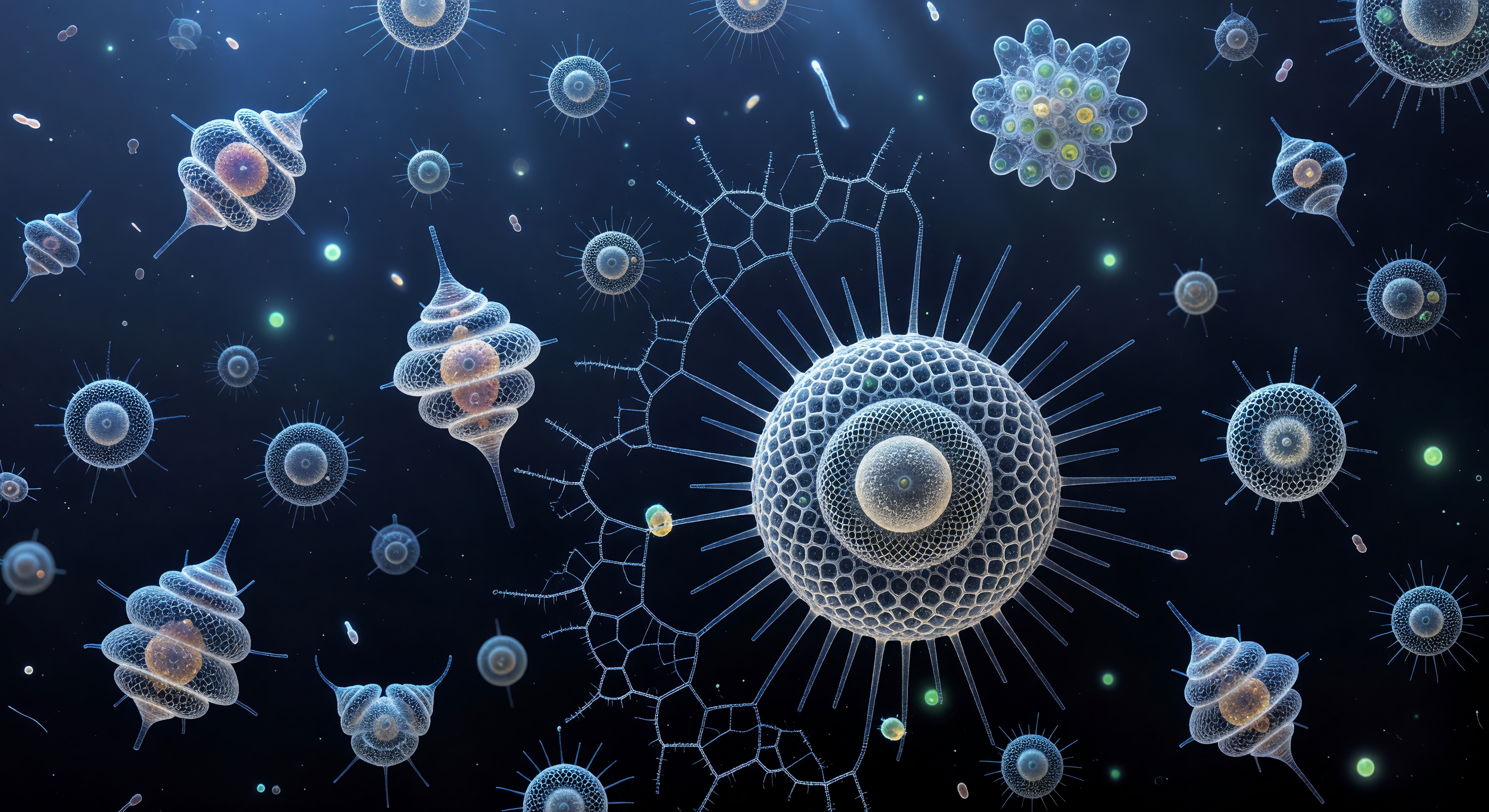
You are suspended at the geometric center of a living mineral cathedral, looking outward through three concentric shells of amorphous opaline silica — the skeleton of *Actinomma asteracanthion*, a spherical radiolarian no wider than a fine human hair. Each shell is a precision lattice of hexagonal pores, each pore a fraction of a bacterium across, and together they nest like Russian dolls of frozen light, their silica struts refracting the cold deep-ocean blue into prismatic halos — pale violet at the margins, warm aquamarine at the center — so that the entire structure reads as a luminous stained-glass vault built at cellular scale. Behind you, the endoplasm glows with a subdued amber warmth, the nucleus and lipid-rich cytoplasm of a living protist radiating honey-colored light that softens the geometry, while twelve triradiate spines — each a crystalline fiber of biogenic silica secreted atom by atom inside membrane-bound vesicles — drive outward through all three lattices and vanish into the indigo haze of the deep ocean beyond. Through every hexagonal pore, the abyss outside resolves into a slightly different refracted window, multiplied across hundreds of tiny lenses, so that depth is felt not as distance but as layering — three superimposed geometric shadows drifting in moiré offset, the progressive darkening of blue between each shell, and the long receding perspective of spines that seem to reach toward a horizon the eye can never quite reach. This is the Stokes regime, where viscosity rules over gravity and inertia, where Brownian jostling is the dominant motion and a drifting bacterium is both landscape feature and prey, and where one of Earth's oldest continuous lineages — half a billion years of siliceous architecture in the fossil record — has solved the problem of life in open water with geometry alone.

