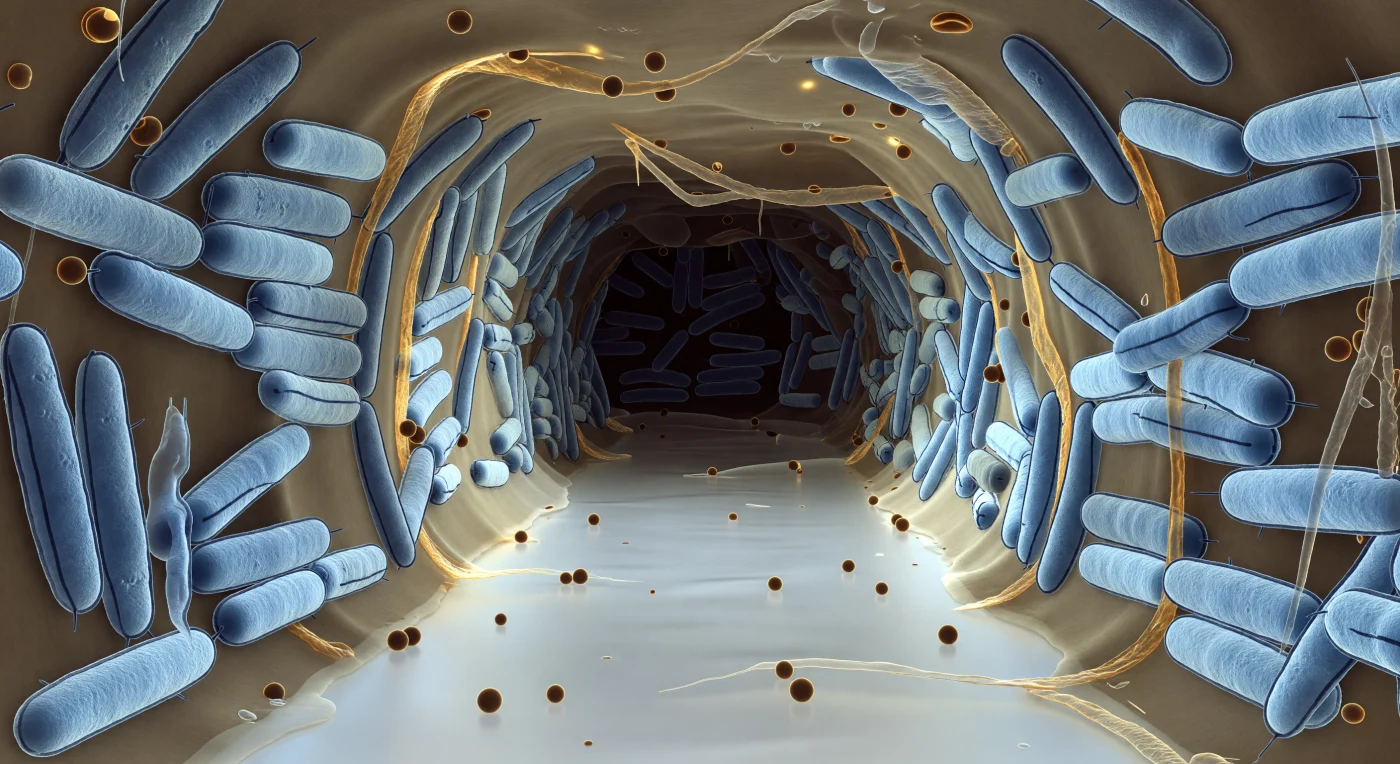
You are suspended at the mouth of a corridor carved through living architecture — a pale, luminous void cutting between walls of densely packed bacterial bodies, each one an ice-blue cylinder roughly the scale of a building to you, their double outer membranes tracing two razor-thin dark lines along every surface, a structural motif repeated hundreds of times into the deepening blue-grey haze where the channel bends toward shadow. The translucent amber-tan exopolysaccharide matrix fills every gap between organisms, binding them into a coherent collective structure — part scaffold, part communication medium — its surface faintly iridescent where hydration layers create thin-film interference, massing into ropy bundles between neighboring cells before thinning to a gossamer film at the channel edge. Scattered across the fluid column and resting against the matrix overhang are outer membrane vesicles, perfectly spherical and amber-glowing, each a sealed lipid bubble carrying signaling molecules and enzymes between organisms that cannot otherwise touch — a form of molecular postal service operating continuously within this chemically active corridor. The light fades with biological purpose: the pale silver-white of your position grades through charcoal toward the channel's vanishing point, tracing the real oxygen and nutrient gradients that govern which cells in this community survive, sporulate, or starve, the entire biofilm less a colony than a differentiated tissue assembled without a blueprint, sustained by nothing but chemistry and proximity.

You are suspended in the extracellular fluid just beyond the boundary of a human T-lymphocyte, and the world around you is defined not by light but by mass, chemistry, and the slow inevitability of molecular drift. The plasma membrane curves away in every direction like the cliff face of a deep-ocean trench — its surface an undulating slate-blue interrupted by the enormous crowns of CD4 receptors and the translucent, carbohydrate-tipped fronds of the glycocalyx, which blur the hard edge of the cell into a zone of increasing molecular density, like silt rising from a seafloor. Dozens of HIV-1 virions drift through the faintly translucent extracellular fluid in Brownian suspension around you, each one a softly glowing sphere roughly 120 nanometers across, its interior a smoky blue-gray housing the faint cone-shaped shadow of a condensed capsid core, its outer envelope studded with trimeric gp120-gp41 spike complexes rendered in burnished gold — the only warmth in an otherwise cold, directionless cryo-EM luminescence that emanates from nowhere and everywhere at once. The closest virions have begun drifting into the glycocalyx forest, their gold spikes extending toward CD4 anchoring sites with a proximity that reads as inevitability: the first molecular handshake of viral entry, playing out in a medium where Brownian noise, receptor affinity, and diffusion gradients are the only forces that matter. Beyond them, more virions dissolve progressively into the pale haze of dissolved glycoproteins, their gold trimers reduced to faint warm sparks fading into the pearlescent murk of a fluid world with no photons, no shadows, and no indifference — only chemistry.

