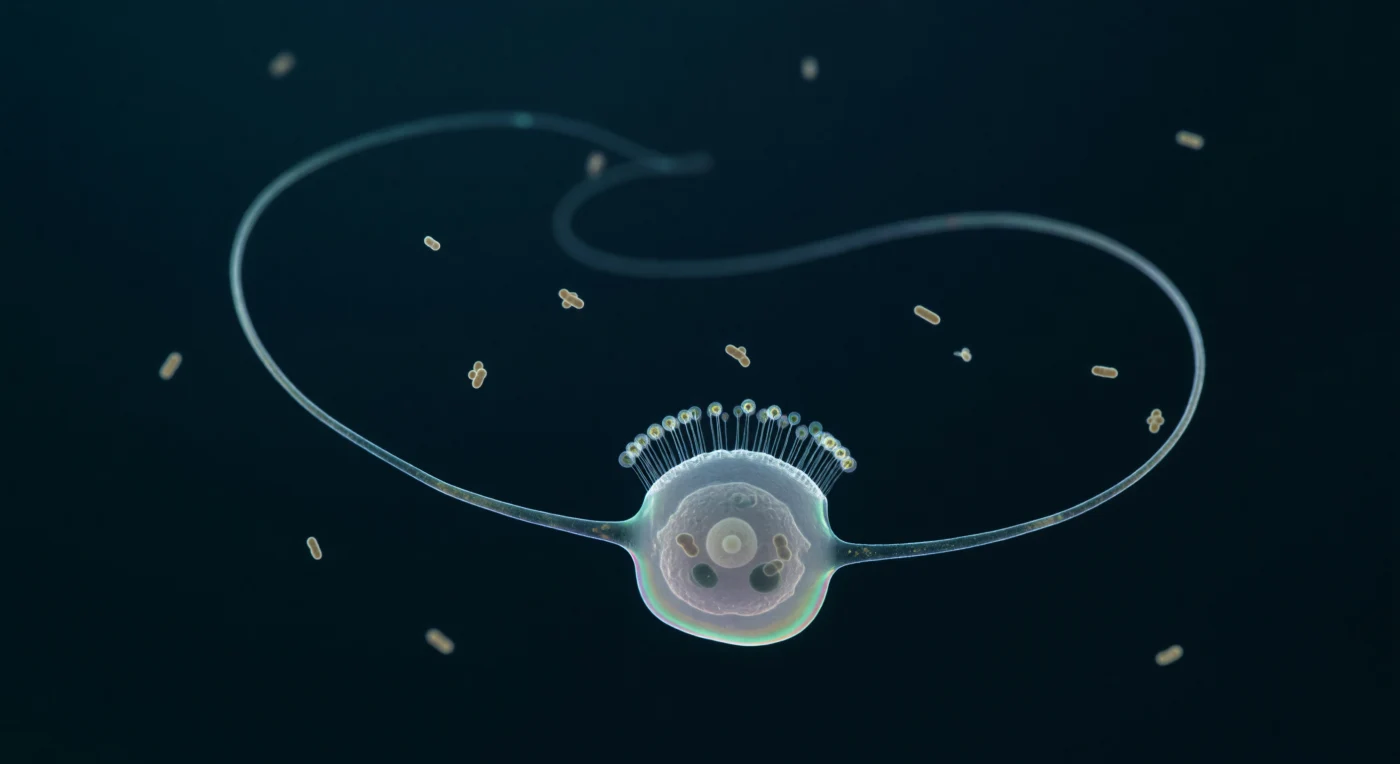
You are suspended inside a medium that behaves less like water than like a cold, faintly resistant gel — at this scale, viscosity overwhelms inertia so completely that the sinusoidal flagellum arcing across your field of view is not slicing through the void so much as sculpting it, driving a slow toroidal current that draws dissolved organics and drifting bacteria gently inward through the near-invisible cylinder of microvilli surrounding its base. This single choanoflagellate cell — its nearly colorless body no wider than a large bacterium is long — represents one of the closest living relatives to the animal ancestor, a unicellular organism whose collar-and-flagellum architecture is mirrored almost exactly in the choanocytes lining the filtration chambers of every sponge alive today, a molecular continuity preserved across perhaps six hundred million years of evolution. The collar itself, a ring of thirty-odd actin-filled microvilli thinner than a wavelength of visible light, functions as a sieve mesh, intercepting bacteria carried on the flagellum's helical wake and passing them into food vacuoles where digestion is already underway — the two dark inclusions visible inside the cytoplasm each still containing the compressed ghost of a rod bacterium mid-dissolution. Around you in the middle distance, a dozen more bacteria drift and tremble in pure Brownian agitation, neither rising nor falling, suspended in a thermal noise field that at this scale is the dominant physical reality, the closest thing this world has to weather.

Suspended in open water at the scale of a large bacterium, you face a structure that fills your entire visual world like a slowly turning chandelier moon — a 32-cell rosette colony of *Salpingoeca rosetta*, roughly forty micrometers across, floating free in the faint blue-black depth of estuarine water. Each cell body is a plump, slightly translucent ovoid rendered in the cool achromatic silver-gray of differential interference contrast, its nucleus visible as a denser ellipsoid within, and from every distal pole a flagellum extends outward — fifteen to twenty micrometers of trembling silver filament, too fine to resolve as a solid rod but unmistakably present, thirty-two of them radiating simultaneously to give the colony the electrified silhouette of a sea urchin frozen mid-breath. The basal poles of all cells converge inward along barely visible cytoplasmic bridge threads — the intercellular connections that make this not a loose aggregate but a coordinated multicellular entity — terminating at a warm amber core of extracellular matrix that glows with faint internal translucence, the only warm tone in an otherwise cool, near-monochrome palette. At the two-o'clock position one cell is mid-division, its body pinched into a peanut form with offset nuclear shadows marking the coming separation, a live record of the clonal cell divisions that built this geometry. Around you, marine-snow flocs drift softly out of focus, their tangled polysaccharide strands and embedded bacteria establishing the particulate, populated world through which this colonial protist — the closest living relative of all animals — quietly filters its share of the sea.
