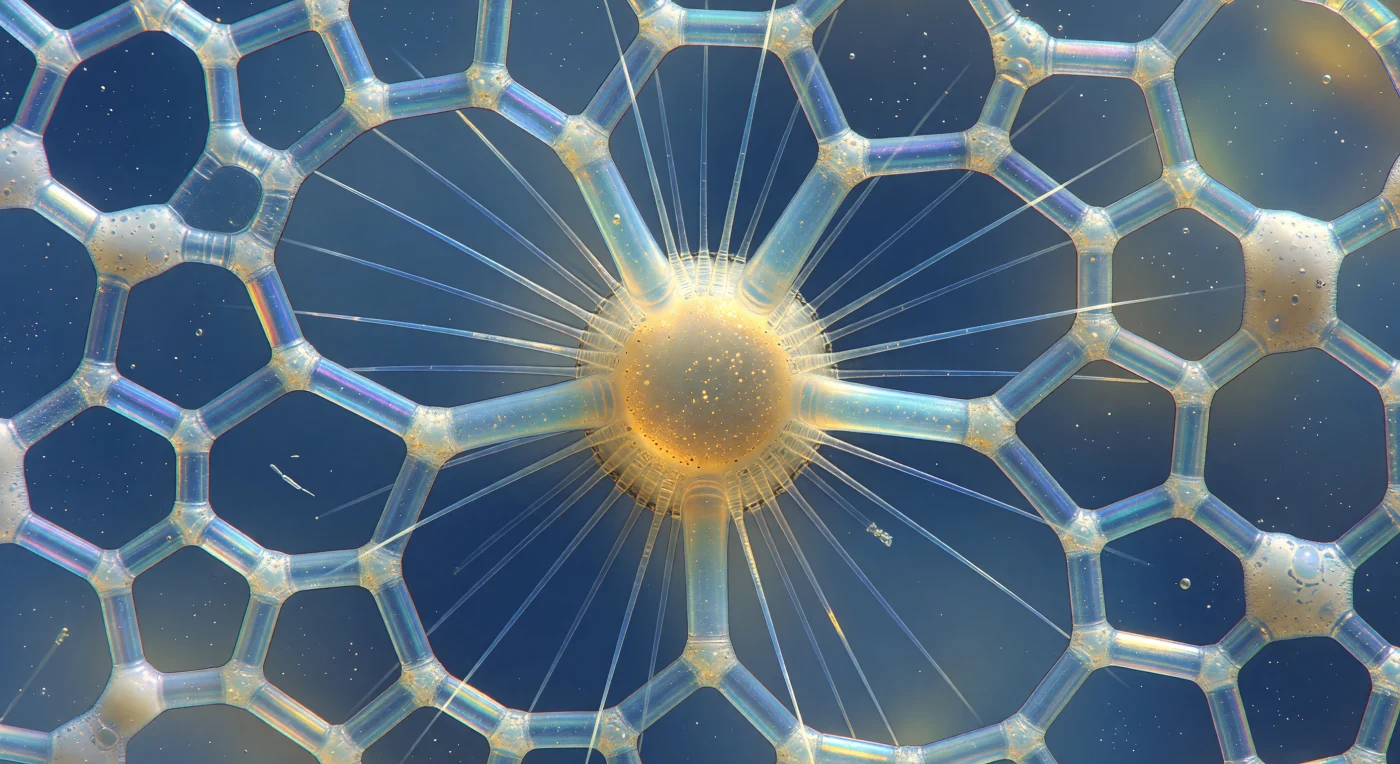
You are suspended at the equatorial flank of a single polycystine radiolarian, your entire field of view consumed by an architecture of amorphous opal silica — beams no thicker than spun glass curving and perforating the water around you like the clerestory of a drowned Gothic cathedral, each strut fragmenting the diffuse blue-green downwelling light into cascading interference fringes of violet, cobalt, and warm amber that shift and dissolve with the faintest thermal shimmer. Every hexagonal and pentagonal pore frames an open window onto the deep cerulean haze beyond, rendering the whole skeleton as an illuminated reliquary suspended against perpetual oceanic twilight, while at the core the central capsule — a perforated organic wall enclosing cytoplasm laden with lipid droplets and crystalline strontium sulfate ballast — glows with a dense amber-gold luminosity like light strained through ancient resin. From that core, axopods radiate outward as perfectly straight refractive needles, stiff bundles of microtubules arranged in a precise geometric array, each one bending the ambient light into a prismatic sliver before vanishing into the haze, while between the outermost skeletal spines a froth of vacuolated ectoplasm clings in translucent blebs, each tiny vacuole momentarily concentrating the interference colors into a bright point. A single bacterial rod tumbles end-over-end through the nearest large pore opening — an involuntary scale marker, impossibly small against the cathedral geometry it is passing through — and fine Brownian particulates drift as silver specks across the chamber windows, reminding you that the water here is not empty but a dense chemical theater in which this extravagant mineral architecture has filtered, floated, and preyed for hundreds of millions of years.