You are suspended roughly two hundred micrometers from the nearest surface of a living Acantharia, a distance that at this scale feels like standing at the edge of a cathedral plaza before an illuminated rose window — and the organism before you fills the entire field of view like a jeweled lantern breathing in cold seawater. At its heart, the central capsule presents as a warm amber-brown ovoid, nearly opaque with the massed bodies of zooxanthellae pressed against its inner wall, their chlorophyll pigments reradiating a faint russet halo of tobacco and gold into the surrounding darkness; from this dense nucleus, twenty spines of strontium sulfate — celestite, a mineral almost unknown in living tissue — radiate outward in precise geometric array, polar and equatorial and tropical positions locked by the Müller's law symmetry unique to this group. Under crossed polarized illumination, each crystalline rod has ignited in a distinct interference color determined by its optical path length and crystallographic orientation: one burns electric cyan along its entire length, its neighbor a saturated fuchsia-magenta, the next a deep cobalt blue fading to violet at the needle-sharp tip, then gold, then lime-green, the birefringence of the sulfate lattice splitting incident light into ordinary and extraordinary rays that recombine at the analyzer to produce colors as saturated as neon against the near-absolute black of the surrounding water. Between the blazing spines, the myoneme contractile cables — actin-based biological guy-wires unique to Acantharia, capable of collapsing the entire spine array to alter buoyancy — run as taut dark threads, their organic composition rendering them invisible to the polarized light that makes everything crystalline incandescent, while at the very periphery of the scene the axopodia extend as ghost-thin silver-white lines into the water column, their microtubule axonemes quivering in the Brownian current, broadcasting the organism's presence into the surrounding broth of dissolved salts and suspended colloids like the trembling strings of an instrument no one is playing.

You stand on a mineral world no wider than a human hair, its floor an endless repeating grammar of hexagonal and pentagonal panes — the outer silica lattice of a living Spumellarian radiolarian, each strut where opaline glass thickens glowing faintly amber, each node catching oblique light as a hard silver point above a razor shadow. Through the dark pore-voids beneath your feet, the cell's intracapsular calymma trembles in pale gold: a vacuolated, living foam in constant submicron motion, the metabolic interior of an organism that has been building such lattices, dissolving them back into the sea, and building them again for over half a billion years. Rising from the pore rims and lattice nodes all around you, a dense forest of axopodia climbs into the near-black liquid overhead — each rod a micron-wide bundle of crystalline microtubule arrays wrapped in glassy membrane, its flanks carrying thin interference fringes of teal and amber where DIC illumination splits around its curvature, the whole forest converging toward tips that dissolve into the diffuse haze of protein-laden seawater fifty microns above. Along three of the nearest shafts, amber vacuoles — membrane-wrapped parcels of captured prey — creep steadily inward at one to five microns per second, drawn by cytoplasmic streaming along the microtubule track inside, each one a warm topaz lantern against absolute cold darkness, carrying digested ocean downward into the architecture below.

You are suspended inside the skeletal architecture of a single-celled organism no wider than a human hair, within a vaulted tunnel of biogenic opal silica — amorphous hydrated silicon dioxide secreted by the cell itself, one molecular layer at a time, into a precise geometry that took hours to mineralize. Around you, the latticed walls of *Eucyrtidium calvertense* curve inward at arm's reach, their elliptical pores arranged in spiraling registers, each aperture flooding the interior with aquamarine ocean light that refracts through the translucent silica into trembling caustic rings, turning the chamber into a shifting mosaic of cold blue-green fire. Above you, five successive septa stack in forced perspective — annular walls of the same frosted opal, each pierced by a central foramen that funnels and concentrates the incoming light, the openings diminishing upward until the fifth is a burning pinprick of turquoise brilliance, above which the apical spine rises as a needle of dense silica, silhouetted in perfect black against the open water column beyond. The extracapsular cytoplasm that coats every inner strut in a barely visible gelatinous film is not decoration but function — axopodia thread outward through these very pores into the surrounding ocean, stiffened by crystalline microtubule bundles, hunting bacteria and dissolved organics in a fluid regime so viscous at this scale that inertia is meaningless and every movement is governed entirely by drag. This is the interior of a skeleton built by a living cell, in a world where gravity barely registers, where Brownian motion is the background noise of existence, and where the entire cathedral around you will one day sink through kilometers of open ocean to join half a billion years of radiolarian fossils on the seafloor below.

You are suspended above a vast grey plain that extends in every direction without relief, its surface packed so densely with the silica remains of radiolarians that it reads less as sediment than as a ruined city compressed flat by geological time. Under the oblique electron beam, intact Spumellarian tests loom like geodesic dome complexes, their nested concentric lattices casting intricate grids of shadow-pits into their own interiors, each hexagonal pore framing a doorway-sized rectangle of absolute black, while broken Nassellarian cones lie overturned among them like collapsed cooling towers, their apical spines still lancing upward through the mineral fill at odd angles. Amorphous clay drifts in smooth accumulations around the bases of intact tests, half-burying some and leaving others elevated on compacted pedestals, the whole surface recording in monochrome physics — electron contrast alone, no colour, no atmosphere — the compression of millions of years of pelagic snowfall into a few centimeters of ooze. The silica of each test is opaline and biogenic, secreted by living cells from dissolved silicic acid over hours of slow mineral accretion, and what survives here is only the most durable fraction: forms whose lattice geometry was strong enough to resist dissolution in the undersaturated deep water during the centuries of sinking and burial. Behind the nearest landmarks, the raking beam loses its power to lift geometry from shadow, and the far field dissolves into undifferentiated grey, a necropolis extending beyond resolution in every direction.