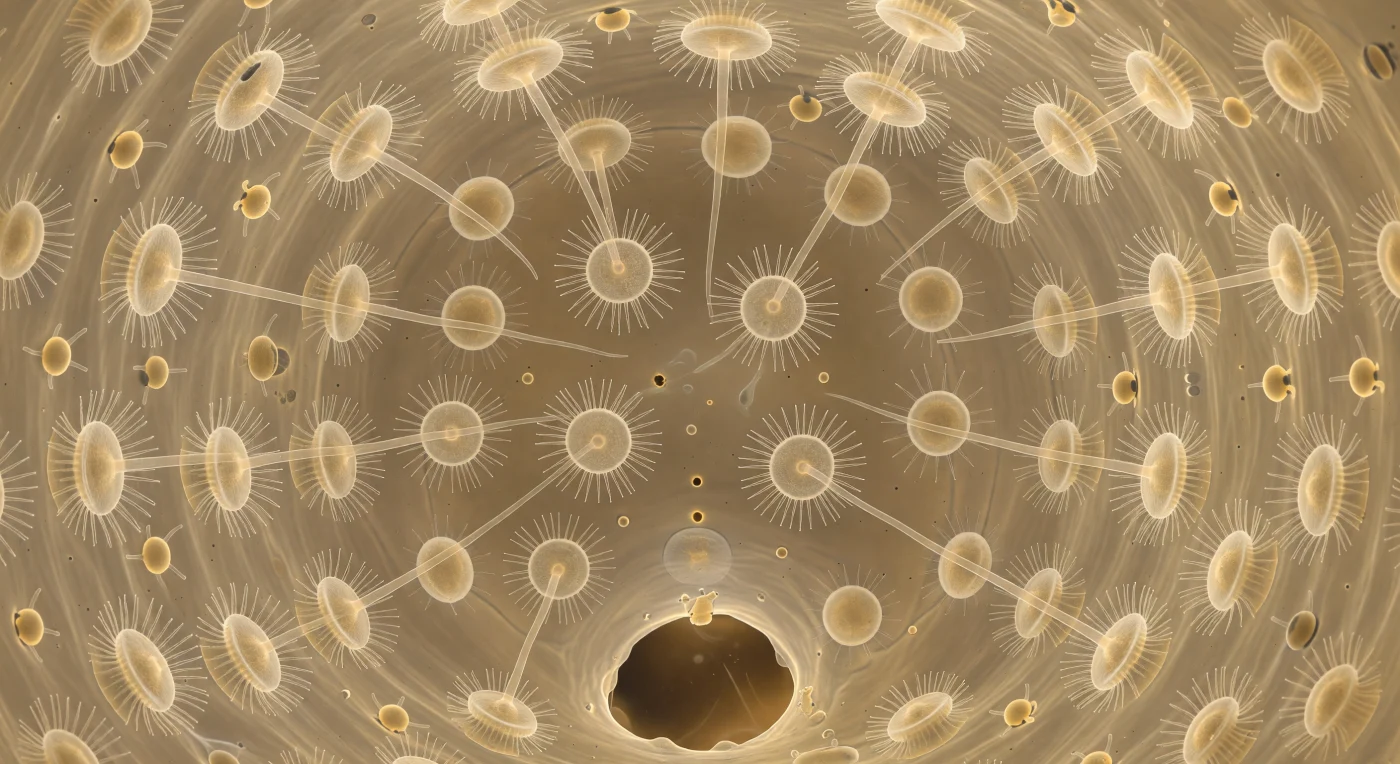
You are suspended at the heart of a living sphere, looking outward through a space barely forty microns across, its entire curved surface tiled without a single gap by choanocytes — translucent, dome-shaped cells each crowned with a crystalline collar of upright microvilli and a single inward-reaching flagellum that catches the diffuse amber light and bends it into faint prismatic tremors. All twenty-five flagella beat in coordinated offset phase, transforming the chamber interior into a continuously rippling luminescence that is neither mechanical nor chaotic but deeply, unmistakably biological — a collective hydrodynamic act in which each cell contributes to a laminar current that converges on the wide apopyle pore gaping directly ahead like an open archway into the canal system beyond. Two prosopyle inlet pores punctuate the wall at oblique angles, each only a few microns across, admitting thin threads of particle-laden seawater carrying ghost-shapes of bacteria destined to be captured against the collar mesh and drawn by endocytosis into the choanocyte body — a filtration strategy so effective that sponges cycle a volume of water twenty thousand times their own body mass each day. Beyond the cell layer, visible through the translucent tissue as an amber-gold fibrous lattice, amoeboid archaeocytes extend pseudopodial arms and drift through the mesohyl scaffold, redistributing digested nutrients and remodeling the extracellular matrix in slow, molten-glass motion. The entire chamber is a working demonstration of the morphological blueprint that unites sponges and their choanoflagellate relatives — the collar-and-flagellum unit whose ancient evolutionary origin sits at the deepest root of animal life itself.