We drift just above a sediment floor of collapsed geometry — cylinders of ribbed silica toppled against boat-shaped keels, triangular shields propped at angles against star-form fragments, every surface perforated with pore arrays so fine they exist at the boundary of visible resolution. The material is diatom frustule, amorphous biogenic opal secreted valve by valve during the organisms' lives and now accumulating in layer after compacting layer, each piece between twenty and a hundred and fifty microns across, its silica body neither fully transparent nor opaque but possessed of the milky internal depth of polished moonstone. Transmitted light pours through every fragment and refracts at each striation boundary into interference fringes — cobalt, pale gold, rose — so that the sediment surface shifts its entire color register when our perspective moves by even a hair, a spectral mosaic generated entirely by nanometer-scale periodicity in the pore geometry rather than by any pigment. Out beyond the nearest ruins, where collapsed frustule columns open into corridors of stacked debris receding into blue-white depth-haze, an Actinophrys heliozoan hangs suspended in the water column like a living chandelier, its axopods — needle-straight rods stiffened internally by organized microtubule bundles — radiating outward in all directions and capturing transmitted light along their length, glowing like fiber-optic filaments above a crystal ruin field that extends, layer beneath layer, in all directions without a visible end.
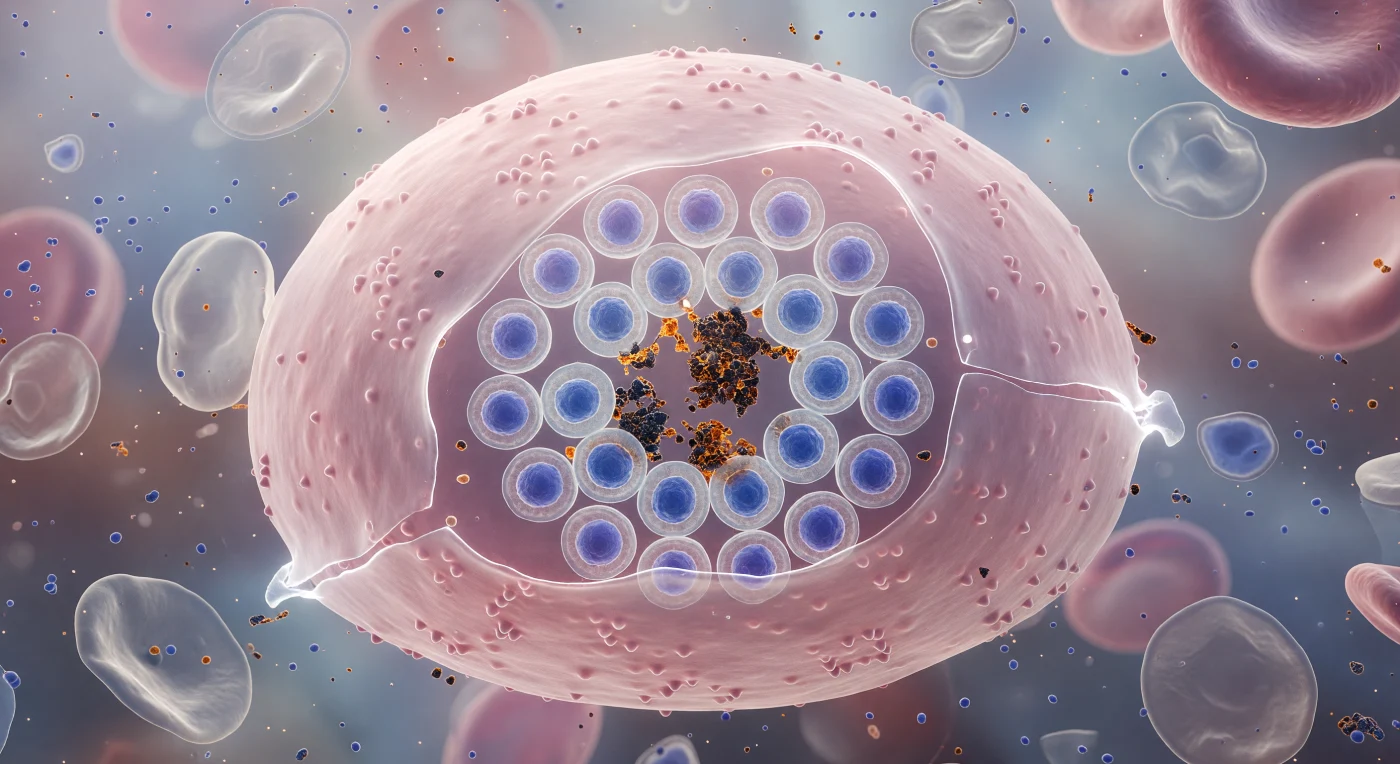
The surface pressing against your imaginary body is the outer membrane of a human red blood cell swollen far beyond its normal discoid shape — a vast pale-rose vault stretched to mechanical near-failure, its lipid bilayer thinned at intervals to a luminous, almost white translucency through which the catastrophic geometry inside is faintly visible. Within, twenty-four *Plasmodium falciparum* merozoites have arranged themselves into a schizont rosette, each parasite a compact ovoid with a cobalt-blue nucleus stained by Giemsa chemistry, the entire wheel-like assembly radiating outward from a central cairn of hemozoin — crystallized heme detoxification byproduct, chemically inert iron-porphyrin polymer, refracting the diffuse biological backlight into brief amber glints against the prevailing blue-violet cold. The host erythrocyte has been