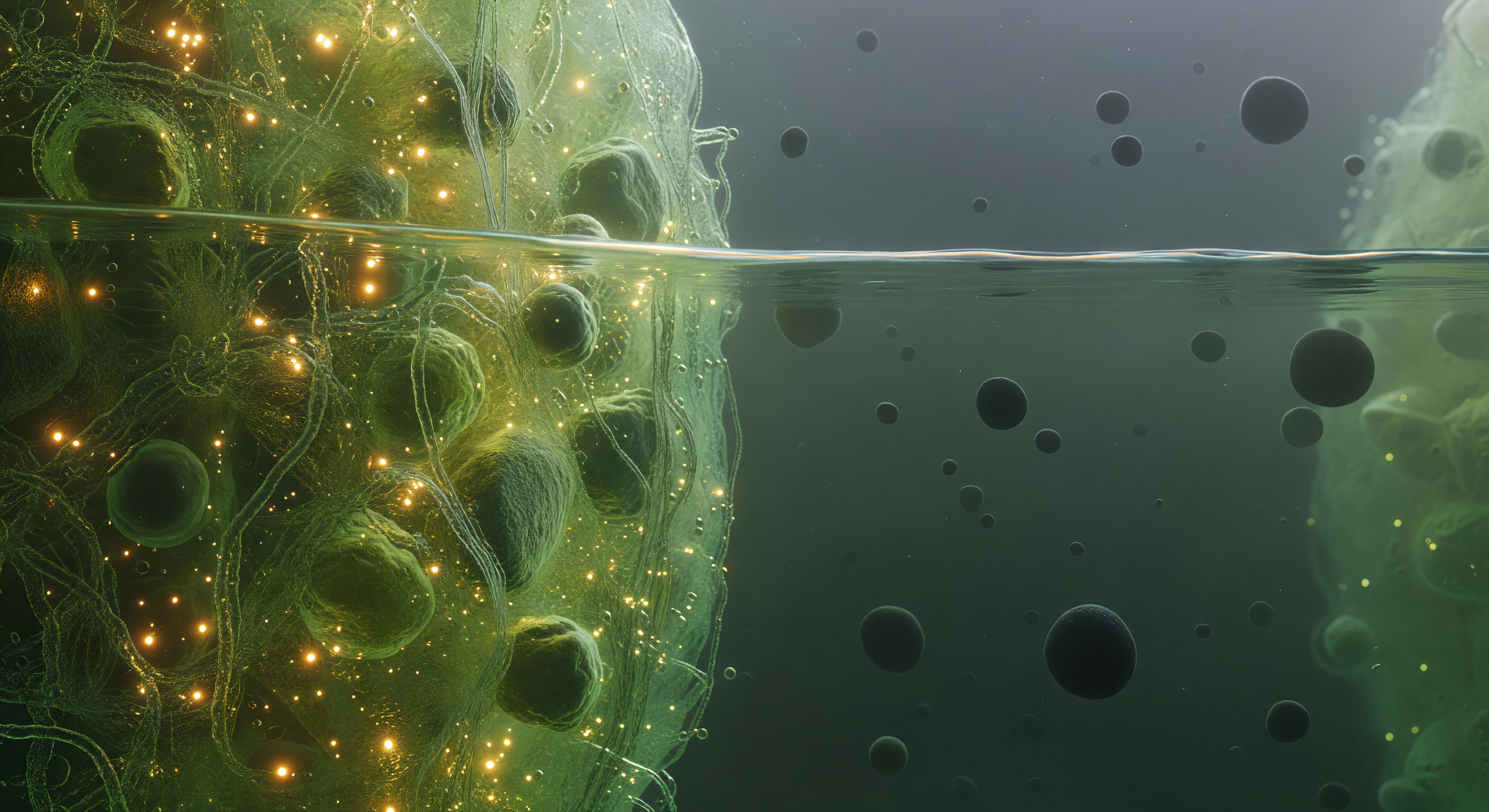
You float precisely at the threshold of two immiscible biological worlds, straddling the phase boundary of a stress granule — a membraneless organelle assembled not by lipid walls but by the thermodynamic logic of liquid-liquid phase separation, where intrinsically disordered proteins and mRNA chains spontaneously partition into a dense, self-organized condensate phase coexisting with the surrounding dilute cytoplasm. To your left, the condensate interior glows with a saturated green-gold luminescence, its molecular mesh so densely packed — proteins and RNA entangled at concentrations approaching hundreds of milligrams per milliliter — that light scatters and pools into an amber warmth, the whole medium trembling with Brownian agitation as ten thousand macromolecules press and slide against one another in a viscoelastic medium that behaves somewhere between liquid and gel. Directly before you, the interface itself holds as a sharp thermodynamic discontinuity, not a gradient but a true phase boundary maintained by surface tension, shivering with capillary fluctuations at the nanometer scale — a faintly iridescent, undulant seam where the dense condensate ends absolutely and the dilute phase begins, catching the interior light in a thin luminous caustic that is the only hard edge in this otherwise fluid and probabilistic landscape. To your right, the dilute cytoplasm opens into a comparative void, a cool blue-green haze where individual ribosome complexes drift as dark matte boulders separated by distances that feel like open ocean after the claustrophobic interior — each one a 25-nanometer asymmetric assembly of RNA and protein, enormous against the diffuse background scatter, fading into a particulate murk where the next organelle boundary is only a vague brightening at the limit of visibility.
Other languages
- Français: Frontière de Gouttelette Liquide
- Español: Límite de Gota Líquida
- Português: Fronteira da Gota Líquida
- Deutsch: Flüssigphasen Tröpfchengrenze
- العربية: حدود قطرة الطور السائل
- हिन्दी: तरल चरण बूंद सीमा
- 日本語: 液相液滴の境界
- 한국어: 액체 방울 경계면
- Italiano: Confine della Goccia Liquida
- Nederlands: Vloeistoffase Druppelgrens