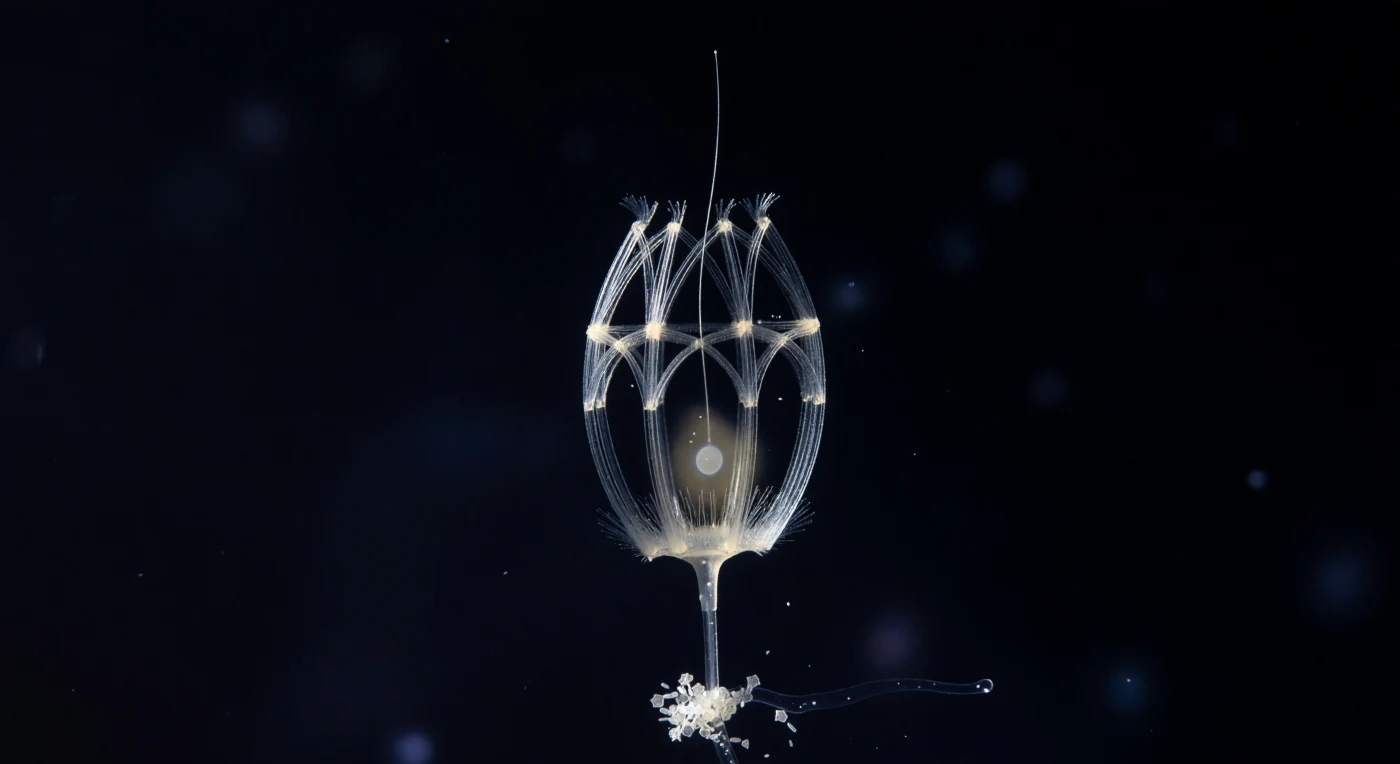
In the lightless reaches of the mesopelagic zone, a single *Diaphanoeca grandis* cell presents itself inside its self-assembled silica lorica — a gothic lantern of costal strips roughly 20 micrometers across, constructed from two interlocking rod systems: longitudinal ribs arching from base to apex like vaulting bones, cinched by transverse hoops, each silica filament blazing white under raking sidelight while the spaces between them breathe open as pure black negative space. The lorica is not decorative but functional, a rigid external scaffold secreted by the cell itself from dissolved silicic acid drawn from the surrounding seawater, its geometry channeling the flagellar current inward through the open anterior pole so that bacteria and dissolved organics are swept through the microvilli collar in a continuous filtration the cell maintains without rest. Within the cage, the living body is barely legible — a pale cytoplasmic haze in the lower interior, a faint nuclear ellipse, a corona of collar microvilli dissolving before they fully resolve, and the flagellum threading upward from that ghost of life into darkness, a single translucent filament whose beat at this frozen instant is invisible but whose consequence — the steady helical vortex drawing water across the collar mesh — is the reason this architecture exists at all. Below, the lorica stalk anchors to a marine snow aggregate whose diatom frustules catch the lateral light as tiny geometric mirrors, and beyond them the surrounding column is not simply empty but dense with implication: bioluminescent haze at the perimeter, bacterial rods too small to fully resolve, and the whole scene locked in the deep cold where choanoflagellates like this one have been filtering the ocean — and where their collar-cell body plan has persisted, essentially unchanged, as the closest living relative of every animal that has ever existed.

Before you, in absolute deep-sea darkness, rises a tower of living glass — the skeletal lattice of *Euplectella aspergillum*, a hexactinellid sponge whose silica spicules are fused at every intersection into a single rigid syncytial whole, the repeating chequerboard geometry so precise it reads as engineered rather than grown. The silica itself functions as true optical fiber, channeling the faint bioluminescent light of the surrounding water along the interior of each rod until the entire mesh glows cold blue-green from within, every spicule a self-luminous filament suspended in blackness, the cumulative structure a Gothic nave built from spun glass. Looking through the lattice wall, concentric shadow grids multiply inward as each cylindrical layer refracts and re-casts the luminescent geometry, while fractured spicule cross-sections at the nearest nodes reveal concentric lamination rings — pale growth bands in biosilica deposited around an organic protein template, the material transitioning from faintly amber at its dense core to glacial white at the outer sheath. Deep within the atrium, two commensal shrimp drift as warm amber-pink silhouettes rendered semi-transparent by the interior glow, living stained-glass figures sealed inside their cathedral for life, the full weight of the water column above communicated not by any sensation but by the perfect stillness of everything and the absolute darkness beyond the structure's halo.
