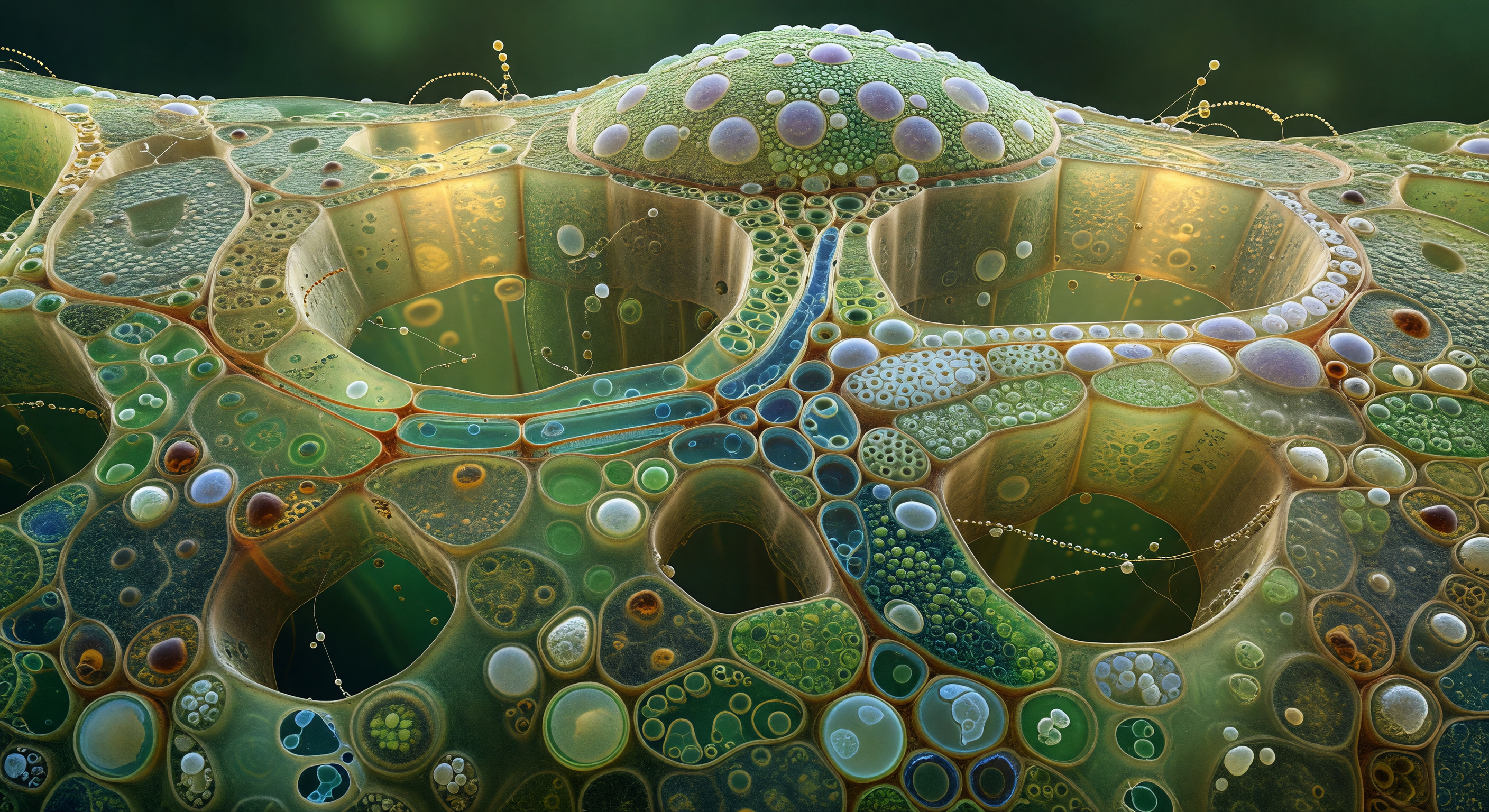
You are standing inside the circulatory infrastructure of a living tree, looking down a hollow lignified cylinder roughly the diameter of a human hair, its amber walls dressed in hundreds of bordered pits arranged with the obsessive regularity of a tiled vault. Each pit is a shallow circular recess only a few micrometres across, its aperture bridged by a translucent membrane ghost that once acted as a pressure-sensitive valve between neighbouring vessels, allowing water to pass under tension while resisting the catastrophic intrusion of air. The vessel around you is dead by design — its cell contents long since autolysed to leave only this secondary wall of lignified cellulose, one of thousands of overlapping pipe segments that together form the xylem conduit system drawing water from root hairs to canopy leaf under negative hydraulic pressure, a column in continuous tension rather than being pushed from below. At the far end of the corridor, that tension has already failed: a single air embolism, convex and mercury-bright, spans the full lumen like a sealed mirror, its surface-tension contact line pinned against the lignin wall in a precise dark ring, halting all further water movement through this segment entirely. The pit gallery behind you is reflected in its curved face in miniature — a hundred tiny circles converging to a point, the whole architecture of cohesion-transport summarized in the distorted image held within a bubble of failure.

