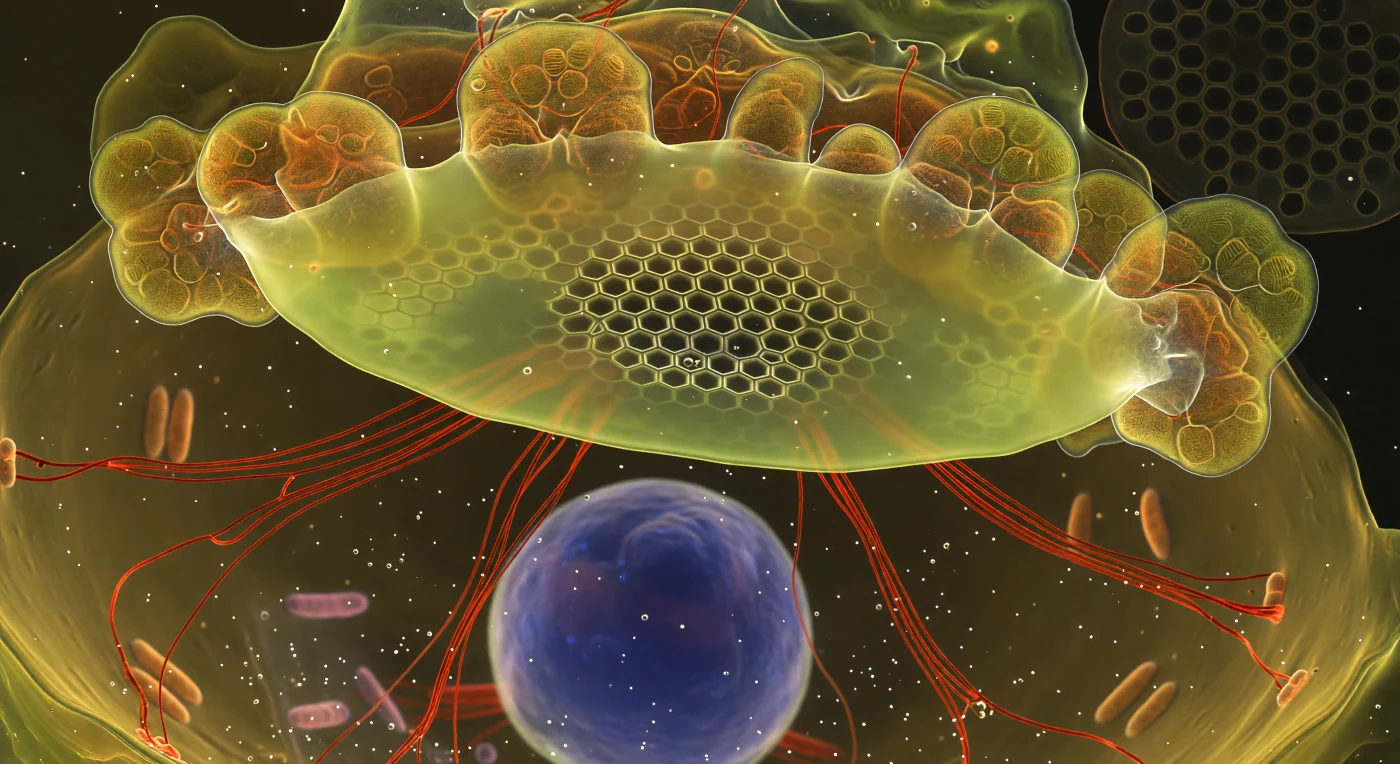
You are suspended inside a living diatom cell at the precise moment of valve birth — an event unfolding over hours yet arrested here in a single luminous instant, the completed silica honeycomb snapping into crystalline focus at the center of the Silica Deposition Vesicle while its outer edges dissolve into a glowing fog of mineral indecision, silica still negotiating its final hexagonal form. The SDV itself stretches like a chartreuse ceiling across the entire field of view, lit from within by PDMPO dye incorporated into freshly polymerizing opaline glass, its resolved core a lattice of dark areolae framed by hair-thin walls burning white-gold — the same architectural logic that has been pressed into ocean sediments for hundreds of millions of years now crystallizing in real time above you. From beneath this forming valve, crimson actin cables radiate outward in taut, bifurcating bundles, their phalloidin-labeled surfaces cutting through the amber cytoplasm like heated wire, transmitting the mechanical forces that sculpt valve geometry with molecular precision. Golden chloroplast lobes press upward against the silica ceiling, their thylakoid lamellae stacked like pages of dense biological text, while far below in the cytoplasmic dark a DAPI-stained nucleus floats like a cobalt moon — the genome that encodes all of this impossible architecture hovering in patient suspension as the cell manufactures its own glass house around it.