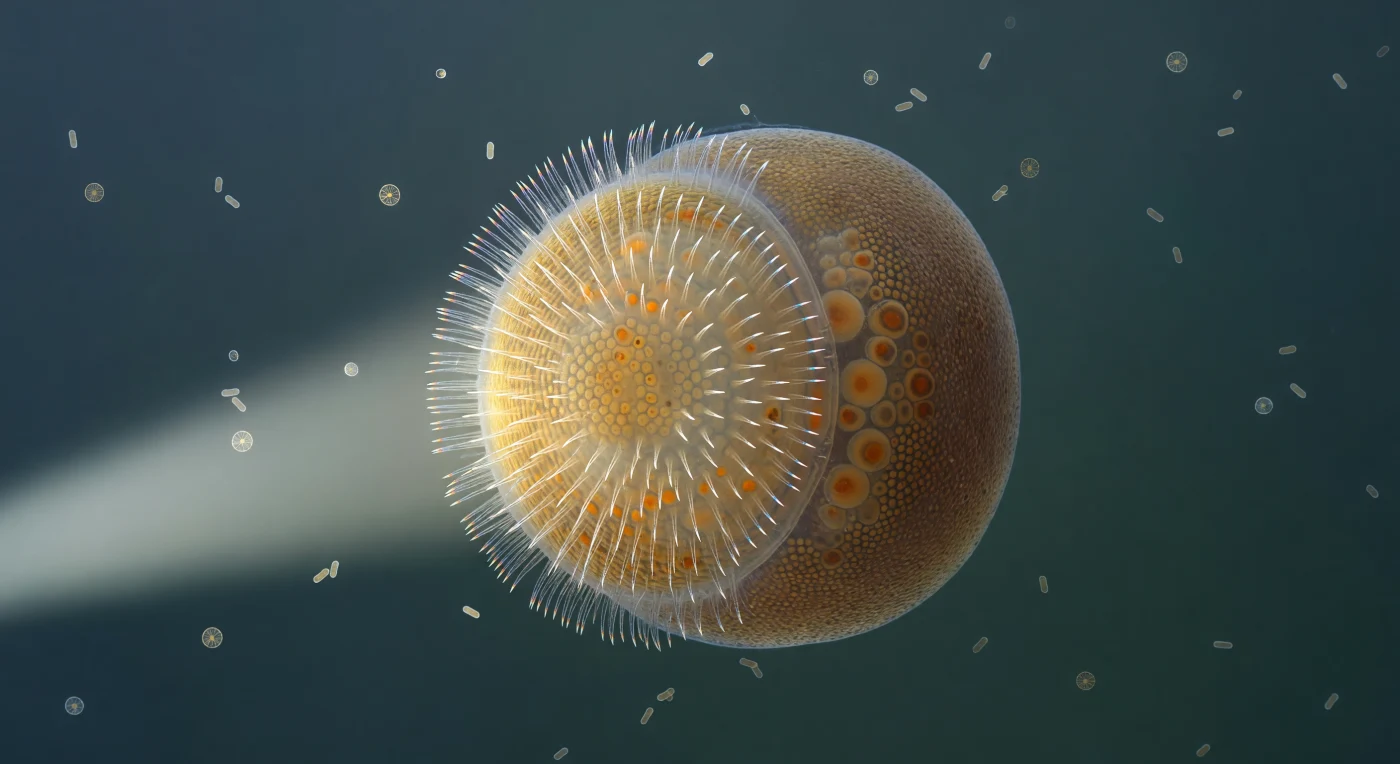
You hover in open water at the scale of a large dust mote, surrounded by a cool blue-green medium that presses against you from every direction with equal Brownian insistence, while before you an oval world roughly 400 micrometers across — the diameter of a fine sand grain — blazes with concentrated biological purpose. This is an amphiblastula larva of a calcareous sponge, a fleeting free-swimming stage that exists for only hours to days before settling onto a reef surface and inverting its own body to begin the sedentary filter-feeding life of its kind; its two hemispheres represent fundamentally different cell fates, the anterior choanoblast-precursors destined to become the flagellated choanocytes that will one day drive water through internal chambers, and the posterior archeocytes carrying dense lipid reserves that fuel the entire settlement and metamorphic transition. A single shaft of side-light carves the near hemisphere into a scintillating golden dome whose surface reads as a continuous vibrating fringe — thousands of cilia beating in coordinated metachronal waves, each transparent as a glass needle, their collective motion diffracting the light into a spectral corona of violet, cyan, and pale gold that wavers and resets in real time around the equatorial rim. The posterior hemisphere recedes into deep ochre shadow, its granular archeocyte masses pressing against one another in dense, yolk-heavy arrangement, their amber lipid inclusions visible as dark punctuations through the translucent wall — an entirely different country of cellular quietude pressed against the luminous, spinning anterior world. Around the larva, diatom frustules glint like engraved medallions and rod-shaped bacteria tumble in slow Brownian arcs, indifferent witnesses to a creature poised at the threshold between the planktonic and the permanent.

You are suspended at the lip of a living chimney — the osculum of a large barrel sponge — peering down into a four-centimeter circular shaft whose burnt-orange, iron-terracotta walls curve steeply into branching exhalant canals below, the entire interior suffused with a wavering cathedral light filtered down through the tropical water column above. What you are witnessing is not passive geology but continuous physiology: this sponge pumps water through its body at roughly 20,000 times its own volume per day, driven by thousands of flagellated choanocyte cells lining spherical chambers deep within the tissue, each cell beating its flagellum 30–60 times per second to draw seawater inward through microscopic ostia pores and exhale it, filtered, through this single chimney opening. The upwelling jet is made visible by micro-turbulence at its boundary with ambient reef water — a shimmering refractive haze laced with organic detritus, pale bacteria clouds, and glinting siliceous fragments, all the suspended material that the sponge has already processed and rejected or simply passed through. The outer surface radiates away in every direction — pitted with ostia, colonized by coralline algae and bryozoan lacework — while soft-focus staghorn corals and reef fish occupy the luminous background, reminders that this single organism, ancient in its body plan and unchanged in its cellular logic for over 600 million years, operates as a structural and biogeochemical anchor for the entire reef community around it.