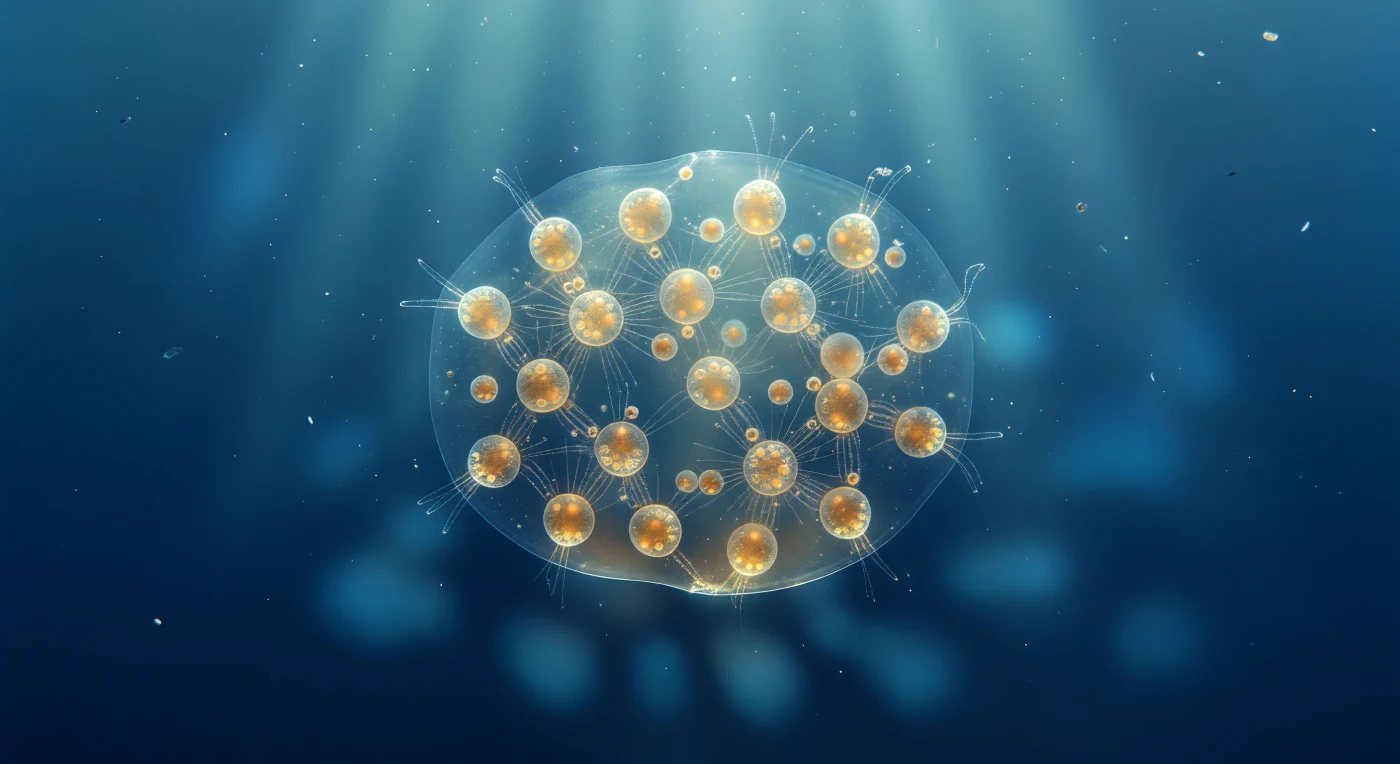
You are suspended one millimeter from the surface of a colonial Sphaerozoum, close enough that the entire colony fills your field of view like a softly luminous planet hanging in open blue water — its eight-millimeter ellipsoidal body so nearly transparent that the boundary between organism and ocean registers only as the faintest shimmer, as though a lens of thickened seawater has quietly organized itself around a community of living amber lanterns. Each individual cell glows a warm golden-brown from the dense population of symbiotic dinoflagellates packed within its ectoplasmic calymma, and from every cell, axopodia radiate outward as whisker-thin filaments — invisible until a shifting column of tropical sunlight catches them at angle and flares them into silver needles, their microtubule axonemes forming a loosely interlocking mesh that threads the whole gelatinous interior. This colony belongs to the silica-free collodarians, and without a hard mineral skeleton, there is no gothic lattice here — only soft, buoyancy-loaded biology: lipid vacuoles scattering light as milky iridescent beads within each cell, and the mucilage boundary itself acting as a weak biological lens, converging downwelling rays into a slowly drifting caustic of brighter and darker blue that shimmers across the water column below like a stained-glass window cast adrift. The surrounding ocean is an almost structureless blue void of near-infinite depth, its fine suspended particles of marine snow — a glinting diatom frustule, a tumbling organic aggregate — only emphasizing the colony's extraordinary stillness, a self-contained microscopic biome held aloft by its own buoyancy and caught between total invisibility and warm interior radiance in an immense, open silence.