You stand at the base of a vertical world, looking up through a living cathedral whose green columns rise seventy micrometers overhead — palisade mesophyll cells packed wall to wall, their semi-translucent amber-green surfaces studded with hundreds of chloroplasts pressed flat against the inner face like emerald tiles in a Byzantine mosaic, biconvex and deep viridian at their cores, each one a self-contained solar collector whose stacked thylakoid grana are just visible as darker internal banding when the light strikes obliquely. Above, the upper epidermis glows like ground frosted glass, its waxy cuticle diffusing the incoming solar radiation into a luminous, near-shadowless downpour that slides between columns in soft curtains, catching the aqueous film on every cell wall as a faint specular gleam before deepening into charcoal in the intercellular crevices — those narrow black alleyways between towers that open laterally into the spongy mesophyll beyond, a labyrinthine gas-exchange network through which carbon dioxide diffuses toward the chloroplast-lined walls and oxygen drifts outward. The sheer density of chloroplasts transforms every wall into a continuous cold-green stained-glass panel, overlapping jade shadows layering one upon the next as lens after lens bends and re-emits the light, and where two chloroplasts nearly touch across an air gap their lime-bright margins glow like neon tubing against absolute blackness. Looking straight up along the canyon between two adjacent columns, the walls converge sharply, the ceiling brightens, and the perspective collapses into a dizzying vertical tunnel — this is photosynthesis not as chemistry but as architecture, a structure of pigment and cellulose quietly converting every photon that survives the journey through the cuticle into the molecular currency of life.
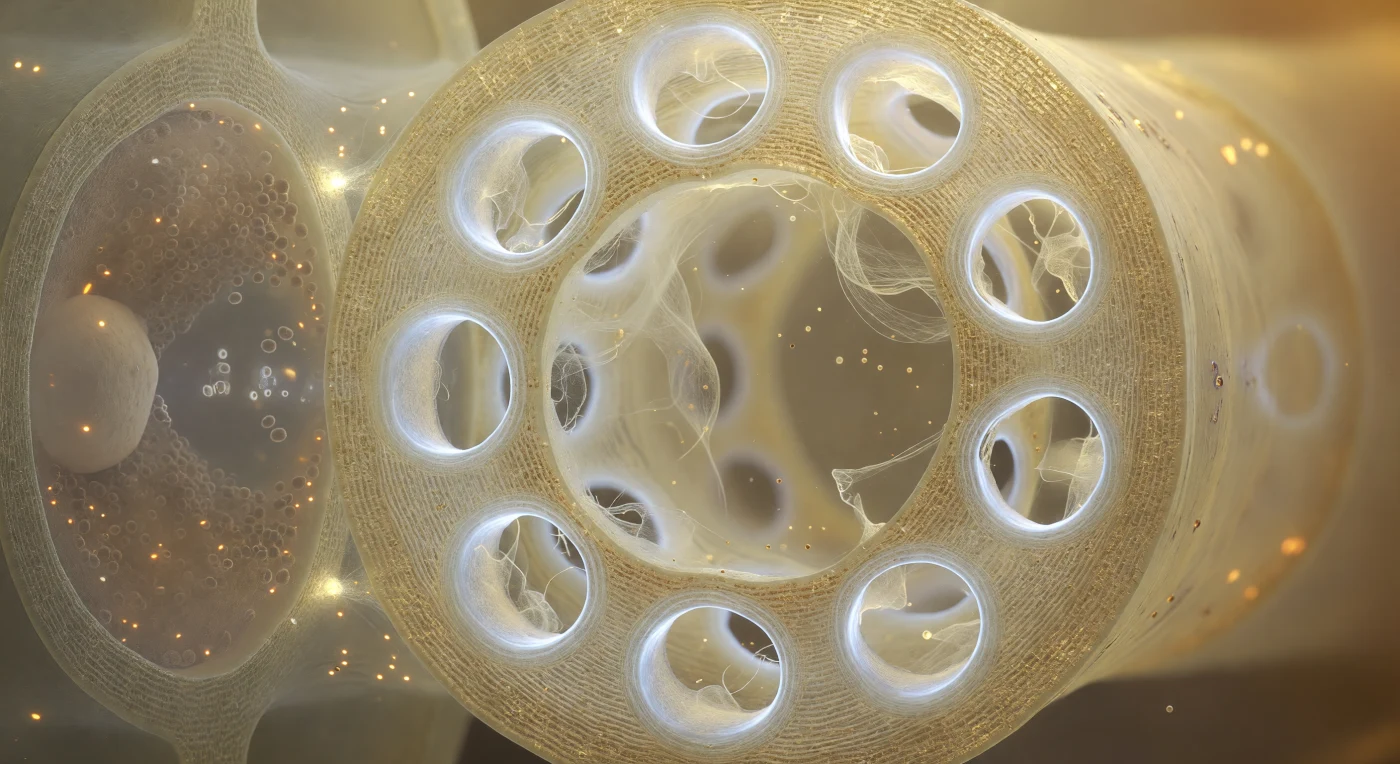