You stand at the floor of a vast biological canyon whose walls are not stone but membrane — the inner mitochondrial membrane rearing up in great convoluted folds, each ridge climbing hundreds of body-lengths overhead before curling back into shadow, its surface a deep charcoal-brown from osmium staining and studded across every visible face with ATP synthase complexes, their rounded F1 heads nodding outward like mushroom caps anchored on molecular stalks, each one a nanoscale turbine spinning proton gradients into the chemical currency of life. The matrix in which you stand is nothing like air or water: it is a dense, amber-tinted colloid packed with enzymes, mitochondrial ribosomes, and loose supercoiled arcs of mitochondrial DNA, the protein concentration so extreme — over 500 milligrams per milliliter — that light scatters into a soft volumetric honey-glow, and globular TCA cycle enzymes press close like half-dissolved boulders whose beta-sheet surfaces you could almost reach out and trace. Far ahead, the cristae junction narrows to a tight slot-canyon bottleneck, its walls so densely lined with ATP synthase heads that they form an almost continuous cobblestone ceiling above the golden-lit throat, the membranes nearly touching in a geometry that concentrates the proton-motive force and restricts the diffusion of cytochrome c, turning topology itself into biochemical machinery. Beyond the junction, the membrane opens again into another chamber, its far wall lost in amber atmospheric depth — a world of pure biological architecture, breathing with slow thermal undulation, crowded with molecular machinery in every direction, alive at a scale where structure and function are indistinguishable.
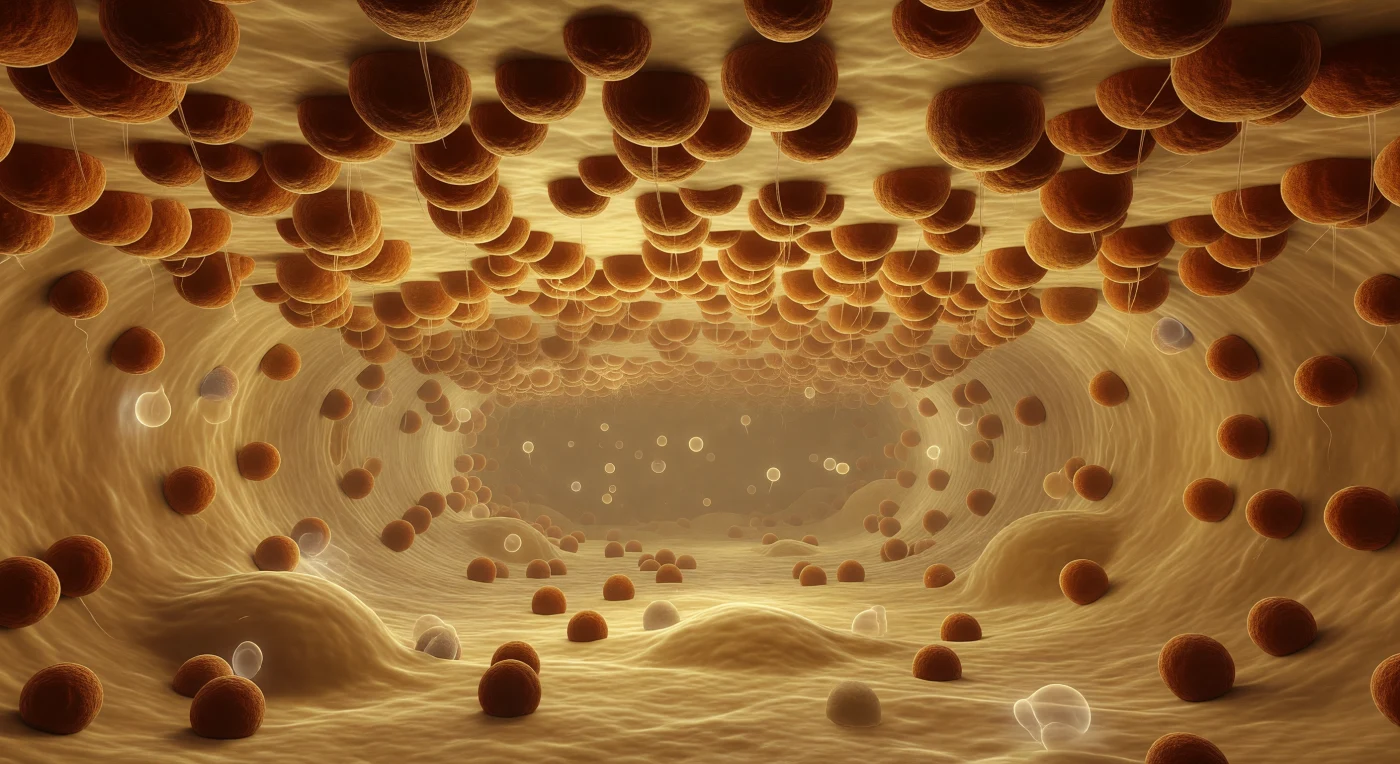
You drift suspended within the lumen of the rough endoplasmic reticulum, adrift in a corridor of warm amber light that seems to emanate from the aqueous medium itself — a space packed with dissolved proteins drifting past like pale, slow ghosts caught in a sourceless glow. Above you, pressing close with the authority of architecture, the cytoplasmic face of the ER membrane has been entirely consumed by ribosomes: thousands upon thousands of rust-brown, asymmetric spheres some 25 nanometers across, packed shoulder to shoulder in an unbroken canopy that obliterates any view of the bilayer beneath, each one a molecular machine threading a nascent polypeptide through the translocon and releasing it, strand by gossamer strand, into the lumen below. These are the factories of the secretory pathway — co-translational translocation in continuous, simultaneous, unselfconscious operation across every surface you can see, the newly synthesized proteins entering this aqueous corridor to be folded, modified, and sorted toward their destinations in the Golgi and beyond. The corridor itself recedes in deep perspective, bounded above and below by twin ribosome-studded membranes converging toward a vanishing point lost in protein-fogged amber murk, its geometry distorted by occasional vesicular bulges bowing gently inward like blisters in wet paper. The density is total and industrial: macromolecular crowding at several hundred milligrams per milliliter surrounds you, and the proliferation of ribosomal bodies overhead — each enormous relative to your position, together constituting not ornament but inevitability — makes unmistakably felt that the cell manufactures its proteins without pause, without awareness, in every direction at once.

You stand at the outermost frontier of a living cell, suspended at the advancing tip of a lamellipodium where individual actin filaments — each just seven nanometers thick — branch from Arp2/3 complexes at precise seventy-degree angles, building a fractal canopy of electric teal and cold jade that radiates inward like an endlessly repeating coral reef frozen in the act of growing. Beneath you, the substrate is illuminated only by an evanescent wave penetrating a few hundred nanometers upward, a cold TIRF glow that catches the warm amber halos of focal adhesion plaques — molecular anchor complexes the size of boulders from this vantage, their integrin-rich surfaces bridging the extracellular matrix to the cytoskeletal machinery driving the whole cell forward. The actin network is not random but exquisitely regulated: Arp2/3 branching, capping proteins, and cofilin-mediated severing balance polymerization at the leading edge against disassembly further inward, generating the protrusive force that pushes membrane against resistance through nothing more than the chemistry of monomer addition. To either side, filopodial bundles — parallel actin arrays distinct from the branched lamellipodial mesh — project outward into the extracellular void like luminous antennae, their tips sensing substrate chemistry and topography in real time. Everything here trembles with thermal energy, macromolecular crowding thickens the medium to a dense gel, and the branching architecture continues inward layer behind layer until density alone swallows the light.