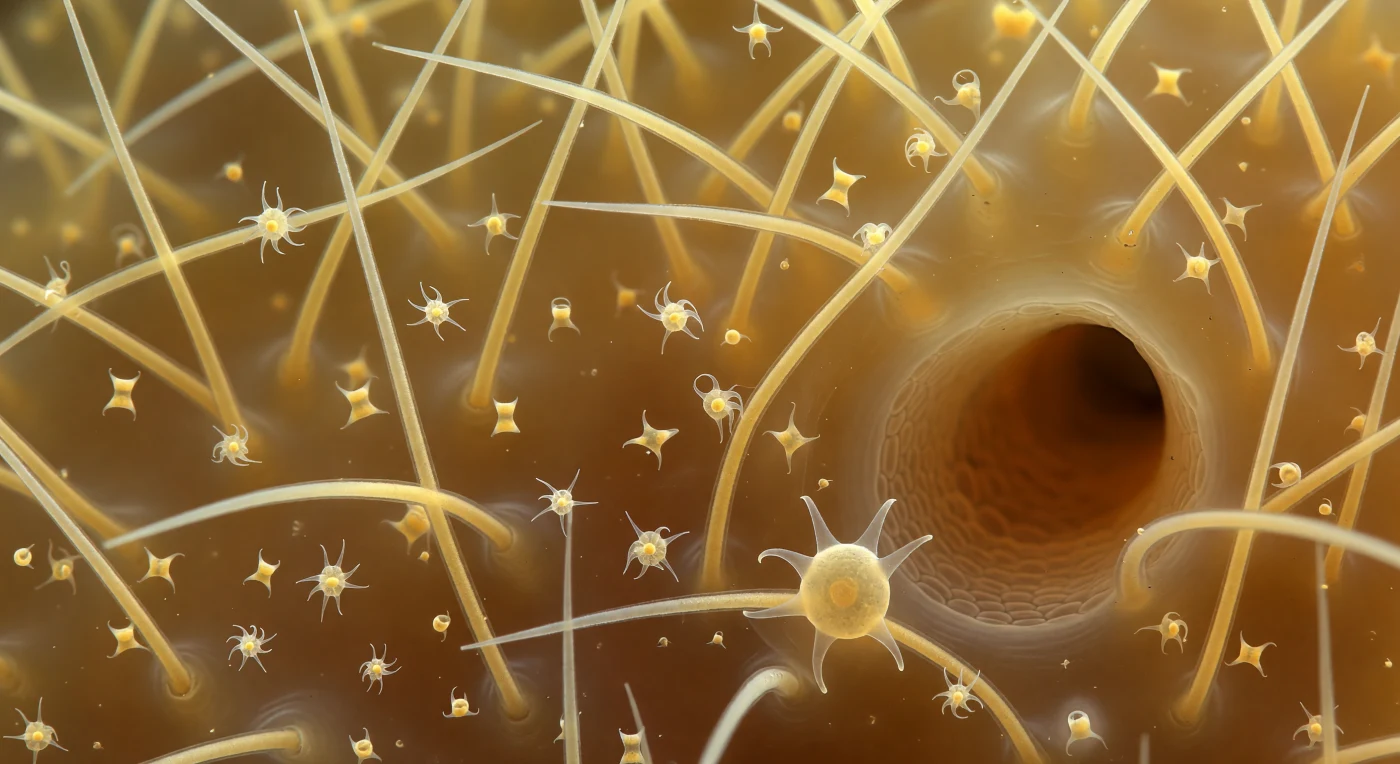
You drift suspended inside a living architecture that dwarfs all sense of normal proportion — the interior of a demosponge mesohyl at a scale where dust becomes cosmos, where the gelatinous matrix around you glows like backlit honey and every direction presents a new corridor of glass. The dominant structures are oxea megascleres, biogenic silica needles secreted by specialized sclerocytes, each tapered to both ends and semi-transparent, their curved lengths blazing with specular highlights as diffracted amber light moves through the gel medium — a colloidal hydrogel of proteins, proteoglycans, and mobile cells that constitutes the sponge's functional extracellular body. Threaded between spicule bases, spongin fibers — composite organic cables of a collagen-like protein unique to demosponges — form the tensile scaffold that binds this skeleton into mechanical coherence, while sigma microscleres and chelae rotating freely in the gel represent a secondary spicular armament whose exact role in structural reinforcement or predator deterrence remains an active subject of inquiry. The dark canal tunnel receding through the mid-ground is a fundamental unit of sponge hydraulic anatomy, a channel through which flagellum-driven current will eventually pass from incurrent ostia to the osculum, sustaining a filtration rate that can exceed the sponge's own body volume twenty thousand times over in a single day. On one foreground spicule, a single archaeocyte grips with extended pseudopods — this totipotent migratory cell capable of differentiating into virtually any sponge cell type, patrolling the mesohyl and embodying, in miniature, the distributed cellular intelligence of an animal without nerves, muscles, or organs.
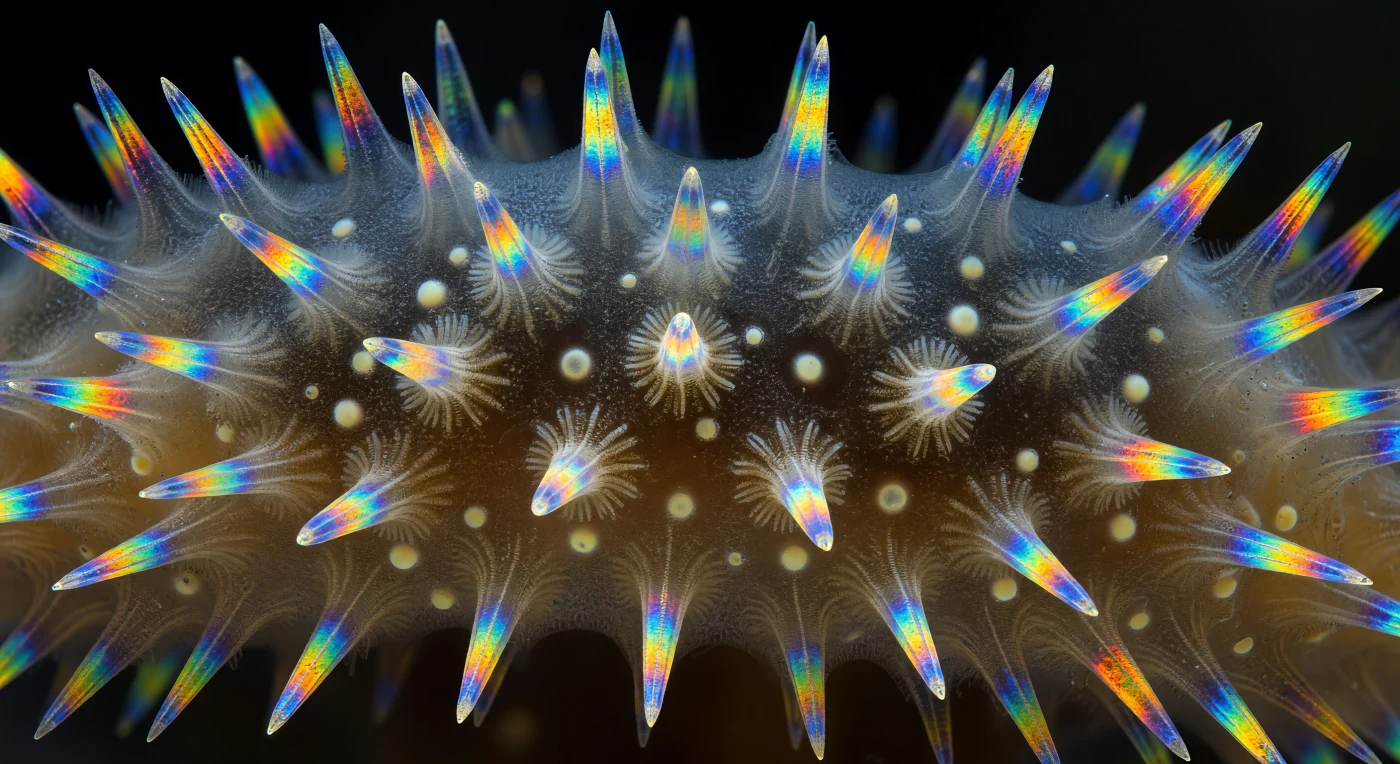
Suspended within the sectioned wall of *Sycon ciliatum*, you look out across an interior architecture that reads less like biology than like a cathedral's clerestory rendered in mineral fire: radial canals bore through the amber-gray mesohyl like nave aisles, their walls furred with choanocyte collars barely visible in the transmitted glow, while calcite triactine and tetractine spicules blaze in hallucinatory birefringent color—electric cobalt, sulfur yellow, molten orange, cold emerald—each crystal lit from within by polarized light against an absolute black void. Two rays of every triactine lie flat in the plane of the wall, throwing hard gem-sharp color gradients along their length, while the third ray punches perpendicularly outward like a needle, its tip catching a specular point of white-gold light. Where spicules cross, their interference patterns stack into iridescent aureoles—amber bleeding into violet into electric teal—the optical equivalent of medieval stained glass, except that each blazing element is a single calcite crystal secreted by a living sclerocyte cell within a matrix of fibrous extracellular gel. The scene unfolds across layered depth: foreground spicules in razor-sharp chromatic definition, mid-ground canal walls dissolving into warm translucency, the far background fading to pure blackness where polarized light cannot penetrate—and the vertiginous realization settles that this entire luminous architecture, part mineral, part living organism, part pure optics, fits within a structure no larger than a human fingernail.