You are standing inside the hollow barrel of a living sieve tube element, and the end wall fills your entire field of view like a cathedral screen — a pale gold partition of cellulose three micrometers thick, perforated by twelve circular pores each rimmed with a luminous callose collar that glows cool and milky-white, narrowing each throat to a fraction of its original diameter through dense concentric lamellae of biopolymer packed like polished bone. Through these portals, P-protein filaments drift as a translucent cream mesh, individual strands billowing gently in the nearly invisible current of phloem sap as it pulses at roughly a meter per hour from one chamber to the next, driven by the osmotic pressure differential between sugar-loading leaves and sugar-consuming roots — the Münch pressure-flow mechanism rendered as a slow, warm tide. To your left, through the thin shared lateral wall, the companion cell presses close like a darkened room seen through frosted glass, its cytoplasm dense with mitochondria and ribosomes that metabolically sustain the enucleate sieve element beside it, the two cells connected by specialized plasmodesmata — pore-plasmodesmata units — that are just barely resolvable as bright punctures transmitting light across the shared boundary. The overall atmosphere is one of immense biological intimacy, warm amber lumen against cold white callose, luminous hollow space pressed by dense metabolic shadow, the entire architecture of long-distance transport in a vascular plant existing at a scale where a single sieve pore becomes a cathedral archway and a drifting protein filament becomes a hanging curtain trembling in an invisible slow tide.
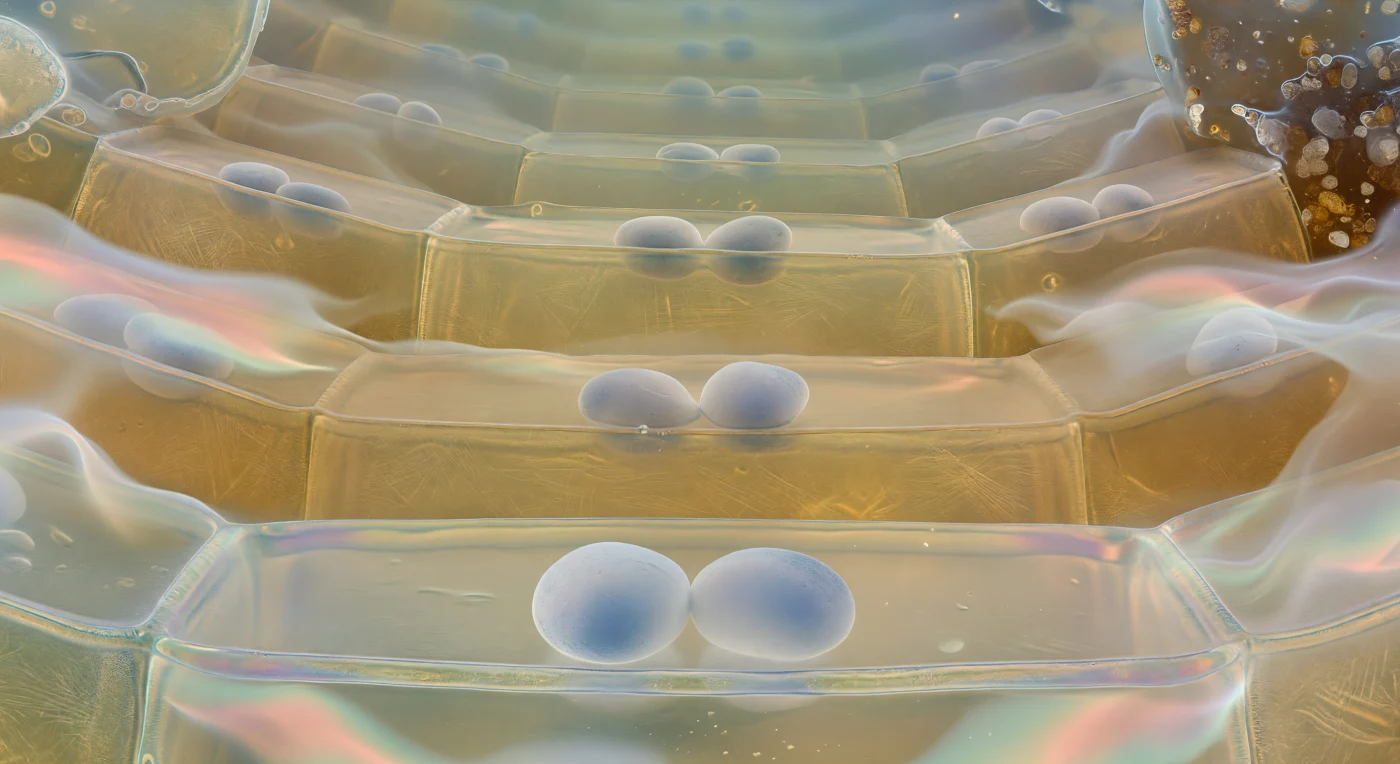