You hover in the viscous nucleoplasm, drifting through a medium dense as cold honey, the constant tremor of Brownian motion pressing against you from every direction as you approach the massive structure that fills your entire field of view. Ahead, the nuclear pore complex rises like a cathedral gateway — a perfect eight-fold rosette of burnished golden-amber protein sub-complexes, each spoke curving inward with organic precision, the whole assembly embedded flush in the dark charcoal double membrane of the nuclear envelope that stretches laterally into the distance like two parallel vaults of ancient stone. From the axial center of the ring hangs a diaphanous curtain of FG-nucleoporin filaments, pale ivory strands beaded with irregular nodular densities that drift and flick with hypnotic slowness, occasionally parting to reveal a deep cylindrical throat of near-total darkness — the selective channel through which hundreds of macromolecular cargoes transit every second, each passage gated not by mechanical doors but by the transient, hydrophobic partitioning of intrinsically disordered proteins. On either side of the portal, massive irregular cliffs of compacted heterochromatin rise in deep indigo and midnight blue, their nucleosome-beaded surfaces absorbing rather than reflecting the ambient phosphorescence, while behind you the nucleoplasm opens into a softer lavender-grey haze of euchromatin — looser chromatin domains and drifting condensate droplets marking the dynamic, phase-separated interior of a nucleus that is anything but inert.

Before you rises a tower of impossible precision — fifteen thylakoid discs stacked in strict register, each one a dense, corrugated slab of emerald-green membrane whose surface roughness resolves, at close range, into a near-continuous cobblestone mosaic of photosystem II and light-harvesting complex heads pressed outward through the lipid bilayer, their chlorophyll-laden protein arrays absorbing incoming quanta and bleeding back a faint warm luminescence. Between each disc, thin lemon-yellow lumen gaps separate the membranes like luminous mortar joints, their acidic interiors hazed with diffusing protons migrating down an electrochemical gradient that is the molecular heartbeat of photosynthesis. Stromal thylakoid lamellae extend horizontally from the base of the stack like emerald ribbons curving away into the gel-like stroma, physically connecting this granum to neighboring towers and ensuring that electron transport chains embedded in geographically separate membranes remain electrically and chemically continuous. The stroma surrounding you has the quality of a luminous, opalescent fog — densely packed with irregular pale masses of RuBisCO complexes on every sightline, and punctuated by small amber plastoglobuli whose lipid-rich surfaces catch the omnidirectional chlorophyll-filtered glow and warm it into honey-gold. The entire scene is lit not from without but from within, each membrane layer both absorbing and re-emitting light so that the atmosphere itself seems to breathe.
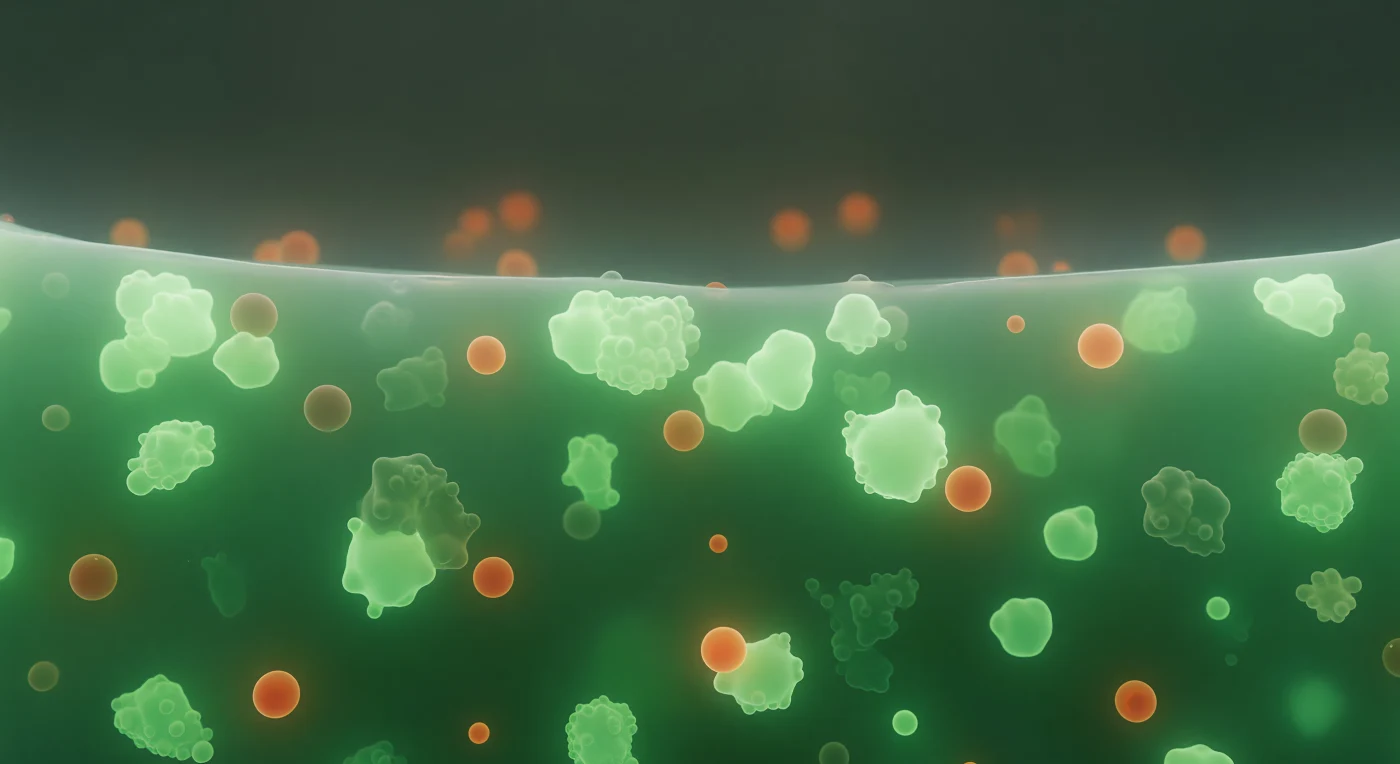
You are suspended inside a stress granule — a membraneless organelle that assembles by liquid-liquid phase separation when the cell halts translation under stress, concentrating RNA-binding proteins and stalled messenger RNAs into a viscoelastic condensed phase distinct from the surrounding cytoplasm. The world around you is an emerald luminescence with no single source: G3BP1 protein, a nucleating scaffold of stress granule assembly, saturates the medium at concentrations far exceeding those of the dilute cytoplasm just beyond, and its GFP fluorescence fills the interior with a cool, diffuse glow that thickens with distance into an impenetrable jade opacity. Closer and farther, mRNA-protein clusters materialize as brighter nodes — not hard objects but local intensifications where RNA and low-complexity domain proteins have co-condensed further, pooling into denser microdomains within the already-dense matrix — while warm amber-orange TIA1-rich co-condensates drift in slow suspension, their surfaces shimmering where two distinct liquid phases meet at an ultralow interfacial tension. Far ahead, the condensate boundary declares itself as an abrupt precipice, a sharply curved interface where the entire luminous green world drops into near-nothingness in a distance of only tens of nanometers — the physical signature of phase separation, a thermodynamic horizon separating two coexisting states of the same cellular matter.