You hover face-to-face with a tower of *Aphrocallistes vastus*, a glass sponge whose entire skeleton is woven from fused silica spicules — not secreted mineral armor but a continuous syncytial lattice of living glass, the same material as optical fiber, built hexagonally at the microscopic level into a structure that at this scale reads as vaulted cathedral architecture rising in cream-white columns from a seafloor carpeted with its own broken spicule debris, shards of biogenic glass glittering in the ROV beam like cold diamonds scattered across gray calcareous silt. These hexactinellid sponges are among the oldest reef-builders in the Northeast Pacific, their colonies dated to thousands of years of incremental accretion, each tower growing only millimeters annually in water near freezing and utterly lightless — yet the silica lattice conducts the artificial floodbeam along each glassy strut by total internal reflection, lighting the structure from within so that nested chambers two to five millimeters across glow like backlit alabaster receding through translucent walls, the whole spire radiating a warm halo into water that has no business being warm at all. Brittle stars drape their banded arms through the mesh openings as though the sponge were scaffolding built for them, rockfish hang motionless in the inter-tower corridors exploiting the architectural shelter, and marine snow descends in absolute vertical stillness through the beam — organic aggregates drifting downward at millimeters per second, the slow rain of surface productivity reaching a filter-feeding reef that pumps tens of thousands of times its own volume through living glass every single day. At the edge of the light, a bioluminescent smear drifts past and vanishes, leaving nothing but the cold, the dark, and the reef extending in every direction far beyond what can be seen.

You are suspended just above a submarine rock face, your entire field of view filled by a terrain that, at this scale, reads as a vast inhabited plateau — the crustose coralline algae beneath you a fractured magenta-crimson continent of calcified polygonal plates, their raised edges dusted with pale lavender, while diatom frustules rise between them as perfect glassy monuments of silica catching the diffuse blue-green light filtering down through the overlying water column. At the center of this world, a calcareous sponge larva roughly 300 microns across is frozen mid-metamorphosis, its ciliated hemisphere collapsing inward along the apical axis in morphogenetic inversion — the ancient developmental program of body-plan construction playing out as the outermost cells flatten into irregular pavement tiles, their boundaries visible as faint pale lines spreading outward from the central attachment point in concentric waves across the living biofilm below. This biofilm is itself a world: a translucent amber-gold matrix of exopolysaccharide, gelatinous trails looping through shallow depressions in the carbonate surface, shimmering faintly with colloidal iridescence as dissolved organics catch the raking light. The larva's thinnest peripheral margin transmits light like stained glass — warm amber where future pinacocytes are pressed to a single-cell thickness — while its invaginating center clusters in deeper ochre, a nascent glycocalyx throwing soft interference colors of pale teal and gold wherever membrane geometry bends the downwelling blue-green light, the entire scene a luminous record of the evolutionary transition between single-celled ancestors and the first animals to build a body.