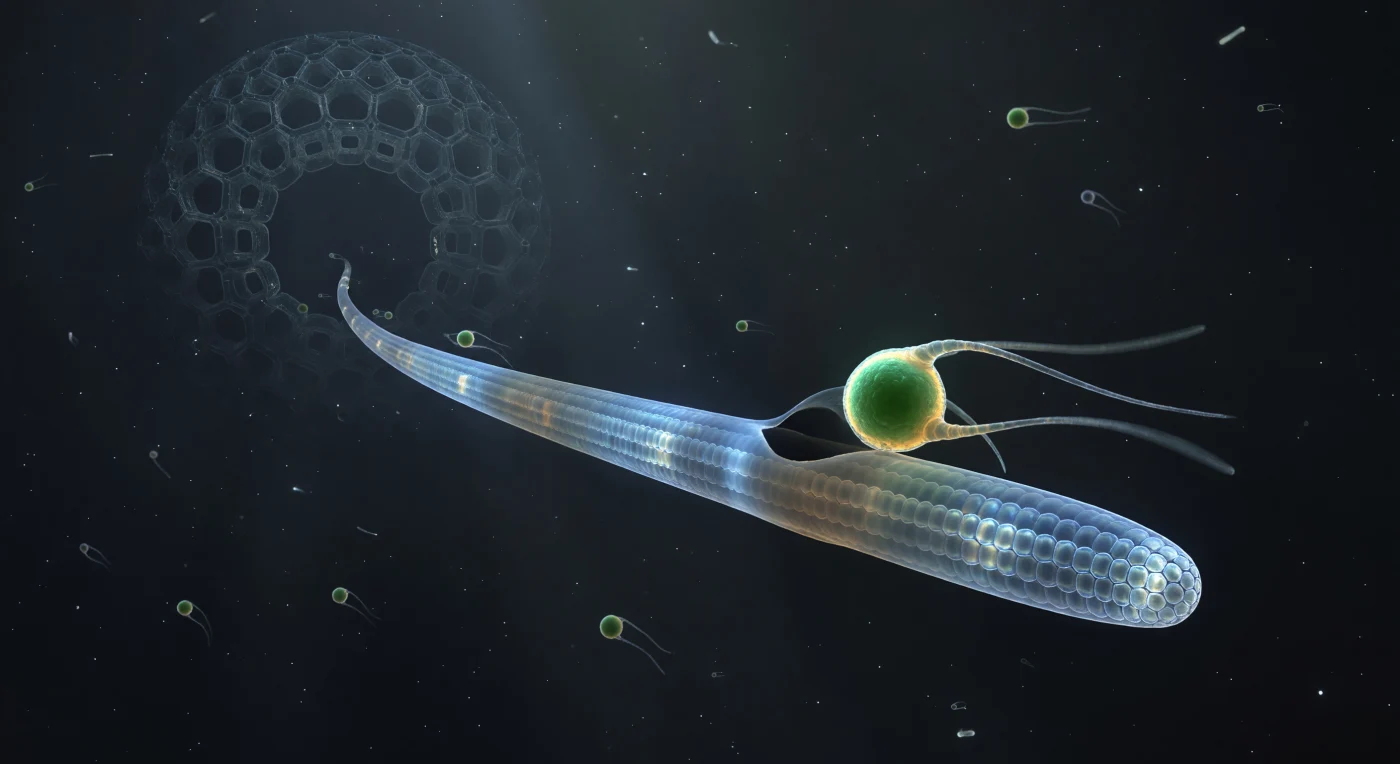
You are suspended at the threshold of a predatory act frozen in time — a crystalline axopodium extends across your entire field of view like a translucent architectural beam, its interior honeycomb of parallel microtubule bundles catching diffracted light and casting cold prismatic halos along its length, while at the point of contact a vivid emerald sphere barely three microns across blazes against the charcoal deep like a small incandescent planet, its two flagella halted mid-beat in arcs of pure filament-light. This is a radiolarian axopodium in the first instant of prey capture: the stiff pseudopodium is braced internally by a crystalline axoneme of bundled microtubules running in precise geometric arrays, and where the flagellate — a Micromonas, one of the ocean's smallest photosynthesizers — has made contact, the glassy axopodial membrane has dimpled inward and begun extruding a dark crescent of new membrane that curls around the green sphere like the opening degree of a slow eclipse. The cytoplasm at the contact zone has shifted from cool glass-blue to a warm turbid amber, betraying the biochemical cascade already triggered: adhesive surface molecules have fired, the engulfment machinery is committed, and the flagellate's fate is decided even as its chloroplast burns at full autofluorescent intensity. Deeper along the axopodium, toward the vaulted silica cathedral of the central capsule barely resolved in the background darkness, earlier vacuolated prey drift inward like dimming lanterns, their pigments faded to rust — evidence that this moment of luminous contact is merely the latest event in a continuous, patient harvest conducted entirely below the threshold of unaided vision, in the sunlit upper ocean.