You are suspended directly above the flagellar motor's central axis, looking straight down through the extracellular medium into one of the most intricate rotary machines in the living world — nested rings of bronze and oxidized gold descending through layered membranes like the plan view of an ancient temple, the outermost L-ring embedded in the deep cobalt sheet of the outer membrane, the P-ring anchored in its faintly amber peptidoglycan stratum below, and at the very center the sculpted MS-ring filling the inner membrane plane with radial ridges and subunit boundaries caught in diffuse, sourceless light. Seventeen stator complexes — each a dense asymmetric mass of MotA and MotB proteins — crowd the rotor's circumference in near-perfect radial symmetry, pressing against the inner membrane like massive turbine buttresses, their pewter-gray surfaces carrying dull highlights where molecular interfaces make contact with the rotor at angles refined by billions of years of selection. This is a proton-powered rotary engine, driven by transmembrane electrochemical gradients that funnel hydrogen ions through each stator unit to generate torque, spinning the rotor and its attached filament at speeds of 100 to 1,000 revolutions per second in living cells. Above you, the flagellum corkscrews away into the turbid, faintly luminous extracellular haze — a helical rope of flagellin subunits whose surface catches cold, directionless light — while the hook junction just above the L-ring seals the rotating shaft to the outer membrane with the functional precision of a mechanical gland. The entire assembly, spanning something close to a large stadium floor in your scaled perception, conveys the deep aesthetic of a structure perfectly evolved to spin, endlessly and invisibly, in the dark interior of a living organism.

You are standing at the base of a living forest that has no trees — only hundreds of smooth, deep blue-cyan cylinders rising from a warm ochre plain in every direction, each shaft roughly 200 nanometers across and six micrometers tall, packed so densely that no horizon exists, only an infinite repetition of vertical forms dissolving into aqueous blue-gray haze above. These are airway epithelial cilia, each one internally braced by a nine-plus-two arrangement of microtubule doublets whose geometry presses faintly through the outer membrane like ribs beneath wet silk, driven by dynein motor proteins consuming ATP in coordinated strokes to sweep the overlying mucus layer toward the throat at roughly ten millimeters per minute. Between the nearest shafts, amber-gold mucus threads hang in slow catenary arcs — luminous, slightly translucent strands of glycoprotein gel whose viscoelastic properties allow them to stretch without breaking, catching diffuse biological light the way honey catches candlelight, anchored where they touch a ciliary surface by the same surface tension that shapes a dewdrop. Far overhead, the metachronal wave is caught in a single frozen instant: an entire colonnade of cilia leaning ten to fifteen degrees in unison while the adjacent rank stands upright again, the phase offset between each row producing a scalloped, rippling ceiling — a breathing rhythm carved into space, generated by a structure that has no awareness it is breathing at all.
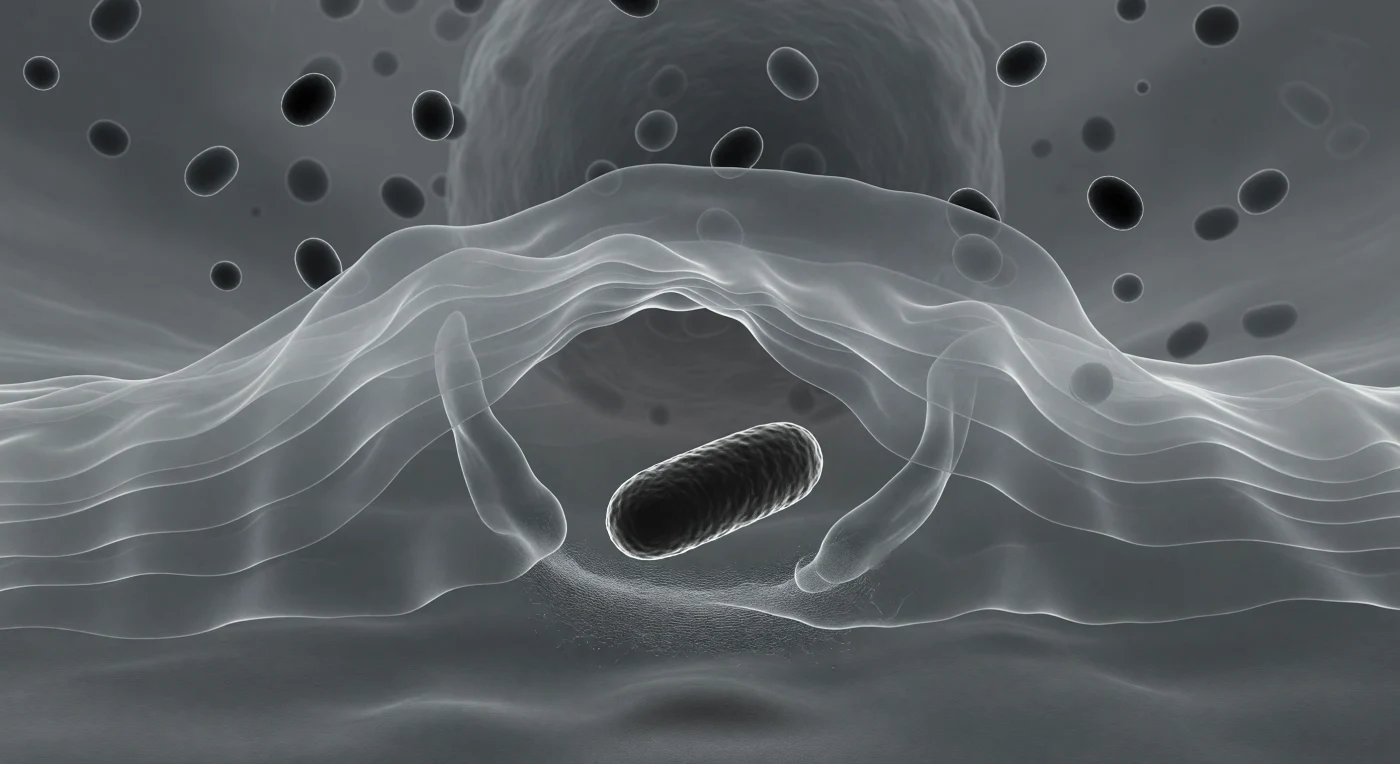
You are pressed against the leading edge of a translucent cytoplasmic veil, watching it arc forward and close around a dense, rod-shaped bacterium like slow cupped hands completing a gesture that has been rehearsed a billion times across evolutionary time. The lamellipodium surrounding you is actin-rich and gel-like — neither liquid nor solid but a living meshwork of polymerized filaments that push the membrane forward through coordinated treadmilling, driven by Arp2/3-nucleated branching just micrometers beneath the advancing tip. The bacterium ahead, roughly two micrometers long, is already nearly engulfed: receptor-ligand binding between surface opsonins and the macrophage's Fc and complement receptors triggered this entire choreography, propagating a zipper-like signal around the prey's circumference that recruits more actin, extends more pseudopod, seals the phagosomal cup. Behind you, the lysosomal granules drifting in the thickening cytoplasm are the bacterium's near future — enzyme-loaded compartments that will fuse with the nascent phagosome and reduce the entire dark monolith ahead to molecular salvage. The geometry of the enclosure is almost complete, and in the cold, directionless DIC light that encodes depth in shadow and mass in tone, the whole scene carries the weight of something architectural and inevitable, a structure solving itself.