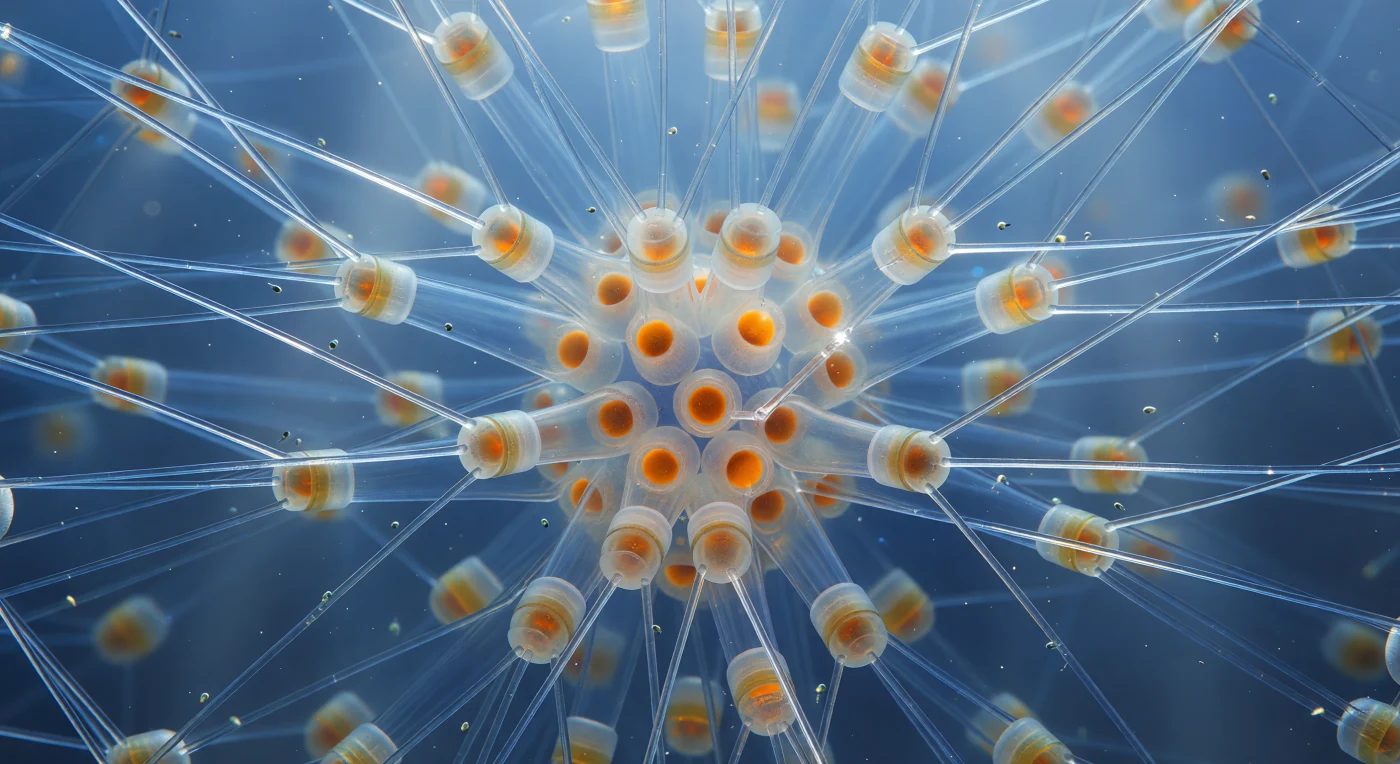
You are suspended inside the geometric heart of a *Chaetoceros* colony drifting in open ocean, surrounded in every direction by hollow silica setae—each needle nearly transparent, no thicker than spun quartz, extending from stacked cylindrical cell bodies in interlocking X-lattices that recede into cold indigo haze. These bristles are biogenic glass: amorphous hydrated silica (SiO₂·nH₂O) deposited by living cells within membrane-bound vesicles, their walls so thin that ambient oceanic blue passes clean through them, broken only by bright hairline caustics where curved surfaces concentrate filtered sunlight into white-gold filaments suspended between the struts like trapped stars. At the colony's dense core, resting spores glow with a deeper, more saturated amber—heavily silicified spheres whose thickened valves scatter light diffusely, warm embers of organic density anchoring the crystalline scaffolding—while the cylindrical cell bodies ringing them reveal their fucoxanthin-rich chloroplasts as burnt-sienna warmth pressed against frosted girdle rings stacked like sections of translucent glass tubing. Nanoscale flagellates drift between the setae on invisible currents, each a pale grey-green mote catching light for a single instant as it turns, the water itself carrying a faint milky haze of dissolved organics and silica debris that softens the far background into gradients of cerulean and slate—the whole structure reading as a living chandelier of biogenic glass, an architectural interior whose microscopic truth is entirely lost from the outside world.