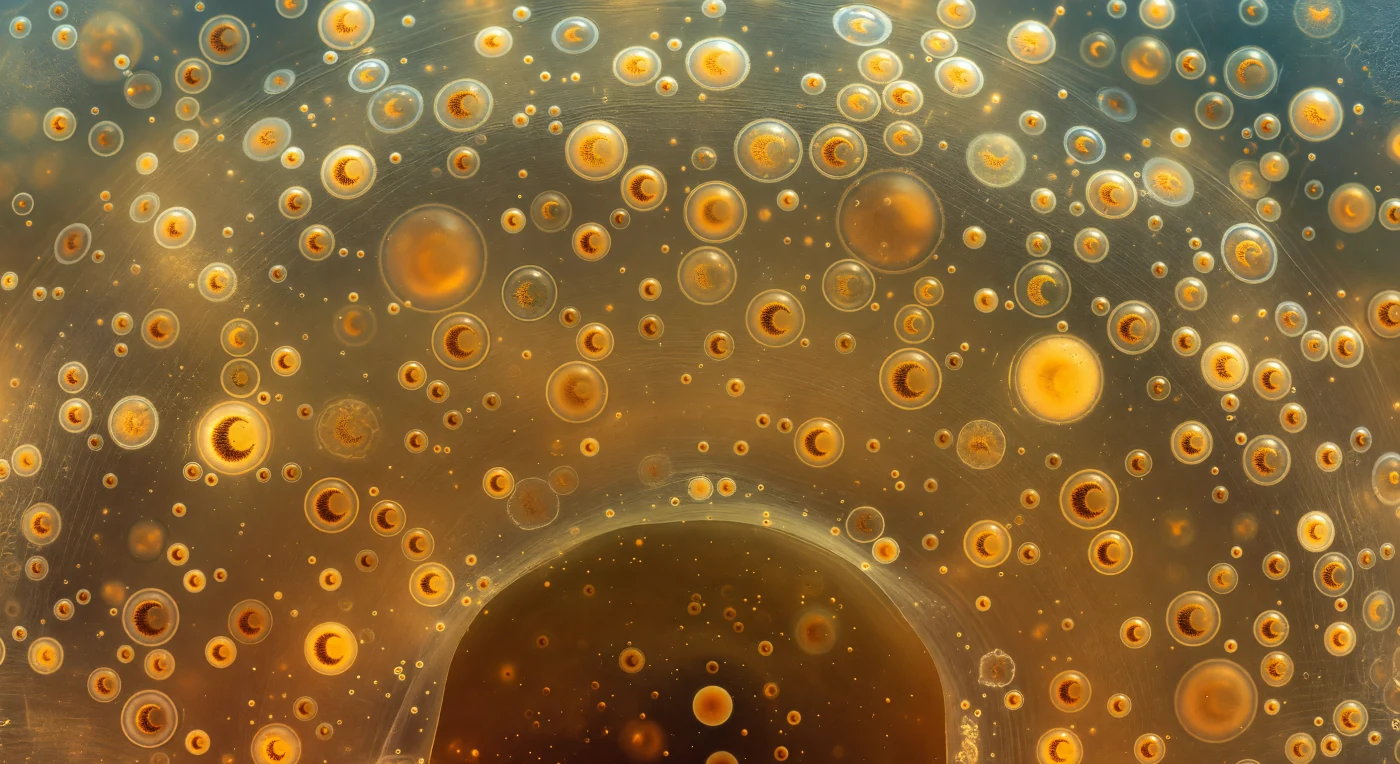
You are suspended within the living interior of a colonial Collodarian's calymma, a trembling amber gel that presses around you with the soft, total intimacy of warm honey — neither liquid nor solid, but a viscoelastic ectoplasm threaded through with barely visible glycoprotein filaments that catch the light like frost on glass. In every direction, zooxanthellae hang motionless in suspension, warm golden-brown spheres of ten to fifteen microns each, stabilized against settling by the gel's own resistance to flow and quivering only with the ceaseless nanometer-scale whisper of Brownian motion; within the nearest cells, C-shaped dinokaryotic nuclei curve like amber crescents, their condensed chromosomes permanently visible as a consequence of the permanently condensed state unique to dinoflagellate chromatin. Scattered among the symbionts, lipid vacuoles swell to thirty or forty microns — enormous clear spheres that refract the ambient light into bright crescents and cast golden caustic rings onto whatever floats behind them, serving the colony as both buoyancy organs and light-concentrating lenses that funnel photons deeper into the symbiont cloud. Far above, diffuse ocean light filters down through the colonial matrix and is transformed layer by layer into the deep amber luminescence filling this space, the photosynthetic pigments of countless zooxanthellae — fucoxanthin, peridinin, chlorophyll — absorbing the blue and remitting chemical energy that flows outward as carbon fixed in the sun-warmed gel. The curved wall of the central capsule looms at the edge of visibility, a sealed organic membrane where ectoplasm ends and endoplasm begins, enclosing the nucleus and silica-deposition machinery in a world you cannot enter — a boundary between two cellular continents, dark and absolute.