You hover at the nadir of an enormous amber basin, the central concavity of a human red blood cell sweeping away in every direction like the floor of a warm, ochre-tinted impact crater — its surface a continuous, gently undulating plain of translucent gold, somewhere between stretched caramel and sun-warmed honey. Just beneath that shimmering membrane, pressed into the underside like etchings in amber glass, the hexagonal lattice of the spectrin cytoskeleton renders itself visible in slightly cooler relief — a geodesic web of biological scaffolding that gives the whole plain the quality of hammered gold foil, the network maintaining the cell's remarkable capacity to deform through narrow capillaries and spring back without rupturing. Raking oblique illumination drives in from the upper left with the hard-edged precision of a scanning electron beam, catching every crest of the membrane's undulation and throwing the concave hollows into pools of deep amber shadow, lending the biological surface the hyper-material tactility of cast bronze. At mid-distance, neighboring red blood cells have stacked themselves into rouleaux — smooth-sided columns of biconcave discs pressed face to face, a reversible aggregation driven by plasma proteins bridging adjacent membranes — their corrugated silhouettes rising like sandstone towers from a straw-colored plasma still carrying its suspended cargo of spiny, projection-roughened platelets, each one a surveillance body circulating in readiness, dwarfed by the great cellular walls around it yet enormous against the spectrin lattice at your feet.
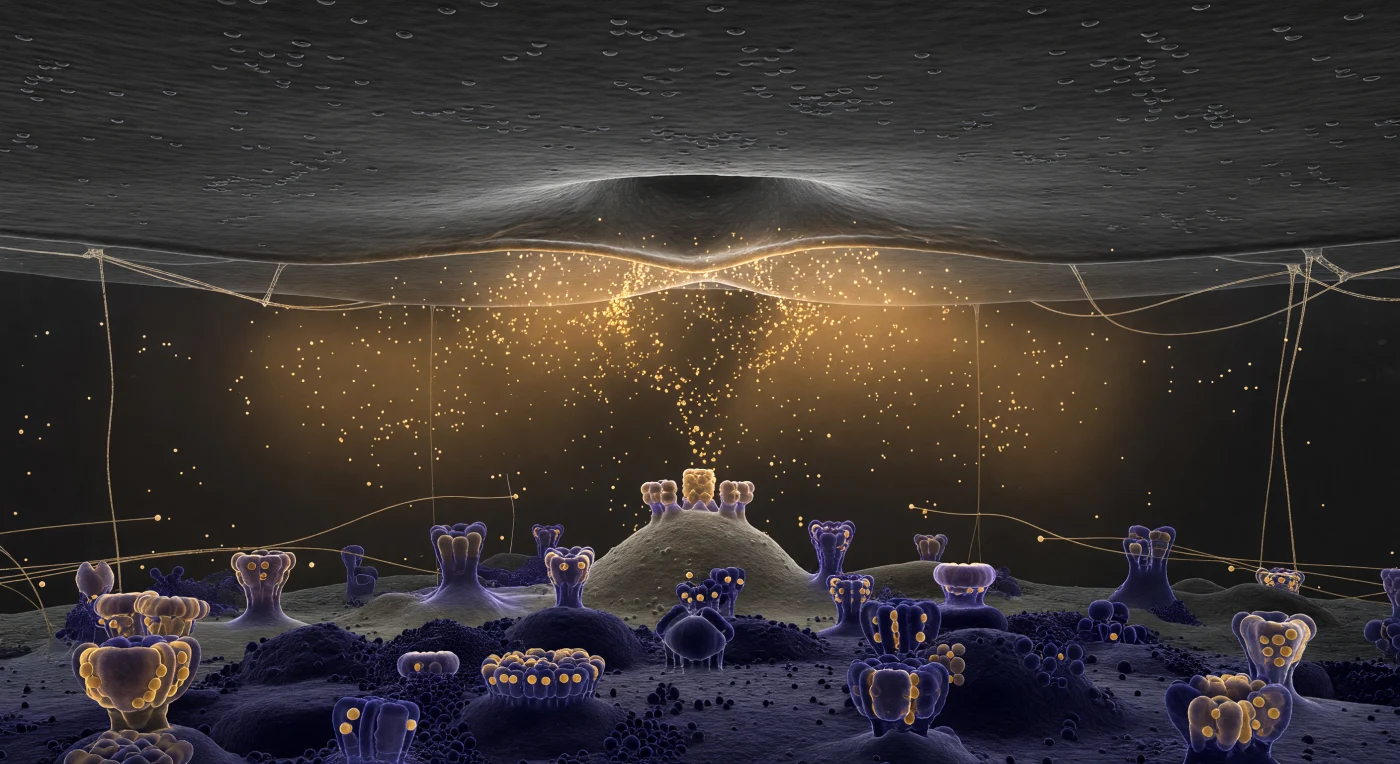
You are suspended inside a corridor that has no horizon — only ceiling and floor, both pressing in from above and below simultaneously, separated by a gap of just 25 nanometers, a distance so violently compressed that the entire synaptic cleft feels less like a space and more like a contact. Above you, the presynaptic membrane stretches as a vast charcoal plain, and at its center a single synaptic vesicle has committed irreversibly to fusion, its lipid bilayer collapsing into the membrane in a smooth omega-shaped dimple as the two leaflet pairs merge into one continuous sheet — a structural event lasting mere milliseconds that nevertheless reshapes the biochemical landscape of the entire corridor. From the fusion pore, thousands of neurotransmitter molecules — glutamate, or acetylcholine, depending on which synapse you inhabit — exhale outward as a warm amber cloud, not discrete objects but a diffuse volumetric tide governed entirely by Brownian diffusion, thinning at the edges as molecules spread laterally across the cleft, their motion unguided and probabilistic. Below, the postsynaptic density rises to meet them: a dark-purple, electron-dense scaffold of extraordinary architectural complexity, bristling with AMPA and NMDA receptor complexes whose extracellular domains protrude upward into the cleft like stone formations, their binding sites oriented precisely to intercept the incoming transmitter flood — a molecular geometry refined over hundreds of millions of years of synaptic evolution, here rendered suddenly, overwhelmingly immediate.