You hover above what appears to be an immense circular floor of luminous glass, every surface carved into a seamless tessellation of hexagonal chambers extending to the curved horizon in every direction — this is a single valve of *Coscinodiscus*, a centric diatom whose frustule of amorphous hydrated silica (opal-A) has been bioengineered at the nanoscale into concentric rings of areolae, each hexagonal cell measuring a few hundred nanometers across its silica walls yet collectively spanning roughly 150 micrometers of open ocean. Transmitted white light rises through the structure from below, refracting through silica walls of varying thickness to produce the warm amber glow at the disc's center and the thin-film iridescence — saffron bleeding into violet-blue — that blazes at the margin where the silica thins to near-transparency, the same interference physics that colors soap bubbles and butterfly wings. At regular intervals across this mandala plain, fultoportulae rise as hollow cylindrical glass chimneys, strutted processes whose open bores vent dissolved gases or mucilage strands outward into the seawater column, each tower catching backlight differently so that some glow amber from within while others rim themselves in cold diffractive blue and cast soft elliptical shadows across neighboring areolae, lending the otherwise flat valve its subtle topographic depth. The surrounding ocean is a faint cyan transparency, carrying wisps of dissolved organics that haze the upwelling light at your peripheral vision, and beyond the disc's beveled lip — flashing a last prism of blue-green iridescence — there is nothing but open water and the weightless suspension of a living glass architecture that, when this organism dies, will drift intact to the seafloor and endure in diatomite sediments for millions of years.

You are suspended just above a polished glass plain that extends to every horizon, its surface barely distinguishable from the seawater film lying across it — a molecular-thin glycoprotein slick giving the substrate a faint iridescent sheen, as if the ocean floor itself had been compressed to a single luminous sheet. Ahead of you, slow and inevitable as a barge crossing a harbor, a *Navicula* cell thirty micrometers long slides rightward through this world: a biconvex hull of amorphous hydrated silica whose valve face is finely corrugated with transverse striae, their diffraction shimmer cycling through steel-blue and warm bronze as oblique phase-contrast light catches the dorsal ridge in a crescent of silver-white. Within the translucent frustule, two large chloroplasts glow with saturated golden-ochre light — fucoxanthin pigment bleeding softly through the silica walls and pooling on the glass beneath the cell in twin lobes of amber warmth — while the raphe slit, a near-invisible seam running the cell's median axis, continuously extrudes the adhesive mucilage that both anchors and propels the organism through the friction of its own secretion at speeds of one to twenty-five micrometers per second. Trailing roughly eight micrometers behind the living hull, a single bacterial rod clings to the pale-yellow refractive wake of that mucilage ribbon — dark, comma-shaped, utterly still — held fast by the same extracellular chemistry that bonds diatom to glass, a reminder that even this solitary gliding cell drags an ecological relationship behind it.

You float at eye level with a single descending column of *Thalassiosira weissflogii* cells, each disc roughly a hundred microns across — vast circular platforms of translucent biogenic silica suspended in saturated cobalt water, their valve faces turned toward you like rose windows in an underwater cathedral, the radial geometry of hexagonal areolae refracting downwelling sunlight into soft iridescent golds and greens that shift with the slightest change in angle. Between every adjacent cell, a single chitin fibril — extruded through specialized fultoportulae and under genuine molecular tension — runs the length of the chain, invisible except where an oblique shaft of light ignites it as a silver filament, the whole structure curling with a slow helical bias so that the thread pulses in and out of visibility as the colony rotates in the current. Through the translucent silica of each valve you can trace the warm ochre lobes of the chloroplasts arranged in a sunburst around the cell interior, lipid droplets catching light like beads suspended in amber resin, while bacteria cling to the outer silica surfaces as dark specks no larger than grains of soot — exploiting the organic coatings and exudates that leak through the pore network into the surrounding ocean. Below the last visible disc the chain dissolves into blue volumetric haze, and a loose aggregate of marine snow — mucilage, broken frustule fragments, and detritus — drifts past to the left, backlit so its gauzy edges glow, a reminder that everything shed or excreted by these cells feeds the ocean's slow downward flux of carbon.