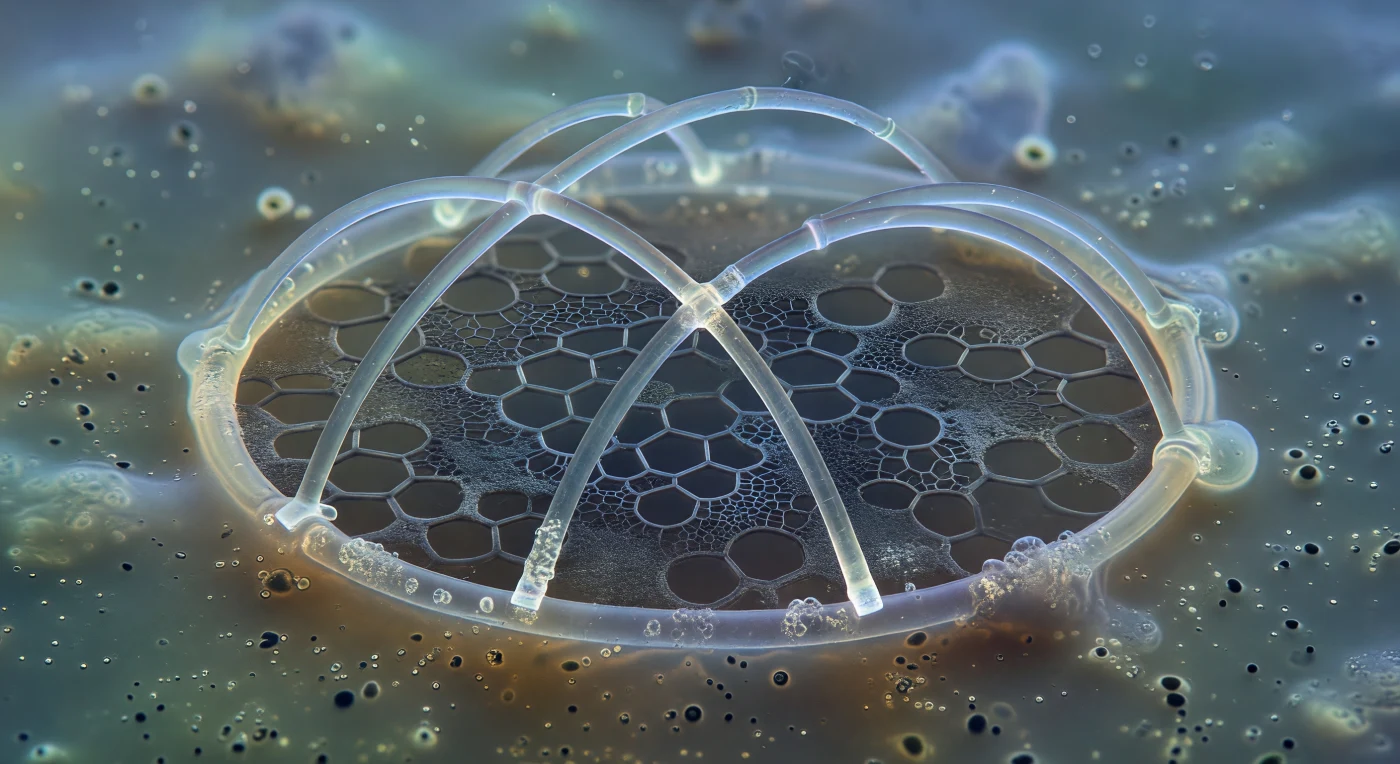
You drift within a medium so thick with biological density that motion itself seems suspended, watching the earliest moments of a skeleton being born. Before you, the nascent sagittal ring of a *Pterocorys* rises in luminous glass arcs — silica no thicker than a few microns already hard and transparent, edged with cold iridescent halos where phase contrast plays along its curves, two primary bars crossing its interior like the first ribs of a cathedral only just begun. Beneath the mineral, barely perceptible against the granular amber-brown cytoplasm, a matte gray meshwork of glycoprotein fibers spreads in uncanny geometric patience — the organic template that precedes and directs silicification, its hexagonal pore outlines already inscribed in soft matter, empty and waiting for the mineral that will follow. This pre-skeletal scaffolding is secreted within silica deposition vesicles, intracellular membrane-bound compartments where seawater-derived silicic acid polymerizes into amorphous opal under precise biochemical control, the growing test never touching open cytoplasm directly. The ring reads as an immense architectural member from this vantage — its curvature implying vast enclosed space — and the silica gleaming with the patient permanence of a structure that, once complete, will outlast the cell that built it by half a billion years.

Pressed flat against the interior surface of a silica deposition vesicle, you are witnessing the slow, irreversible act of mineral construction — amorphous silica gel advancing in a pale grey-white mass across a rust-orange lattice of silaffin-like protein fibrils, burying the scaffold strand by strand as silicic acid polymerizes into the hydrated glass that will eventually become a radiolarian's intricate test. The silicalemma membrane curves overhead as a razor-precise double line of darkness, the boundary between this tightly controlled biomineralization chamber and the amber cytoplasm beyond, where cobalt-blue mitochondria and emerald-green granules glow like lamps behind frosted glass. The scaffold beneath you is not passive scaffolding but an active template — the silaffin proteins carry the polyelectrolyte chemistry that catalyzes silica precipitation, drawing dissolved silicic acid out of the vesicle fluid and locking it into solid mineral along their surfaces, the fibres darkening from terracotta to deep sienna as mineral encases them. Where the gel front is thinnest its translucency still permits a glimpse of the compressed protein lattice below, the gradient of encroachment as unhurried and inevitable as a tide darkening sand. The entire space — enclosed, amber-lit, saturated with the diffuse cream luminosity filtering through the membrane above — is a foundry operating at the threshold of chemistry and geometry, building one of Earth's most exquisite mineral architectures one nanometer-thick silica layer at a time.

You stand on the outer shell of a radiolarian — a single-celled planktonic organism no wider than a few human hairs — its surface rendered in the cold, sourceless illumination of a scanning electron microscope as a vast curved plain of amorphous opal silica, ash-gray and faintly vitreous, the horizon bending away in every direction with the undeniable curvature of something spherical and very small. The ground beneath you is not ground at all but a geometric lattice: a near-perfect hexagonal tessellation of raised silica bars three micrometers wide, each framing a pore roughly fifteen micrometers across that plunges into absolute blackness, so that more of the surface is void than solid and you are walking a web of bridges over abyssal wells. Six triradiate spines erupt from the lattice at equidistant nodes, tapering to needlepoints forty or fifty micrometers overhead — at this scale, structures the height of skyscrapers, their lit edges catching a single silver line of highlight while their shadow faces dissolve into matte darkness. This skeleton, secreted by a living cell through a slow intracellular process of silicic acid polymerization inside dedicated membrane vesicles, is not crystal but biogenic amorphous silica — opal — precipitated with a geometric precision that no purely chemical process could achieve, the organism imposing biological order on mineral chaos. Through the pores, if you angle your gaze downward, you can just discern the suggestion of an inner concentric shell, its own hexagonal lattice misaligned with the outer one, a moiré of overlapping voids hinting at the nested architecture below — the radiolarian building its test not as a single wall but as a series of concentric spheres, each a new mineral commitment laid down over hours of slow, silent accretion in the open ocean.