You are suspended just above the floor of a single columella cell, looking upward into a cathedral of living architecture that recedes tier upon tier into luminous amber-green distance. Directly before you, two or three amyloplast statoliths rest motionless against the lower plasma membrane — dense, chalky-white ovoids the size of boulders at this scale, their matte surfaces shading to faint blue-grey where transmitted light filters through, pressing gently into the membrane like polished stones dropped onto a drumhead; these are the plant's gravisensors, organelles dense with starch whose sedimentation toward the cell's gravitational floor is the root's only means of knowing which way is down. The membrane beneath them is barely visible, a soap-film meniscus shimmering with pearlescent interference colours — pale violet, ice-blue, gold — its tension holding the clear, almost optically empty cytoplasm in suspension, a medium so transparent it reads as pure water coloured only by a ghost of golden warmth where diffuse light grazes through. Rising overhead, the anticlinal cell walls curve upward as broad planes of translucent amber glass, the cross-hatched weave of cellulose microfibrils caught inside like a ghostly grid in thick honey-coloured resin, and through the semi-transparent ceiling you can make out the floor of the next cell tier, its own statoliths appearing as cloudy white crescents hazed by the intervening wall — a geometry that repeats, tier above tier, the successive ranks of cells growing smaller and more distantly lit, the whole stack receding to a cool blue-green twilight at the pyramidal apex where the columella dissolves upward into the meristematic zone. At the periphery, the outermost border cells are losing structural integrity, their membranes unravelling into a shimmering hydrogel haze of root mucilage — a polysaccharide secretion that refracts transmitted light into drifting spectral ribbons of salmon-pink and pale aquamarine, coating the surrounding mineral grains with a glistening biochemical halo that lubricates the root's passage through soil and negotiates the chemical boundary between plant and earth.