You are suspended at the heart of a Victorian diatomist's masterwork, surrounded by a constellation of silica architecture hovering in absolute black — each frustule a self-luminous reliquary of amorphous opal-A glass, acid-cleaned to pure mineral skeleton and glowing with cold interference light. These are the frustules of single-celled algae, organisms whose bodies are entirely enclosed in precisely nanofabricated silica shells built in two interlocking valves, each hexagonal areola a diffraction grating only nanometers thick that splits incident light into electric cobalt, smelted amber, and shivering violet across its surface. The *Triceratium* blazing before you spans perhaps forty times your simulated body length, yet it is a single cell's worth of architecture — its terraced silica ramparts and pierced lantern pores the product of a biological silica deposition vesicle working with molecular precision over a matter of hours. Around you, the *Coscinodiscus* wheel, the pearl-spoke *Arachnoidiscus*, and the sigmoid calligraphy of *Pleurosigma* each glow with their own spectral identity, their striae densities and pore geometries the very characters taxonomists use to read species identity like text. In the void between them, fragments of *Navicula* and *Pinnularia* return borrowed diffracted light as faint blue sparks — the scattered remnants of organisms whose shells, once sedimented, will outlast the cells themselves by millions of years.

You hover above a continent-wide plateau of sculptured glass, its surface hammered into parallel ridges by polarized light that reads every nanometer of relief as though raking across cold metal at dusk — this is the valve face of *Pinnularia nobilis*, a pennate diatom roughly 150 micrometers from pole to pole, its frustule cast from amorphous biogenic silica deposited inside living membrane vesicles with molecular precision. The transverse costae march across the full width of the plateau in strict regimentation, each a low ridge of opaline material separated by rectangular bays where the silica thins and the differential interference contrast wavefront shifts from bone-white to dove-grey, while the central sternum runs the entire axial length as a pale luminous highway — the structural backbone from which the raphe slit is incised like a deliberate canyon, its absolute darkness hinting at the nanometer-wide groove through which this living cell extrudes adhesive mucilage to glide across substrates at speeds measured in micrometers per second. Beneath the cool metallic surface, visible through the silica as warmth bleeds through amber glass, two H-shaped chloroplast lobes glow with fucoxanthin-orange biology, and between them the nucleus sits as a frosted ellipsoid of sourceless interior light — two worlds stacked one over the other, the cold mineral architecture of a frustule that will outlast its maker by millions of years enclosing the warm, metabolizing, dividing life that built it.

At one centimeter above the surface, the mudflat has ceased to be mud — it is a continuous burnished sheet of living glass, a biofilm of packed pennate diatoms whose silica frustules press flank to flank across every horizon, each cell acting as a sliver of hammered foil that catches the low morning sun at a grazing angle and throws it back as deep amber and bronze, the whole plain rippling in slow tonal waves where populations have migrated upward en masse to thicken into raised ridges of concentrated pigment and silica that glow richer than the surrounding mat. These organisms are photosynthetic single cells encased in two-part shells of amorphous hydrated silica — opal glass sculpted at the nanoscale into striated vaults and hexagonal pore arrays — and their vertical migration through the extracellular polymer matrix they secrete is a coordinated behavioral response to light and tidal exposure, driven by gliding motility along a raphe slit pressed against the substrate, the whole carpet alive with movement too slow to register as motion but constant nonetheless. Closer in, sinuous Gyrosigma cells curve through the transparent EPS gel like polished mahogany canoes leaving faint mucilage contrails, while sand grains rise as boulders tiled with flattened Cocconeis valves whose regularly spaced areolae — near the wavelength of visible light — scatter the sun into faint spectral fans of blue and gold along their edges. Beads of retreating seawater cling between the biofilm ridges, their curved undersides magnifying the golden carpet beneath into warm amber lenses before wind or evaporation takes them, and at the far distance the mat grades from bright gold into deep olivine where a thin draining film still submerges it, the boundary marking the slow recession of the sea from a surface that has been alive and photosynthesizing here, in this same way, for perhaps two hundred million years.

You are suspended in the amber-green twilight of a freshwater epilimnion, hovering at the tip of one arm of a perfect eight-armed silica rosette that dominates your entire field of view — an *Asterionella formosa* colony, its arms each a slender rod of biogenic amorphous silica tapering like a cathedral buttress, translucent as fire-polished borosilicate glass and edged with thin-film interference halos of pale gold and cool silver where downwelling daylight catches the striated areolae along each valve face. At the colony's centre, a dense amber knot of mucilage polysaccharide binds all eight frustules into their characteristic star geometry, the same biopolymer adhesion that allows colonial diatoms to construct architecturally precise superstructures from individually nanoporous silica shells — each cell wall a two-part epitheca-over-hypotheca assembly deposited in silica deposition vesicles over hours of quiet cellular labour. Two paired chloroplasts run the length of every arm like amber ingots, their fucoxanthin pigmentation glowing warmly through the glass and casting faint brown-gold luminescence into the surrounding water, driving the photosynthesis that makes freshwater epilimnion blooms of this species visible as golden-brown surface flocs to the naked eye. Beyond the colony, soft-focus Staurastrum desmids and cyanobacterial filaments populate the luminous haze of the water column, their presence a reminder that this medium — viscous, tannin-tinged, chemically alive — is a competitive ecosystem in which the Asterionella's silica architecture both concentrates light for photosynthesis and slows sinking through Stokes drag, buying time in the illuminated zone at the cost of a mineral investment that will outlast the cell itself by millions of years.