You are suspended in a column of water so cold it registers in the body as pressure rather than temperature, and around you, in every direction, the mesopelagic zone is conducting its quiet architecture. Nassellarian tests descend on all sides — conical helmets, stacked pagoda-chambers, elongated multi-segmented forms between one hundred and four hundred micrometers across — their amorphous opal silica lattices catching the last attenuated photons from the surface world and breaking them into cold blue-grey glints along every pore-edge and radial bar, each skeleton a cathedral of spun mineral that took its occupant days to deposit one silica vesicle at a time. Where cells still live, amber and tawny cytoplasm presses warm against the inner lattice walls, leaking through hexagonal pore-arrays as faint biological light — stained-glass lanterns adrift in near-total darkness — while the many empty tests beside them read as pale silver-grey, their hollow chambers open to seawater, cold and eyeless. Between these sinking forms, marine snow flocs — transparent aggregates of shed mucus, dead organic matter, and released radiolarian cytoplasm — drift in slow spiral paths through the deep indigo water column, occasionally snagging on a protruding silica spine before releasing, their semi-luminous surfaces catching ambient bioluminescence from somewhere unseen and passing it along as a diffuse pearlescent shimmer. This is the biological pump made visible: each test that sinks carries sequestered silica and organic carbon toward the abyssal floor, a millimeter-scale freight contributing to the planetary-scale transfer of matter from the sunlit ocean surface to the sediment record below, where after tens of millions of years it will compress into radiolarite and re-enter the geochemical cycle of the Earth.

The field before you is an ancient seafloor rendered in stone — a dense, interlocking mosaic of recrystallized silica tests pressing against one another in every direction, their original spherical and conical geometries still legible as ghost-forms beneath 150 million years of burial and mineral replacement. These are the compressed remains of radiolarians, single-celled planktonic organisms whose intricate silica skeletons once drifted through a Jurassic ocean that has long since vanished, accumulating on the seafloor over millions of years before being entombed, pressurized, and transformed into radiolarite chert. Transmitted light floods upward through the section in a cold, diffuse wash, suffusing the pale chalcedony and microcrystalline quartz of each recrystallized test with a faint phosphorescent glow — ash-white and bone-cream forms welded seamlessly to their neighbors, the delicate pore lattices of living architecture now flattened into subtle surface relief by lithostatic pressure. Between the tests, hematite-stained cements bleed rust-orange and dried-blood crimson through the interstices, tracing the ghost geometry of pore spaces once filled with seawater, now mineralized into iron-veined amber gradients that pool and trail like rust-water through cracked stone. The world extends in all directions as an infinite compressed archive — denser and more opaque at center, dissolving toward the periphery into granular translucence — the entire visible field a planetary surface made of fossilized plankton, an ocean's biological productivity frozen into mineral silence.

You float within an absolute blackness that reads more like deep space than ocean water — the crossed polarizing filters have erased all ambient light, leaving only what the mineral architecture itself chooses to radiate. Scattered across this darkness in every direction, Acantharia cells detonate like suspended fireworks: each of the twenty strontium sulfate spines fanning outward from a single organism functions as a birefringent rod, converting polarized illumination into blazing interference color, so that one cell burns in magenta and gold while its neighbor erupts entirely in electric teal and cobalt, the icosahedral symmetry of their spine arrangements mapping each cell's chromatic identity as precisely as a fingerprint. Between these chromatic explosions, the amorphous opaline silica of Spumellaria tests surrenders nothing to the polarized field, instead appearing as cold pewter lattice-spheres — nested concentric cages of hexagonal pores connected by radial struts, readable in exquisite structural detail yet chromatically inert, haunting the dark between the Acantharia like Gothic cathedral ruins preserved in silver fog. The strontium sulfate composing the Acantharia spines is itself a rarity: unlike the silica skeletons built by virtually all other radiolarian lineages, it is unstable in seawater and dissolves almost completely within days of the cell's death, meaning these incandescent forms leave almost no fossil record — what burns so brilliantly here vanishes without geological trace.

You hover in absolute darkness two thousand meters below the surface, your entire existence compressed to five hundred microns of cold saltwater, and a narrow cone of blue-white light cuts through water so clear it has no color — only depth — until *Aulacantha scolymantha* resolves at the beam's edge, its five hundred hollow siliceous spines fanning outward in every direction like the arrested explosion of a glass chandelier, each translucent tube conducting your spotlight inward and leaking it back out at the distal tips as cold fiber-optic pinpoints, a spherical constellation of pale blue-green coordinates scattered across otherwise empty water. At the organism's core the phaeodium sits like a bruise — an opaque, dark-brown mass of partially digested material dense enough to block light entirely, giving the cell an unsettling interior weight, while around it the capsular wall is barely legible as momentary silver threads of opaline mineral, architecture present only as inference and glint, the frothy ectoplasm beyond studded with vacuoles that scatter the beam into a soft aureole of diffuse cold luminescence between the dark nucleus and the radiating spine-field. This is a phaeodarian radiolarian, belonging to a lineage that exchanged the pristine mineral lattices of its polycystine relatives for this darker, more organic body plan — the phaeodium itself a uniquely derived organelle, a communal waste-and-reserve mass shared across the extracapsular cytoplasm, the hollow spines not merely skeletal struts but functional light guides evolved in a world where the only photons are bioluminescent and therefore worth capturing. The void beyond the illuminated halo is not merely dark but structurally dark, a material the light tests and fails to enter, the beam falling off into black with no scattering horizon and no floor, the glowing spine-tips the only coordinates in a space that otherwise refuses to be measured.