A star-burst fortress of calcium oxalate fills the entire visual field, its forty individual facets radiating from a dense central nucleus like the blades of a rose window carved from living mineral — each spike tapering to a stiletto point that dissolves into absolute darkness. This is a druse crystal, roughly 55 micrometers across, precipitated inside a parenchyma vacuole when dissolved calcium and oxalate ions reached saturation and cascaded outward from a seed nucleus in a burst of crystallographic inevitability; the plant uses such inclusions to sequester excess calcium, neutralize organic acids, and deter herbivores, locking metabolic waste into insoluble geometry. Under crossed polarizers, the surrounding aqueous vacuolar contents vanish into a perfect void, and only the birefringent crystal generates color — deep cobalt blue across the broadest faces where light retardation is maximal, burning through burnt sienna and raw amber at oblique angles, erupting into near-incandescent gold along the razor ridgelines where constructive interference stacks wavelengths from two adjacent facets simultaneously. At the very periphery of this black cathedral, a faint pale-gold curve betrays the parenchyma cell wall — compressed cellulose microfibrils forming a laminate fortification whose own birefringence is a whisper against the mineral spectacle at center stage — a reminder that this entire blazing structure exists sealed and motionless inside a single living cell, a frozen record of ion chemistry pulsing with color in the biological dark.

You are suspended weightless above a gently convex hemisphere roughly the width of a human hair, its surface tiled in a near-perfect honeycomb of isodiametric cells — each about twelve micrometers across — whose cellulosic primary walls form a mint-ivory lattice, translucent enough that diffuse light scatters through them like fog through rice paper, illuminating each facet from within without casting a single hard shadow. At the center of every hexagonal tile, a dark oval nucleus presses against the frosted interior like a smooth river stone seen through glass, betraying the extraordinary density of these cells: barely vacuolate, packed with ribosomes and organelles, their cytoplasm a faintly granular amber-gel that leaves no empty space, because these are among the youngest cells the organism will ever produce. This is the shoot apical meristem, the organism's generative summit — a dome of pluripotent initials whose division planes are still fresh, sustaining a pool of stem-like cells in the central zone while the peripheral zone recruits founder cells outward, a living geometry of potential continuously consuming and renewing itself. At the flanks, two leaf primordia swell upward like warm-gold foothills, their cells already elongating along a directional axis, the regular honeycomb giving way to a brick-course geometry across a gradient zone just two or three cells wide — the boundary where identity negotiates itself in real time. The entire scene carries the quality of something both ancient and perpetually unfinished, a summit that has never stopped building itself outward into form.

Looking upward from the substomatal cavity, you find yourself suspended inside a living architectural vault — two kidney-shaped guard cells arch overhead like the keystones of a cathedral door, each one swollen taut with osmotic pressure, their surfaces gleaming with an aqueous film that transforms oblique light into long specular ridges of wet jade. The inner walls of these cells are visibly thickened, their cellulose microfibrils packed into pale ivory laminations whose structural asymmetry is the sole mechanical reason the pore gapes open rather than collapsing — a seven-micron slit of cold blue-white sky visible at the apex, backlit by open atmosphere and sharply contrasting with the warm chlorophyll-green pressing in from either side. Along the inner face of each guard cell, twelve chloroplasts are marshalled in a loose procession, lens-shaped bodies four to six microns long, their photosynthetic membranes glowing with an almost self-generated luminescence, pressed there by the very turgor pressure that holds the stoma open — a feedback written directly into the cell's geometry, since the same osmotic engine that drives guard cell inflation also energizes the chloroplasts performing the photosynthesis that fuels it. Around this incandescent frame, the surrounding epidermal pavement cells recede as pale, featureless panes, large-vacuolated and nearly colorless, while below your vantage point the substomatal cavity dissolves into a spongy mesophyll labyrinth where wet cell surfaces catch condensed vapor into gleaming meniscus films and scatter light into a soft biological fog.

You stand suspended inside the vascular cambium at the peak of spring's generative surge, enclosed within a corridor of cells so attenuated they seem less like walls than suggestions of enclosure — each fusiform initial stretching before you like the nave of a submerged cathedral, its pale straw-yellow cytoplasm glowing with a lantern's diffuse warmth, its nucleus a large opalescent sphere drifting mid-cell through amber haze. This is the vascular cambium, a single meristematic layer of fusiform initials poised between two diverging fates: to your left, daughter cells already committed to xylem are armoring themselves with concentric bands of lignified secondary wall, the amber deepening cell by cell into tawny ochre as living protoplasm is replaced by hollow, engineered tubes built for water under tension; to your right, nascent sieve elements dissolve themselves purposefully — nuclei fading, cytoplasm thinning to a permeable gel, callose halos forming around sieve plate pores as each cell surrenders its individuality to become a conduit for photosynthate flowing at nearly a meter per hour. The compression between these two solidifying empires is immense and quiet: the cambium itself is under mechanical stress from expanding xylem on one side and the bark's resistance on the other, yet these cells — barely twenty micrometers across — sustain the division rates that will build an entire season's growth, their walls so thin they register less as surfaces than as the living membrane of a world caught perpetually between becoming wood and becoming flow.