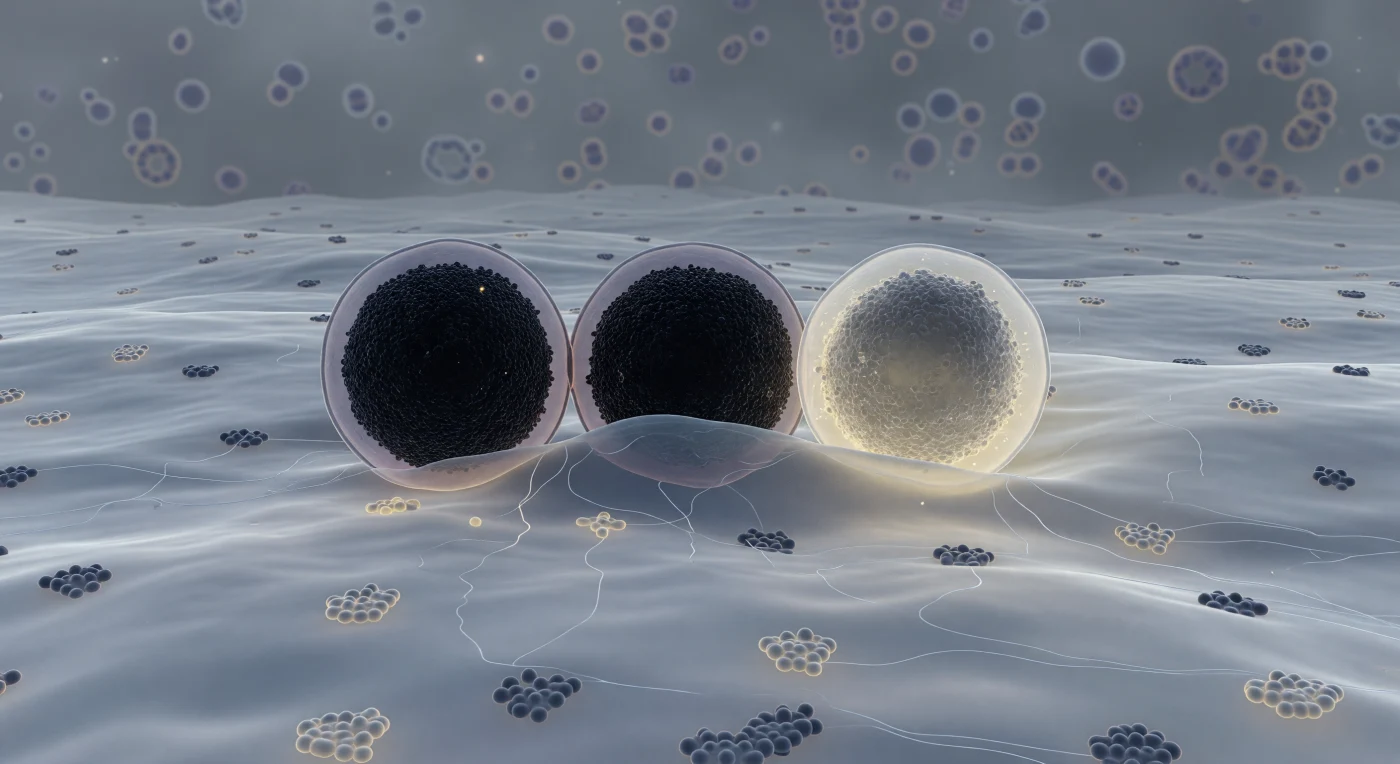
You exist pressed against the inner face of a plasma membrane that stretches to every horizon like an immense, softly luminous plain — a fluid bilayer of blue-grey lipid faintly threaded with embedded protein complexes, its surface alive with slow rolling topography where cholesterol-rich raft domains pool in subtly lighter, more ordered patches. Three granules dominate the scene, each the scale of a small building: the first sits docked in full contact, its near-black crystalline core of zinc-insulin hexamers so densely packed they absorb almost all light, ringed by a thin lavender halo where its limiting membrane holds; the second bows the plasma membrane gently inward in a hemi-fused collar where lipid identity dissolves into indeterminate grey; the third has already surrendered, its once-opaque core unraveling from deep charcoal to a dispersing warm ivory-yellow haze as insulin molecules escape into the pale amber luminosity of the extracellular space beyond. This is the final choreography of regulated exocytosis, driven by SNARE protein complexes — syntaxin, SNAP-25, and synaptobrevin — that zipper the two bilayers together with enough mechanical force to overcome the energy barrier of membrane fusion, a process completed in milliseconds yet representing the culmination of hours of granule biogenesis and priming. Threading through the cytoplasmic space between the granules, ghostly actin filaments form a loose cortical mesh, while the deeper cytoplasm recedes into a structured murk of macromolecular crowding — a single frozen instant in a thermally inevitable act, caught mid-breath, that cannot be undone.

You are suspended at the very tips of the brush border, looking obliquely across a landscape that extends in every direction like an engineered plain — thousands upon thousands of cylindrical microvillus shafts, each roughly 100 nanometers across, packed into near-hexagonal regularity and rising to meet you from the enterocyte surface far below. These projections are not passive scaffolding: each shaft is stiffened internally by a tightly cross-linked bundle of actin filaments anchored through villin, fimbrin, and myosin-1a into the dense terminal web of the cell cortex beneath, the entire array constituting the intestinal brush border — a structure that amplifies the absorptive surface area of the small intestine by a factor of roughly twenty. From every shaft tip rises the glycocalyx, a soft three-dimensional pelt of O- and N-linked oligosaccharide chains extending from transmembrane glycoproteins including alkaline phosphatase and the peptide hydrolases embedded in each villus membrane, their combined mass forming a continuous extracellular matrix several tens of nanometers deep that functions simultaneously as molecular sieve, enzymatic surface, and protective barrier against luminal proteases. Oblique warm light rakes across the field from the upper right, casting precise crescent shadows in the narrow intervillus channels — those aqueous gutters between shafts, rich in ions, partially hydrolyzed peptides, and monosaccharides, the very molecules that will cross the membrane below through SGLT1 and GLUT5 transporters in the next fraction of a second. The slight tilt of one shaft, the asymmetric thickening of a glycocalyx cloud on its windward face, the faint oscillatory tension encoded in this frozen moment — all of it declares living tissue caught mid-breath, restless and continuous beneath the stillness of the image.