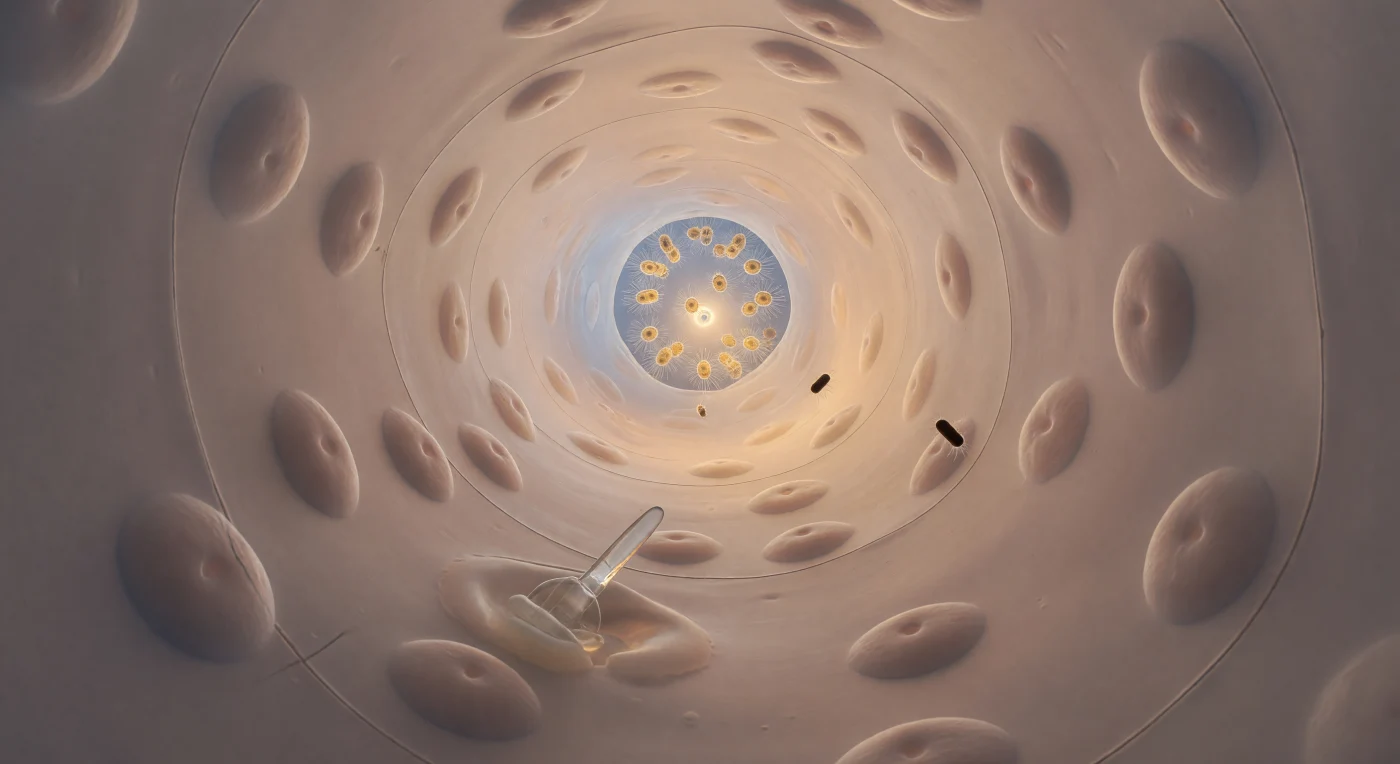
You are suspended at the entrance of a living conduit, gazing down a gently curving cylinder of flesh and fluid whose far end glows with the warm amber light of a choanocyte chamber — a small, burning aperture that anchors your perspective the way a candle anchors a dark room. The walls enclosing you are built from endopinacocytes pressed impossibly flat against the underlying mesohyl, their surfaces polished and faintly translucent, interrupted only by the soft dome of each nucleus rising like a submerged stone beneath wet silk, and by the hairline seams where cell margins meet in nearly invisible junctions. Here, water does not flow so much as it is persuaded forward: at this diameter, viscosity reigns absolutely, and the Stokes-regime current carries its passengers — rod-shaped bacteria drifting past like lacquered cylinders, one tumbling end-over-end as Brownian noise briefly claims it before the laminar flow reasserts its patience — without turbulence, without urgency, with a quality closer to procession than motion. Embedded in the lower wall, a spicule of calcium carbonate catches transmitted light along its entire length, and pressed against its base, an amoeboid archaeocyte deforms around the obstacle in slow, warm increments, cytoplasm bulging through a cell junction as it migrates through tissue that has been filtering ocean water through passages exactly like this one for the better part of six hundred million years. Behind you, cold blue-white ocean light floods through the canal mouth; ahead, through the prosopyle no wider than five microns, the choanocyte chamber blazes with the collective industry of dozens of flagellating cells whose beating, were sound possible here, would be the quietest and most ancient whisper in animal history.
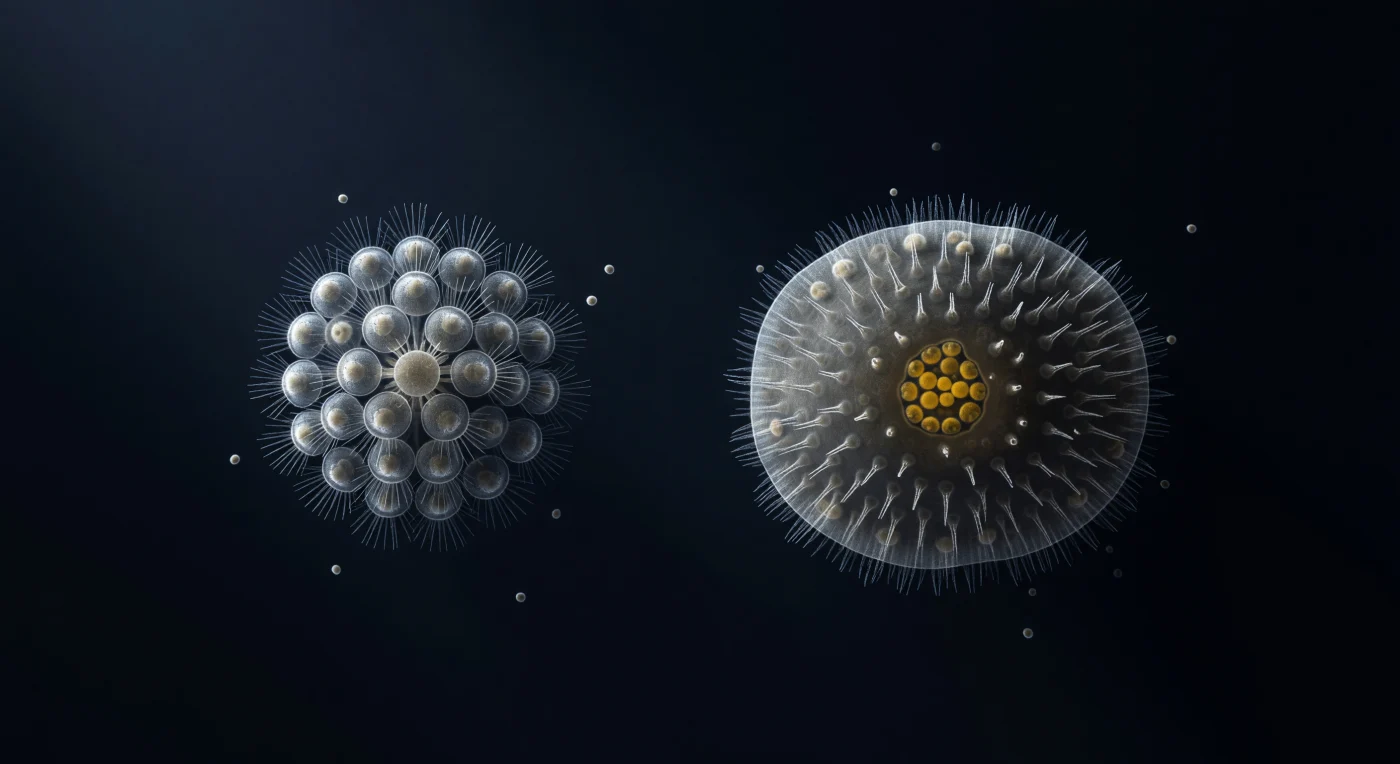
You hover in a pressurized darkness that reads as oceanic, a DIC-illuminated void of deepest blue-black through which a single oblique light rakes from upper-left, sculpting every membrane into bas-relief — silver-white highlights blazing on exposed cell faces, the opposite edges dissolving into shadow so complete they seem to merge with the surrounding medium. To your left, a 32-cell rosette colony of *Salpingoeca rosetta* hangs in precise biological architecture, each choanoflagellate cell presenting its collar-and-flagellum unit outward in an achromatic arrangement of silver needles and pale translucent cytoplasm, the intercellular bridges at the colony's basal core constituting some of the oldest known molecular machinery for multicellular cohesion — a structural preview of every animal body that will follow across the next seven hundred million years. One hundred microns of open water separates this colony from a calcinean sponge parenchymella larva that occupies the right side of the field in dense, semitransparent mass: its exterior layer of ciliated cells catches the DIC light as a silver fringe at the periphery, while its interior cell mass grades toward a posterior cluster of yolk-laden cells burning in the only warm amber tones in an otherwise monochromatic scene, and within the anterior hemisphere, a handful of deeper cells already betray incipient collar-cell morphology — minute apical condensations rehearsing the choanocyte plan before larval settlement has even begun. The gap between colony and larva reads as empty, yet the shared architecture of collar microvilli, the common logic of flagellum-driven filtration, and the near-identical ultrastructure of choanoflagellate cells and sponge choanocytes charge that hundred-micron interval with the entire conceptual weight of animal origins, the two organisms functioning simultaneously as living taxa and as the closest available material witnesses to the transition that made complex multicellular life possible.