entirely hollowed: its hemoglobin digested, its cytoskeleton remodeled from within by exported parasite proteins that have pushed up dense knobby protrusions across the outer membrane surface, each knob an anchor point for cytoadherence to blood vessel walls. Osmotic pressure has been building for roughly 48 hours of intraerythrocytic development, and the membrane is now at mechanical failure — the contrast between its warm salmon-rose and the dense jewel-indigo of merozoite nuclei pressing outward reads as both exquisitely beautiful and functionally violent, the entire scene suspended in the last instant before rupture releases twenty-four invasion-competent daughters into the surrounding plasma.

You are suspended alongside a structure that has ceased to be microscopic — it has become geological. The armored flank of *Ceratium tripos* stretches across your entire visible world, its thecal plates glowing amber and burnt sienna as transmitted blue-green light filters upward through living cellulose, turning each polygonal panel into a luminous tile of warm organic gold. These are not simple surfaces: each plate is a flattened mesa of layered cellulosic material, its edges defined by raised suture ridges — geometrically precise fault scarps where neighboring plates meet at angles that would not look out of place in a satellite image of tectonic terrain — and across each mesa face, parallel micro-ridges run in orderly striations interrupted by perfectly circular pores, each one a dark well descending through the theca's laminated wall like a drainage shaft bored through sedimentary rock. Cutting across the mid-frame, the equatorial cingulum opens as a canyon of cobalt-indigo shadow, its steep walls exposing the plate material in cross-section as faint concentric banding, and deep within the trench the transverse flagellum lies coiled in near-darkness, a thread of pale iridescence catching scattered light — the mechanism of the organism's spiral locomotion resting in its groove, invisible to any world beyond this one. At the left edge of frame, the apical horn rises and curves away out of sight, a tapered spire of the same amber armor sweeping a long shadow back across the near plates, while behind it the open water column dissolves into featureless blue-green translucency, the background of a world where every surface is alive and every wall is a carefully assembled architecture of organic chemistry.