You are suspended just beyond the wreckage of two spent lives — the paired frustules of *Fragilariopsis* drift to either side like shed armor, their striated silica walls

You are suspended inside a narrow fracture in Antarctic sea ice, its walls rising around you like the inner faces of a geode cut from polar glass — each crystallographic plane a slightly different angle, splintering the dim light filtering down from the surface into overlapping wedges of cobalt, pale violet, and cold silver. The channel exists because brine, excluded as seawater freezes, concentrates into these sinuous veins where the salt lowers the freezing point enough to keep liquid water at –2°C, creating a hypersaline microhabitat whose slightly viscous fluid carries a faint amber optical warmth against the surrounding blue ice. Drifting through this enclosed world are ribbon-chains of *Fragilariopsis cylindrus*, each cell an 8-micron silica cylinder so glassy it nearly vanishes, made visible only by the saffron-bronze chloroplasts burning inside like compressed embers — fucoxanthin pigments shifted toward warmer wavelengths to harvest the dim, blue-attenuated polar light reaching this depth. Around each chain, an invisible sheath of antifreeze exopolysaccharide gel hazes the frustule outlines and anchors gossamer mucilage threads to the ice-crystal walls, the biological strategy that keeps these organisms locked within the channel rather than flushed into open water. Below, the channel floor is stained dark amber-bronze by generations of accumulated diatom biomass — a compressed living mat that marks this frozen architecture as something more than geology: a vertical canyon of cold light and biological warmth, microhabitat and organism co-constructed at the boundary of ice and liquid.

You drift above a tilted world of glass, its surface curving away in every direction like the hull of an enormous buried planet, tessellated from edge to invisible edge with thousands of perfect hexagonal pits — each one a six-sided shaft dropping two hundred nanometers into absolute silica shadow, their velum meshwork floors visible only as ghostly lattices revealed by the low-raking electron-beam illumination that turns every ridge into blazing white and every interior into matte black. This is the valve face of *Coscinodiscus wailesii*, a centric diatom, and what reads as monumental architecture is the organism's frustule: a shell of amorphous biogenic opal deposited with nanometer precision inside a silica deposition vesicle during the cell's division cycle, its areola geometry encoding taxonomic identity in the very periodicity of the hexagonal array. From the plain, fultoportulae towers rise at irregular intervals — hollow silica columns open at their bores, each flanked by three satellite pores through which the cell once secreted chitin fibrils outward into the water column, their directional shadows sweeping across the hexagonal field like sundials marking a frozen moment. At the valve margin, rimoportulae appear as dark elongated slits cut cleanly through the silica precipice, mechanosensory structures whose inner chambers once connected cytoplasm to sea, while below the mantle's curve the smooth cingulum bands begin — featureless hoops of silica receding into depth, the plain girdle holding epitheca to hypotheca in a joint refined across two hundred million years of evolutionary pressure.

You hover a hundred micrometers above the surface of a giant kelp blade, *Macrocystis pyrifera*, which spreads beneath you like an undulating continent of olive and amber — its enormous plant cells tessellating the surface in a waxy, translucent mosaic, the whole landscape trembling as slow caustic curtains of blue-green shallow-water light sweep through the column above and set every surface flickering between gold and shadow. This blade is not bare: a layered biofilm civilization coats it in pale amber extracellular polysaccharide and mucilage, a barely visible matrix that catches low-angle light with a faint iridescent sheen, within which several distinct diatom species have colonized every available niche of substrate, light, and flow regime. Dominating the middle distance, *Licmophora* colonies rise on slender mucilage stalks — each a perfect flabellate fan of twenty to forty elongate silica frustules arranged in golden-brown arcs thirty to sixty micrometers wide, their finely striated valves glowing warm amber and throwing interference colors of teal and copper as caustic light refracts through them, swaying almost imperceptibly in the slow surge current like a microscopic forest echoing the kelp canopy far above. Between them, *Cocconeis* valves lie flush and nearly invisible against the kelp cells — elliptical silica shields pressed tight to the substrate by their own raphe system, camouflaged in the green-brown EPS — while *Rhabdonema* ribbon bundles coil in loose zigzag spirals through the mid-ground, their bright silver-gold edges catching the light against the darker matrix. Cutting across the background, a pale scrape scar exposes raw kelp surface where a grazing amphipod — crouching at the far margin of its own devastation, enormous at this scale, a mountain of jointed translucent armor — is still working its mandibles through the biofilm fringe, sending slow pressure waves through the water that set every *Licmophora* fan trembling in unison, golden antennae tuned to the same ancient, animal frequency.