You are suspended at the exact center of a dividing cell during anaphase, and surrounding you in every direction is a cathedral of electric-green light — the mitotic spindle rendered in fluorescent tubulin, its microtubule cables radiating from two blazing centrosomal poles far above and below like the rigging of some vast biological machine under full tension. The kinetochore fibers are the most dramatic structures: thick, taut, intensely luminous bundles of polymerized tubulin, each one locked onto a chromosome mass and pulling it poleward with measurable force, the blue-violet chromatin receding into the polar glow like storm clouds retreating into twin suns. Directly at your level, the midbody cuts across the visual field as a bar of concentrated white-green fire — the antiparallel overlap zone where interpolar microtubules from opposite poles interdigitate and are cross-linked by motor proteins including the kinesin Eg5, so densely packed with tubulin that it outshines the rest of the spindle — while all around you, interpolar fibers cross in overlapping diagonals, forming a three-dimensional lattice of living polymer through which you float weightless. Beyond the spindle's glow, the cytoplasm is absolute black, yet not empty: macromolecular crowding at 300–400 mg/mL scatters the microtubule fluorescence into soft aureoles of milky green mist at every fiber's edge, giving biological mass to the darkness and reminding you that this entire luminous architecture — chromosome segregation, spindle tension, the midbody's fire — is a protein machine operating in real time, measured in minutes, inside a space you could cross in a single human stride.

You are suspended in the pale silver immensity of blood plasma, drifting toward an endothelial wall whose true surface remains almost entirely hidden — buried beneath the glycocalyx, a dense, cathedral-like forest of proteoglycan and glycoprotein chains standing half a micron to two microns tall, their heparan sulfate strands arcing and forking like winter birch branches rendered in cold blue-silver under the diffuse glow of ruthenium-red false color. This living coat is no passive barrier: negatively charged sulfate and carboxyl groups embedded along every filament generate steep electrostatic gradients that regulate which molecules may pass, trap signaling proteins, and mechanically buffer the shear forces of flowing blood, while bound water sheathes each strand in a hydration layer that gives the whole structure its viscoelastic, fog-made-solid quality. Your own shadow — the broad biconcave dome of the approaching red blood cell — sweeps across the canopy below you, turning ice-blue filaments to indigo and near-black at the center of the eclipse while the illuminated margins continue to glow with that precise cold aqueous light, depth accumulating as strand density thickens from sparse at the canopy top to an effectively impenetrable lacework just above the charcoal membrane. The glycocalyx is not merely a structural feature but the cell's sensory interface with the circulatory world: it transmits fluid shear stress to the endothelial cytoskeleton, modulates vascular permeability, and mediates leukocyte rolling — and as contact becomes inevitable, its outermost filaments are already beginning to deform around your leading edge, the forest yielding with a slowness that makes the scale of what you are entering impossible to doubt.
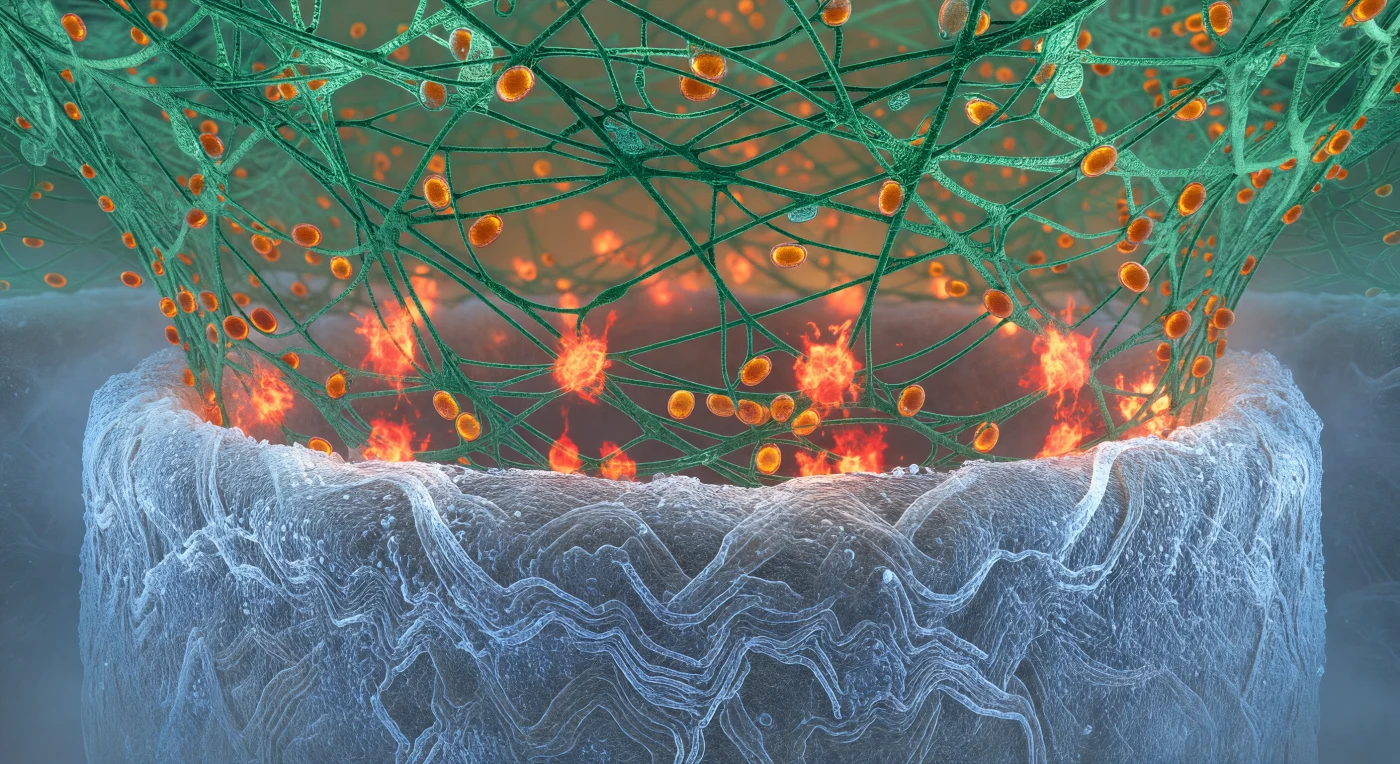
You stand pressed into the killing zone between two of the most formidable biological architectures the living world produces — beneath you, the fungal hypha curves away like the hull of a vast pale vessel, its chitin wall radiating cold blue-white luminescence where calcofluor dye has rendered every interlocked polysaccharide strand incandescent, a surface dense and granular as ancient salt that disappears into haze at both flanks under its own geological weight. From above, the neutrophil's actin cradle descends in a viridian forest of branched F-actin cables, each filament crosslinked under tension and gleaming with a resinous wet light, the entire scaffold gripping the hypha with the organized ferocity of a living vise. At the interface you inhabit, reactive oxygen species erupt in irregular red-orange detonations — concentrated bursts of oxidative chemistry igniting like slow flashbulbs, their amber cores bleeding outward into copper coronas that momentarily backlight the actin mesh from below, the product of NADPH oxidase complexes assembling at the phagosomal membrane to flood this narrow space with superoxide and its downstream derivatives. Threading through the scaffolding above, azurophilic granules drift in warm amber procession along actin cables, each one a sealed reservoir of myeloperoxidase and elastase converging on the brightest ROS fronts — the entire scene a coordinated molecular siege in which chemistry, mechanics, and targeted biochemical delivery converge at a contact zone measured in micrometers but experienced here as an vast, smoldering battlefield.