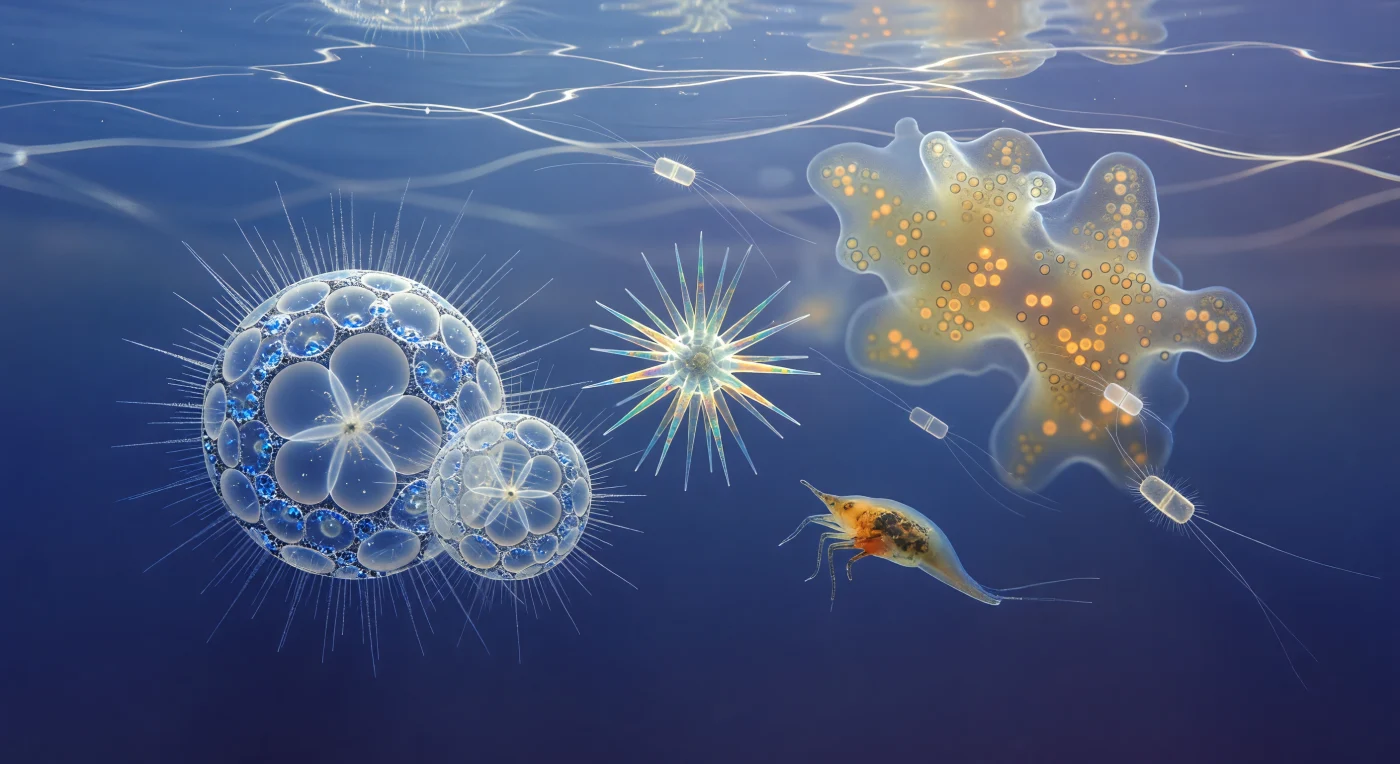
You are suspended thirty meters below the tropical Pacific surface, scaled down to the size of a single radiolarian cell, adrift in a luminous cerulean void where gravity has surrendered its authority to viscosity and diffuse light floods in from every direction above like a slow celestial broadcast. Around you, Sphaerozoum individuals hang in the water like paired cathedrals of opal silica, their nested lattice spheres catching the restless caustic nets drifting down from the surface and fracturing them into cold prismatic sparks at each hexagonal pore, while their axopodial halos — each filament a crystalline microtubule axoneme thinner than a bacterium — ignite briefly into silver wire wherever a shaft of refracted light sweeps through. An Acantharia rotates slowly nearby, its twenty strontium sulfate spines arranged in precise icosahedral geometry and shifting between teal, amber, and rose through birefringence as they turn, the myoneme fibers anchored invisibly at their bases ready to adjust the cell's buoyancy in response to pressure or light. Behind you, the Collodarian colony fills peripheral vision like a warm amber nebula, its gelatinous shared matrix backlit by surface sun filtering through thousands of embedded symbiotic dinoflagellate cells — each a five-to-fifteen-micron sphere burning with chlorophyll warmth — while Chaetoceros diatoms drift past like frosted glass shuttles trailing hair-thin silica setae, and a copepod nauplius the apparent size of a school bus tumbles through mid-water, its translucent body and beating limbs moving in the slow-motion choreography of a world ruled entirely by Stokes drag.