You are suspended inside a vaulted calcite chamber roughly the width of a human hair, the curved cream-white walls rising around you like the nave of a Romanesque cathedral, their surfaces faintly granular where individual calcite crystals form a mosaic of buff and ivory at this resolution. The calcite is softly translucent rather than opaque, transmitting a diffuse warm glow from the sunlit carbonate sand world outside, so the chamber seems lit from within its own walls — a sourceless ambient luminescence that edges every surface with a pearlescent halo and turns the dark pore openings into pure black archways through which pale pseudopod threads extend like drawn glass filaments into the open seawater beyond. This is the interior of an Amphistegina foraminifera test, a single calcified chamber among many in the multi-chambered shell architecture of this benthic protist, and the pale beige-gray cytoplasm filling the lower space is not inert — it is a living gel maintaining active pseudopodial networks that exchange gases, nutrients, and signals with the surrounding reef environment. Settled throughout this cytoplasm like passengers at rest are the Nitzschia symbiont diatoms, each one a slim pennate frustule of biogenic silica 20–40 micrometers long, their valve margins returning the filtered light as a barely-there silver-blue interference shimmer, while their interiors glow with the deep amber warmth of fucoxanthin-rich chloroplasts — broad lobes of photosynthetic pigment the color of old honey held to candlelight, sequestered here in non-motile productive calm, sheltered from the turbulence of the water column by walls of stone the foraminifera built around itself one ion at a time.
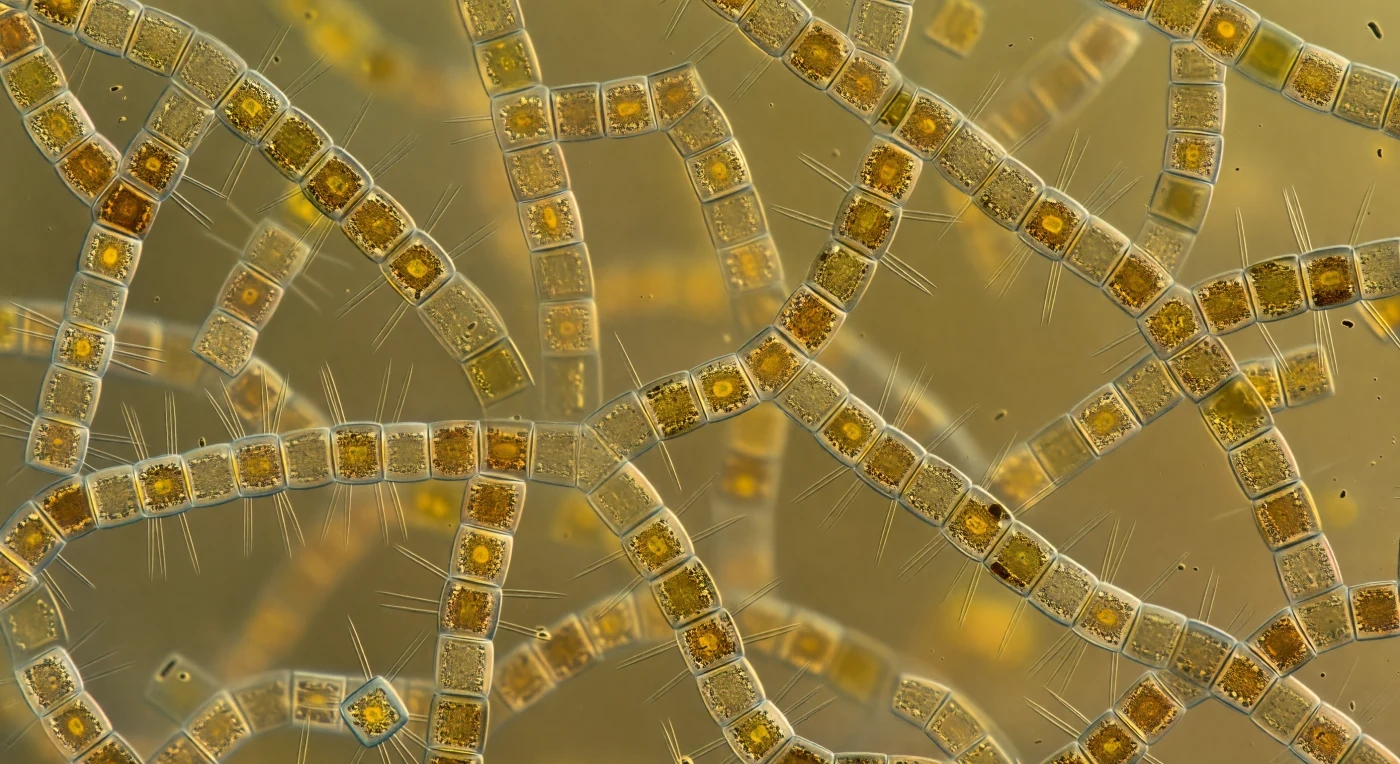
You are suspended inside a living fog of glass and gold, every direction occupied by cylindrical silica towers — *Skeletonema costatum* cells linked end-to-end by radiating arrays of fultoportula struts, each rod a needle of cold clarity catching diffuse light against the warm amber haze that fills every sightline. The illumination has no source: surface sunlight, filtered down through millions of fucoxanthin-laden chloroplasts, has been scattered, partially absorbed, and re-emitted so many times that the water column itself glows from within, a saturated amber-green radiance arriving equally from all directions and eliminating shadow almost entirely. Visibility collapses within half a millimeter, distant chains dissolving into the luminous haze before they can resolve, because the medium between them is not open water but a colloidal soup of extracellular polysaccharides, dissolved pigment, and bacterial cells — dark rod- and comma-shaped forms coating nearly every frustule surface in a dense organic mat that absorbs light where clean silica would scatter it. This is a spring bloom at its peak: a community of single-celled photosynthesizers dividing every twelve to thirty-six hours, knitting the upper ocean into an open ribcage lattice of biogenic silica that stretches, without interruption, in every direction — claustrophobic, gorgeous, alive in every cubic micrometer.