You are suspended just inside the bisected wall of a freshwater sponge's survival capsule, looking inward across a space that reads, at your scale, like the nave of a cathedral cleaved open by a geological fault. The wall to your left is a laminate fortress in three distinct zones: an outermost membrane of taut, faintly iridescent amber; behind it a palisade of amphidisc spicules standing in pale cream spongin cement, each one a dumbbell of crystalline biogenic silica whose flared rotular disc-ends interlock like ornate frosted-glass rondels, casting prismatic glints of icy white and ghost-blue as transmitted light refracts through their shafts; and finally a smooth, honey-gold inner spongin layer that curves inward to open onto the interior. Beyond that boundary, the cavity glows with saturated amber and saffron warmth — archaeocytes packed shoulder-to-shoulder, each cell a swollen globe gorged with lipid-droplet reserves that make them shine like oil-filled lanterns, the entire mass held in chemical suspended animation against winter. To the far right, a micropyle pore tube breaches the armor in cross-section, its cylindrical passage currently stoppered by a tight column of pale cells pressed membrane-to-membrane, the circular opening at its far end gazing outward into murky, cold olive-grey sediment water — the contrast between that cold exterior and the incandescent interior giving the whole scene the visual weight of a geode cracked open in deep winter, every surface radiating pressure, density, and latent biological potential.

You hover centimeters above the dark, waterlogged timber, your entire field of view consumed by a vivid grass-green crust spreading across the wood's shadowed underside — Spongilla lacustris, a freshwater sponge whose color comes not from its own cells but from the dense populations of symbiotic green algae packed into its living tissue, converting every shaft of dappled stream light into photosynthetic energy that suffuses the surface with an almost interior glow. The texture is deceptive: what reads as soft velvet is in fact a living matrix of cells and silica, the sponge's skeletal spicules protruding as fine glass needles across every millimeter of surface, each silica tip catching light like a frost crystal and scattering it into a soft halo of microglints that gives the whole crust a faint luminous bristling. Embedded within this green tissue, dark mahogany spheres press upward like buried cobblestones — the gemmules, each roughly half a millimeter across and armored in their own radial spicule architecture, ancient survival capsules capable of lying dormant through winter and drought while the living tissue around them actively pumps thousands of times its own volume of stream water each day, filtering bacteria and dissolved organics through choanocyte chambers that have operated on this same fundamental plan for over six hundred million years. Tiny ostracods roll across the surface like seed pearls through velvet, pale flatworms glide in translucent arcs, and above everything the stream water rises as a cathedral of cool green clarity, its surface a shifting silver ceiling that sweeps alternating light and shadow across the sponge in slow waves, the entire scene a reminder that this unassuming green crust on dead wood represents one of the oldest surviving animal body plans on Earth.

You are hovering less than a millimeter above the outer skin of a living tropical demosponge, and the world beneath you fills the horizon in every direction like an alien tidal flat seen from a low drone — except every surface breathes. The exopinacoderm spreads as an undulating mosaic of flattened polygonal tiles, each cell boundary raised into a shallow ridge saturated in deep burnt-orange and saffron carotenoid pigment, their surfaces glistening with a thin aqueous film that gives the landscape a wet, lacquered quality somewhere between freshly glazed ceramic and sun-warmed coral rubble. Scattered across this plain at irregular intervals, the ostia punctuate the surface like volcanic calderas — circular to subtly oval depressions, some fully dilated with porocyte walls retracted to reveal the absolute darkness of inhalant canals descending into the sponge's interior plumbing, others contracted to pale-rimmed slits barely admitting light, the surrounding pinacoderm tiles faintly depressed and paler at their rims as continuous negative pressure draws water downward at rates that will collectively process tens of thousands of times the sponge's own body volume each day. Between the ostia, a three-dimensional biofilm of rod bacteria, coccoid clusters, and filamentous cyanobacteria weaves through strands of mucopolysaccharide that catch the downwelling reef light as iridescent threads, while a small polychaete has surfaced from one open canal mouth with palps fanning in slow arcs through the boundary layer, and a copepod arriving at the upper edge of the scene refracts ambient blue through its transparent carapace before being deflected, invisibly but decisively, by the pressure gradient exhaled from a distant osculum.
















