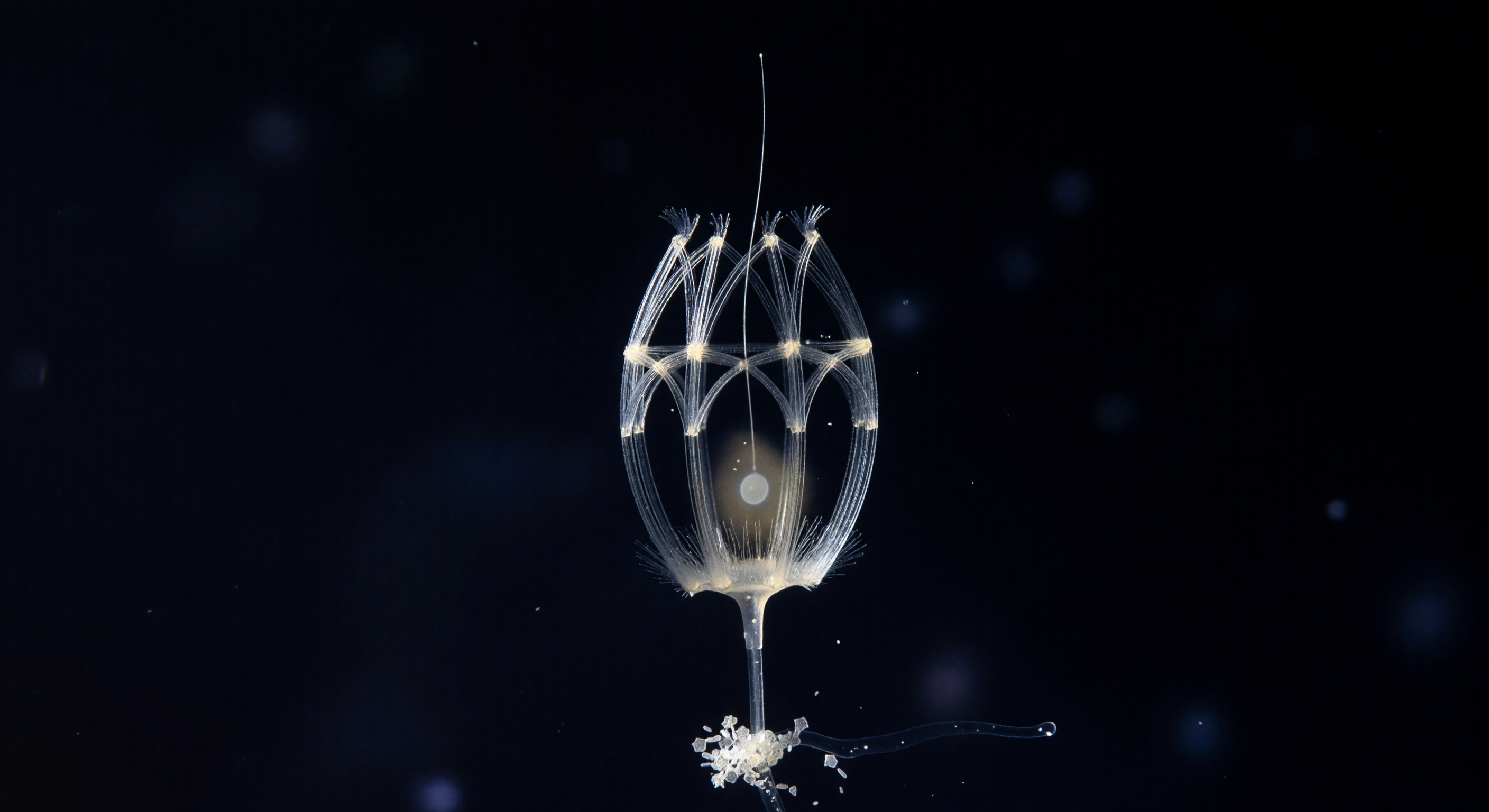
In the lightless reaches of the mesopelagic zone, a single *Diaphanoeca grandis* cell presents itself inside its self-assembled silica lorica — a gothic lantern of costal strips roughly 20 micrometers across, constructed from two interlocking rod systems: longitudinal ribs arching from base to apex like vaulting bones, cinched by transverse hoops, each silica filament blazing white under raking sidelight while the spaces between them breathe open as pure black negative space. The lorica is not decorative but functional, a rigid external scaffold secreted by the cell itself from dissolved silicic acid drawn from the surrounding seawater, its geometry channeling the flagellar current inward through the open anterior pole so that bacteria and dissolved organics are swept through the microvilli collar in a continuous filtration the cell maintains without rest. Within the cage, the living body is barely legible — a pale cytoplasmic haze in the lower interior, a faint nuclear ellipse, a corona of collar microvilli dissolving before they fully resolve, and the flagellum threading upward from that ghost of life into darkness, a single translucent filament whose beat at this frozen instant is invisible but whose consequence — the steady helical vortex drawing water across the collar mesh — is the reason this architecture exists at all. Below, the lorica stalk anchors to a marine snow aggregate whose diatom frustules catch the lateral light as tiny geometric mirrors, and beyond them the surrounding column is not simply empty but dense with implication: bioluminescent haze at the perimeter, bacterial rods too small to fully resolve, and the whole scene locked in the deep cold where choanoflagellates like this one have been filtering the ocean — and where their collar-cell body plan has persisted, essentially unchanged, as the closest living relative of every animal that has ever existed.
Other languages
- Français: Cage Lorica dans l'Obscurité
- Español: Lorica Geométrica en Oscuridad
- Português: Lorica em Trevas Profundas
- Deutsch: Lorica Gitterwerk im Dunkel
- العربية: قفص لوريكا في الظلام
- हिन्दी: अंधेरे में लोरिका जाल
- 日本語: 暗闇のロリカ格子
- 한국어: 어둠 속 로리카 골격
- Italiano: Lorica Wireframe nell'Oscurità
- Nederlands: Lorica Draadmodel in Duisternis