You drift centimeters above the Southern Ocean floor at three thousand meters depth, where the world resolves into a pale grey-beige plain built not from rock or sand but from the accumulated silica and carbonate remains of a hundred million years of surface blooms — a sediment called diatomaceous ooze, composed almost entirely of biogenic opal and foraminifera tests settling through the water column in a slow, perpetual snow. Tilted at random angles from this surface, intact *Coscinodiscus* frustules protrude like translucent manhole covers half-buried in ash, their concentric rings of hexagonally packed areolae still geometrically perfect, the amorphous hydrated silica of each valve catching interference colors — pale aquamarine, faded gold, cold ivory — in a world where no sunlight has ever reached; between them, *Eucampia* girdle-band fragments and feather-edged pennate valves of *Fragilariopsis* show the work of corrosive abyssal water operating just above the lysocline, their margins dissolved into lacy gradients where structured glass becomes suggestion, while white foraminifera tests sit like small architectural ruins among the siliceous material, some intact, some already collapsing into chalky stain. From several points embedded in the ooze, slow pulses of cold blue bioluminescence bloom and die in the bacterial colonies metabolizing within the sediment matrix, their light diffusing upward through overlying frustule valves and briefly illuminating the areolae like fiber-optic arrays before darkness reasserts itself. Far toward the haze-limit of visible depth, a pale plume rises in a soundless column where a polychaete moves beneath the surface, its passage lifting decades of delicate accumulation into a slow-motion cloud of frustule fragments and carbonate dust that drifts upward through the dark water, each tumbling shard of diatom valve catching a blue bacterial flicker for a fraction of a second before the column of darkness swallows it completely.
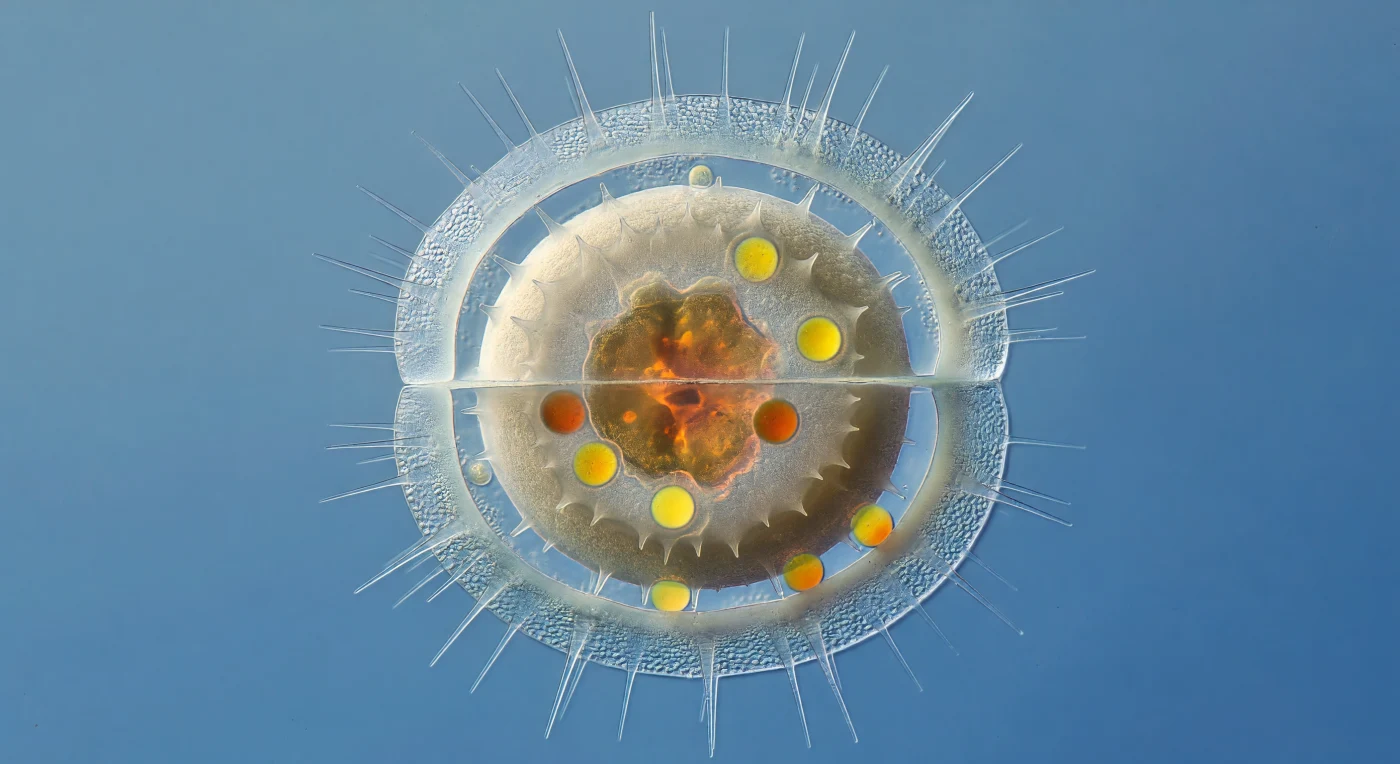