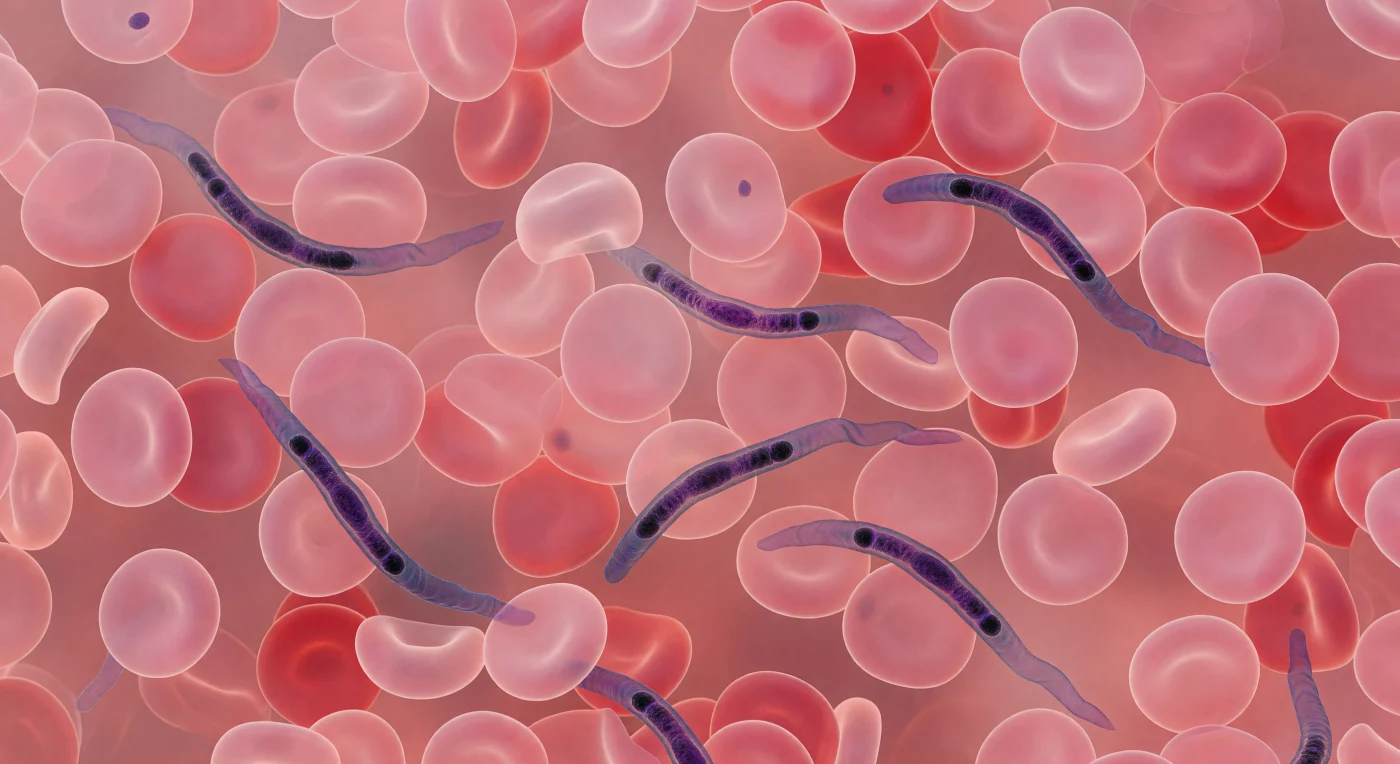
The view is an overwhelming immersion inside living tissue — in every direction, soft rose-pink biconcave discs press close, their thinned centers transmitting amber light as faint aureoles, their thickened carmine rims overlapping until the visual field dissolves into a blush fog of stacked geometry with no horizon and no escape. Between these densely crowded erythrocytes, each approximately seven micrometres across, Trypanosoma brucei navigate the narrow interstices like sinuous calligraphy — elongated blue-violet bodies fifteen to thirty micrometres long, their undulating flagellar membranes propagating slow silk-ribbon waves that represent the parasite's primary means of locomotion through a fluid medium governed entirely by viscous drag rather than inertia. Giemsa staining saturates the scene with its characteristic palette, concentrating in the dense chromatin of the nucleus and the compact kinetoplast — that extraordinary mitochondrial DNA mass unique to kinetoplastid protists, sitting near the posterior end of each parasite body like a dark seed, its position marking the base of the flagellum and anchoring the entire motility apparatus. These organisms are not incidental passengers but active architects of pathology: Trypanosoma brucei crosses the blood-brain barrier to cause African sleeping sickness, and the undulating movement visible here is the same swimming behavior that allows the parasite to evade host immune surveillance while traveling through exactly this claustrophobic corridor of red cells and plasma.

You hover two hundred micrometres below a ceiling that looks like liquid mercury — the underside of the pond's surface film — which shimmers and ripples with the micro-wakes of dozens of organisms pressing against it from below, returning afternoon light as a broken, amber-tinged mirror. The community suspended around you represents the neuston, a thin biological stratum exploiting the physics of the air-water interface: here, dissolved organic matter concentrates, dissolved oxygen peaks, and light intensity is highest, drawing photosynthetic protists upward until they pack the surface film at densities rare anywhere else in the water column. Euglena, each a tapered emerald spindle between fifteen and eighty micrometres long, jostle with bead-like Chlamydomonas cells whose brick-red eyespots — dense carotenoid granule arrays tuned to detect photon direction — burn like heated coals against the green, while barrel-shaped Coleps tumble slowly among them, their armour of interlocking calcium carbonate platelets fracturing the caustic light into hard facets. Threading through the bacterial haze that softens every distant form into a blueish-silver fog, a Spirogyra filament angles across the background like an industrial girder, its cellulose wall bending the ambient light into a bright specular edge and revealing the spiralling jade-green chloroplast ribbon within. This is a world governed entirely by viscosity and chemistry rather than by inertia: at Reynolds numbers far below one, every organism swimming through this crowded amber medium feels not open water but something closer to warm syrup, every flagellar stroke and pellicle contraction a deliberate negotiation with fluid that forgets no movement instantly.