Before you hovers an architectural wonder of botanical chemistry — the glandular secretory head of a tomato trichome, its four disc-shaped cells arranged in a low crown, each one a slightly flattened dome of translucent green-gold cytoplasm pressing faintly against the inner face of a cuticle that has been forced outward into a taut, amber-tinted blister of accumulated essential oils, refracting oblique light across its equator in a slow liquid shimmer, poised at the threshold between containment and release. Within that blister, fine density gradients betray the layered secretion of different terpenoid compounds — monoterpenes, sesquiterpenes, and volatile phenolics synthesized in the leucoplasts of the stalk cells below and actively transported upward through the crown cells into this subcuticular reservoir, where hydrostatic pressure builds until the cuticle ultimately ruptures or an organism brushes against it. The six-tiered stalk descends below the crown in diminishing cellular tiers, each pair of cells anchored by thickened pectin-cemented primary walls, the whole column rooted in a reinforced base cell splayed into the surrounding epidermis — a dicot surface of jigsaw-puzzle anticlinal walls glazed with rod- and plate-form wax crystals that cast hard shadows across a matte bluish cuticle, making the luminous blister above blaze in contrast against an otherwise muted, textured terrain of translucent walls and epicuticular frost.

You stand inside a living cathedral of water and pressure, a corridor of stacked vacuolar chambers stretching ahead into a pale cerulean infinity, each cell towering roughly two hundred and fifty micrometres from floor to ceiling, its interior almost entirely consumed by a single vast vacuole — a volume of colourless, optically perfect fluid held under such hydraulic tension that the surrounding cellulose walls bow gently outward, glowing a translucent blue-white under transmitted light like frosted sea-glass under a shallow tropical sun. Life itself has been pushed to the absolute margins: the living cytoplasm survives only as a gossamer pale-green film pressed against the interior face of each wall, interrupted occasionally by a small amber-tinted nucleus roughly ten micrometres across, a dense sphere drifting at the edge of an ocean that makes the surrounding vacuolar space feel vertiginous by comparison. This is the root elongation zone in full hydraulic extension, a region where cells are no longer dividing but instead inflating — importing water through aquaporins, building turgor pressure that can exceed several atmospheres, and using that force to drive irreversible cell wall loosening and elongation through the coordinated action of expansins and xyloglucan endotransglycosylases acting on the cellulose microfibril network. Far down the luminous corridor, fifty cell-lengths into the aqueous haze, the central stele appears as a deep indigo axial column — the vascular core, dense with lignifying proto-xylem, absorbing rather than transmitting the light that pours through everything else around it, a structural spine marking the hydraulic highway that will eventually carry this water column upward toward the canopy.

You are standing inside the layered skin of a tree, oriented radially inward, surrounded by rank upon rank of dead cork cells whose suberized walls glow with the warm mahogany-amber of old resin — each one a sealed hexahedral chamber, its lumen a lightless absolute void, the suberin lamellae laid down by a cell that completed its own construction and then vacated entirely, leaving nothing behind but the architecture. These are the cells of the periderm, produced outward by the phellogen, a lateral meristem that generates cork toward the bark surface and phelloderm parenchyma inward, the whole system replacing the epidermis once secondary growth begins and the stem circumference expands beyond what a single outer layer can contain. The cork walls are chemically extraordinary: suberin is a polyester-lipid composite, hydrophobic, nearly impermeable to water and gases, cross-linked into lamellae that resist enzymatic digestion — it is, functionally, biological waterproofing and armor simultaneously, and its amber coloration intensifies with age and impregnation depth, the outermost cells rendered almost sienna in the oldest layers. Ahead of you, the fortress breaks: the phellogen stratum appears as a single translucent sheet of living cells, thin-walled and faintly luminous, the only dividing layer in this entire system, generating dead cork outward and living phelloderm inward in a slow annual rhythm invisible at any scale other than this one. Off to one side, the sealed geometry dissolves into a lenticel — a channel of loosely packed, incompletely suberized complementary cells where the otherwise impermeable periderm admits gas exchange, an exhaling wound in the wall through which the living tissue inside communicates, however faintly, with the atmosphere outside.