You are suspended ten micrometers from the outer wall of a dying *Chaetoceros* frustule, close enough that its long hollow setae radiate past you like the spokes of a shattered chandelier, each one a tapering needle of nearly vanished silica scattering ambient blue-green light into faint spectral halos. The parent cell's walls have thinned to gossamer — hydrated amorphous opal barely a hundred nanometers thick, its areolae bleached of interference color, the whole cylindrical lantern form held together more by geometric habit than by any remaining structural integrity, its copulae rings splayed slightly outward by the pressure of what swells within. Inside, the resting spore fills the dying shell the way a river stone fills a soap bubble: two full micrometers of granular amorphous silica stacked in concentric laminae, its surface roughened with blunt hexagonally clustered spines, its color the matte warm ivory of unglazed ceramic — opaque where the parent frustule is ghostly, dense where the outer walls are dissolving back into the silicic acid they borrowed from the sea. And yet sealed within that heavy ceramic vault, visible only because the thinning parent transmits light like a paper lantern, the interior of the spore glows: a collapsed lobe of deep amber chloroplast tissue — fucoxanthin concentrated to near-opacity — surrounded by brilliant cadmium-to-ember lipid droplets, each one a bead of stored solar energy pressing gently against the thick inner wall, the entire living treasure radiating warmth against the cold blue oceanic water fading to deep cobalt at the edge of the field.

You are suspended within the pale mineral body of a Miocene diatomite, enclosed on every side by ten million years of compressed biological architecture rendered in silica and stone. The dominant structures around you are the frustules of countless diatom species — *Melosira* chains stacked as coin-columns of pale opal-A silica, each valve's concentric radial striae still resolved at microscopic precision; a massive *Stephanopyxis* disc occupying the field like a cathedral wall, its honeycomb loculi preserved in cross-section as chambered voids set into white stone. Between and around these forms, void pore spaces — once pathways for interstitial water during early diagenesis — appear as absolute cold darkness, the negative space that makes the surrounding silica matrix glow by contrast with an ivory, sourceless luminescence. This rock is diatomite: a sedimentary deposit formed when biogenic silica accumulated faster than it dissolved, the frustules compacting under burial pressure but retaining taxonomically identifiable ultrastructure across geological time, their amorphous hydrated silica slowly recrystallizing yet never fully erasing the biological geometry encoded within. Nearby, the smooth translucent curve of a fossil fish scale redirects the diffuse cold light into a faint ochre warmth — the only organic remnant in a world otherwise given entirely to mineral silence, every surface a record of organisms that lived, sank, and became the ground itself.


