The world you are looking out from is orange-amber and articulated — chitin ridges curving into frame at the edges of vision, stub-limbs trembling against something that should not be strong enough to hold anything, yet does. Around you, in every direction, the reticulopod network of *Globigerina bulloides* radiates inward like silver fractures in blue-green ice: threads of 0.2–0.5 µm diameter, far below the threshold of easy resolution, each one revealed only by the continuous bead-like procession of organelle cargo drifting steadily along it — amber vesicles, dark mitochondria-like particles, all moving with unhurried metabolic purpose toward the chalk monolith hanging in the upper distance. That structure, the foram's calcite test, is a cold architectural fact: spherical, chambered, its surface pocked with hundreds of apertures from which these threads emerged and now converge back, bearing you with them in increments too slow to feel as motion but too geometrically coherent to be anything but intent. The surrounding medium is not emptiness but a colloidal reality — marine snow drifting in Brownian arcs, bacterial specks catching light, dissolved organics casting amber warmth into the blue-green column — a fluid world in which foraminifera routinely capture copepod nauplii many times their own reticulopod diameter, their webs exerting cumulative adhesive and contractile force across dozens of simultaneous contact points. There is no sudden violence here, only the slow mechanical closure of a trap that was already complete before the struggle began.

You are suspended in the open ocean at twenty meters depth, looking upward through a column of water that has filtered sunlight down to its last surviving wavelength — a saturated cobalt blue near 460 nanometers that fills the entire visual field like the interior of a luminous gem. Throughout this column, Acantharia drift at multiple focal depths, each one a starburst of twenty strontium sulfate crystal spicules arranged in the precise geometric symmetry of Müller's law, their birefringent shafts shattering the downwelling light into prismatic coronae of ice-white and pale violet that bloom outward like cold halos — these crystals are among the only biological hard parts on Earth built from strontium sulfate rather than silica or calcium carbonate, and they dissolve completely when the organism dies, leaving no fossil trace. Scattered among them, tintinnid ciliates hang at oblique angles within their hyaline lorica vases — tapered tubes of agglutinated coccoliths that transition from amber at the base to near-perfect transparency at the open rim, where a barely-resolved fringe of cilia marks the threshold between organism and ocean. Between all these crystalline architectures, soft flocs of marine snow — mucus aggregates, colonial debris, and detrital particles — drift across the frame, their warm amber and cream surfaces a quiet counterpoint to the mineral precision of the spicules, everything suspended together in a water column that functions not as empty space but as a living medium, faintly particulate, chemically complex, and threaded through with dissolved organic matter that gives the blue its almost electric warmth.

Across a vast fractured plain of desiccated clay, warm oblique light rakes over a mosaic of polygonal mud plateaux separated by shadowed crevasses, illuminating the scattered survivors of a vanished pond: amber spheres and chitin domes of dormant protist cysts that rest against the granular substrate like polished beads dropped on broken stone. Each Colpoda cyst is a masterpiece of biological engineering — a double-walled chitinous capsule roughly 15 micrometres across, its outer wall chemically hardened against osmotic assault and UV damage, enclosing a metabolically arrested cell that has shut down nearly all biochemical activity and can persist in this state for years, even decades, through cycles of freeze, desiccation, and chemical stress. The Arcella tests nearby are domed helmets of self-secreted organic material, their apertures sealed with a translucent plug, their surfaces bearing a fine geometric lattice of protein subunits — each one a biological vault whose inhabitant has withdrawn entirely from the world. Between the solitary cysts, collapsed sheets of dried Euglena palmella mucilage catch the raking light with a faint biological sheen, their olive tints betraying dormant chloroplasts still holding their pigment inside cells that have surrendered motility and individuality for collective encystment in a communal gel. Everything here is suspended in a kind of deep biological time, waiting — each smooth, perfected form a stark, luminous anomaly against the fractured geological disorder of the substrate — for the chemistry of water to return and unlock them.




