You hover suspended in warm, saturated air at the heart of a vast cylindrical corridor — the aerenchyma canal running the length of a water lily petiole, its curved wall an unbroken mosaic of swollen chlorenchyma cells roughly 200 µm across, each one a chlorophyll-dense chamber whose shared cellulose partitions glow faintly amber at the edges and whose inner faces burn a deep, mottled emerald. Aerenchyma tissue like this is not incidental architecture but an evolutionary solution to aquatic life: these gas-filled lacunae form a continuous internal atmosphere connecting leaf to root, allowing oxygen produced by photosynthesis to diffuse downward through flooded sediment where it would otherwise be unavailable. Spanning the full 800-µm breadth of the canal directly ahead, a diaphragm floats like a lace medallion cast in living glass — a stellate array of arm-shaped cells radiating from a central hub, their tapering tips stopping just short of the wall and of one another, leaving open polygonal gaps large enough to drift through, the whole structure back-lit by diffuse light filtering along the canal so that each arm is rimmed with a golden-green halo and the intervening voids frame receding views of the tunnel beyond. These diaphragms are thought to provide structural reinforcement against the collapse of such large air spaces while preserving gas continuity — a compromise between mechanical integrity and atmospheric openness, solved here with extraordinary cellular economy. Thin iridescent films of water tremble across some of the gaps, casting violet and copper interference colors against the green mosaic walls, and in both directions the canal dissolves into a teal-blue atmospheric haze, the cellular texture fading to unresolvable fineness like city lights seen through fog — an immensity contained entirely within the stem of a single leaf.
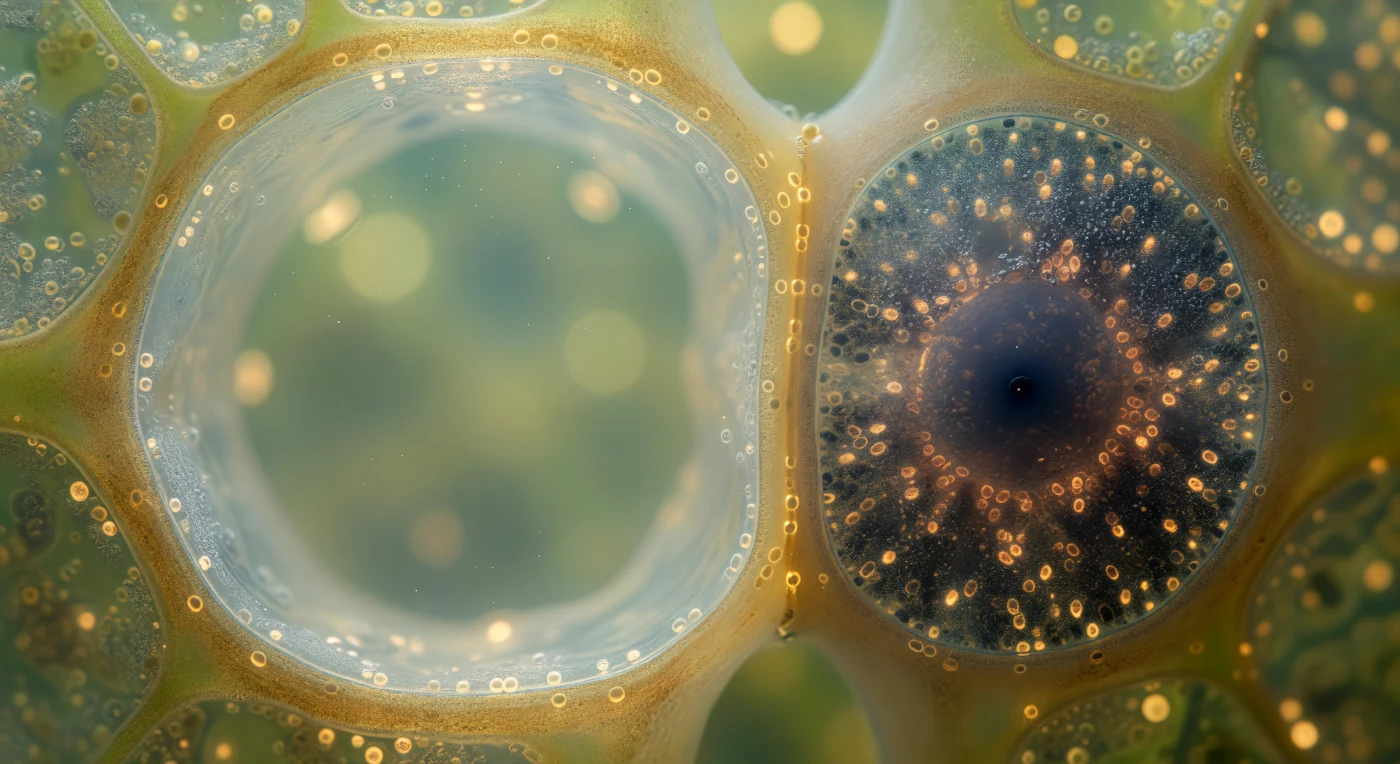
You are suspended at the living interface between two of the most functionally opposed cells in the plant kingdom, where the contrast is not metaphorical but absolute and structural. To your left, the sieve tube element extends like a flooded nave — its lumen stripped of nearly all organelles during maturation, an act of cellular self-sacrifice that leaves a patent, low-resistance conduit through which dissolved sucrose flows at roughly a meter per hour, driven by osmotic pressure gradients established between leaf sources and root sinks in a process described by Münch's pressure-flow hypothesis. Pressed against it to your right, the companion cell has compensated entirely for its neighbor's metabolic nakedness: this single small cell expresses the full transcriptional machinery, drives active sucrose loading through symplastic plasmodesmata and apoplastic H⁺-ATPase-coupled transporters, and sustains the sieve tube energetically through the very nanoscale channels — those faint amber perforations visible at the shared wall — that stitch the two cells into a functional unit despite their radical cellular asymmetry. The intensity of that dark, ribosome-dense, mitochondria-crowded companion cytoplasm is not incidental but obligatory: it is running the metabolism of two cells in the volume of one, a biological forge maintaining the slow sweet river next door.