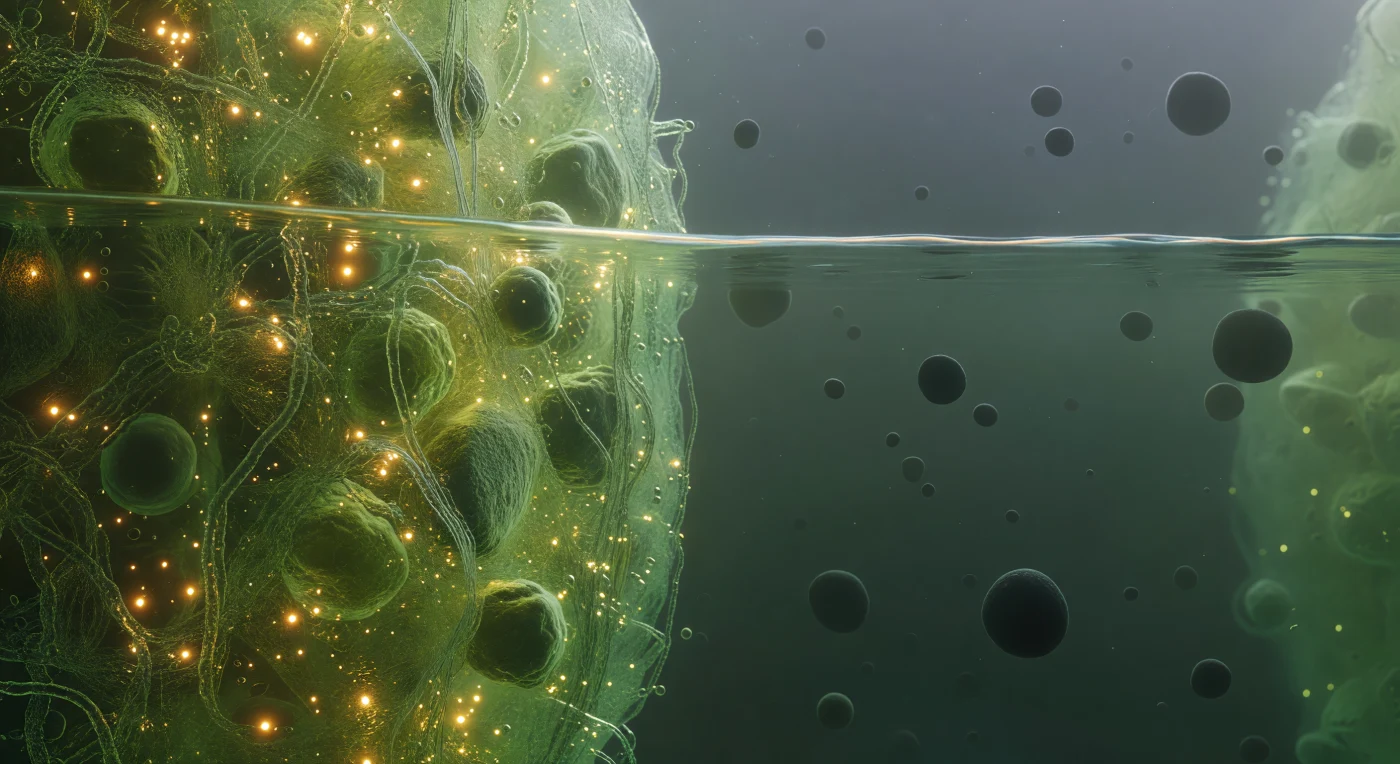
You float precisely at the threshold of two immiscible biological worlds, straddling the phase boundary of a stress granule — a membraneless organelle assembled not by lipid walls but by the thermodynamic logic of liquid-liquid phase separation, where intrinsically disordered proteins and mRNA chains spontaneously partition into a dense, self-organized condensate phase coexisting with the surrounding dilute cytoplasm. To your left, the condensate interior glows with a saturated green-gold luminescence, its molecular mesh so densely packed — proteins and RNA entangled at concentrations approaching hundreds of milligrams per milliliter — that light scatters and pools into an amber warmth, the whole medium trembling with Brownian agitation as ten thousand macromolecules press and slide against one another in a viscoelastic medium that behaves somewhere between liquid and gel. Directly before you, the interface itself holds as a sharp thermodynamic discontinuity, not a gradient but a true phase boundary maintained by surface tension, shivering with capillary fluctuations at the nanometer scale — a faintly iridescent, undulant seam where the dense condensate ends absolutely and the dilute phase begins, catching the interior light in a thin luminous caustic that is the only hard edge in this otherwise fluid and probabilistic landscape. To your right, the dilute cytoplasm opens into a comparative void, a cool blue-green haze where individual ribosome complexes drift as dark matte boulders separated by distances that feel like open ocean after the claustrophobic interior — each one a 25-nanometer asymmetric assembly of RNA and protein, enormous against the diffuse background scatter, fading into a particulate murk where the next organelle boundary is only a vague brightening at the limit of visibility.

You hover in the narrow aqueous corridor between two immense epithelial walls, pressed nearly flush against one another, their paired membranes running like dark parallel cliffs into the depths below you — a space so confined that the extracellular fluid filling it registers less as a gap than as a faint golden humidity, thickened with glycocalyx sugars and dissolved ions. Girding the full perimeter of this interface at your eye level, a continuous crimson belt of tight junction proteins — claudins, occludins, ZO-1 — forms an unbroken molecular seal, a glowing cinnabar weld fused directly into the bilayers of both cells simultaneously, its slight granularity betraying the crystalline clustering of individual protein complexes repeating along the seal like stitches in living metal. Just below it, a second band resolves in cooler emerald light: the E-cadherin adherens junction, its ectodomains reaching across the 20-nanometer intercellular gap and interlocking like interlaced fingers, mechanically coupling the cytoskeletons of both cells into a single tensile system that coordinates epithelial integrity across the entire tubule circumference. Above you, the tight junction belt marks an absolute boundary — beyond it, the tubule lumen opens without transition into a cavernous black void, its aqueous contents held back entirely by the molecular seal at which you hover — and below you, through semi-opaque cytoplasm radiating diffuse indigo from enormous DAPI-blue nuclei, the cell bodies recede like the deep panels of a stained-glass window seen from within the lead came, the junction proteins themselves the luminous armature holding the living architecture together.



