You are suspended at the exact threshold where the living root enforces its most absolute law: nothing passes this border unchecked. Looking along the curve of the endodermal cell file, you see prismatic chambers pressing close on every side, their translucent walls glowing faintly greenish-ivory, and running continuously through each shared radial wall — cell to cell without interruption — a belt of amber-orange fire, the Casparian strip, composed of suberin and lignin deposited with geometric precision into the wall midline, forming an unbroken luminous necklace that encircles the entire vascular cylinder. Behind you the cortex opens into a looser, cooler architecture of pale parenchyma with generous intercellular air spaces, while ahead the stele compresses into a dense indigo and violet mass of procambial cells, their nuclei prominent, the whole structure reading as a jewel of biological intensity — the chromatic contrast between the warm amber seal and the cold blue interior making the functional boundary visible as pure sensation. This strip is not decorative: it forces all water and dissolved ions moving from soil into the vascular system to pass through the plasma membrane of the endodermal cell itself, bypassing the leaky apoplastic route through the cell walls, so that the plant exercises molecular-level selectivity over every ion that enters its circulation — a living checkpoint encoded in the chemistry of a single, precisely positioned band of wall material no more than a few hundred nanometers thick.

You stand at ground level on a world constructed entirely of interlocking amber walls, their material sporopollenin — one of the most chemically resistant biopolymers known to biology, capable of surviving millions of years in sediment without degradation — shaped here into a continuous reticulate lattice of ridges enclosing irregular polygonal basins each a few micrometers across, the whole architecture pressed outward from the surface of a single rose pollen grain no wider than a human red blood cell is long. Each raised murus curves with a geological honesty, its inner and outer slopes shadowed unevenly by raking illumination that autofluoresces gold from within the polymer itself, pooling warm ochre in the lumina and brightening each crest to pale honey. Running from horizon to horizon across the mid-distance, a colpus groove interrupts the mesh — a germinal aperture where the exine thins toward translucency, structurally predestined as the site where the pollen tube will eventually breach the wall to deliver its cargo — its floor smooth and reflective against the textured geometry surrounding it on all sides. The reticulate exine sculpturing visible here is not decorative but taxon-specific, its pattern determined during microsporogenesis by the geometry of the callose-walled tetrad in which the grain matured, each ridge crystallized under competing pressures into this imperfect, irreproducible equilibrium. Clarity is absolute at this scale: no moisture, no atmospheric scatter, only the sharp geometry of a fossilized architecture that evolved to protect a living cell across distances measured in meters while its internal machinery operates in nanometers.
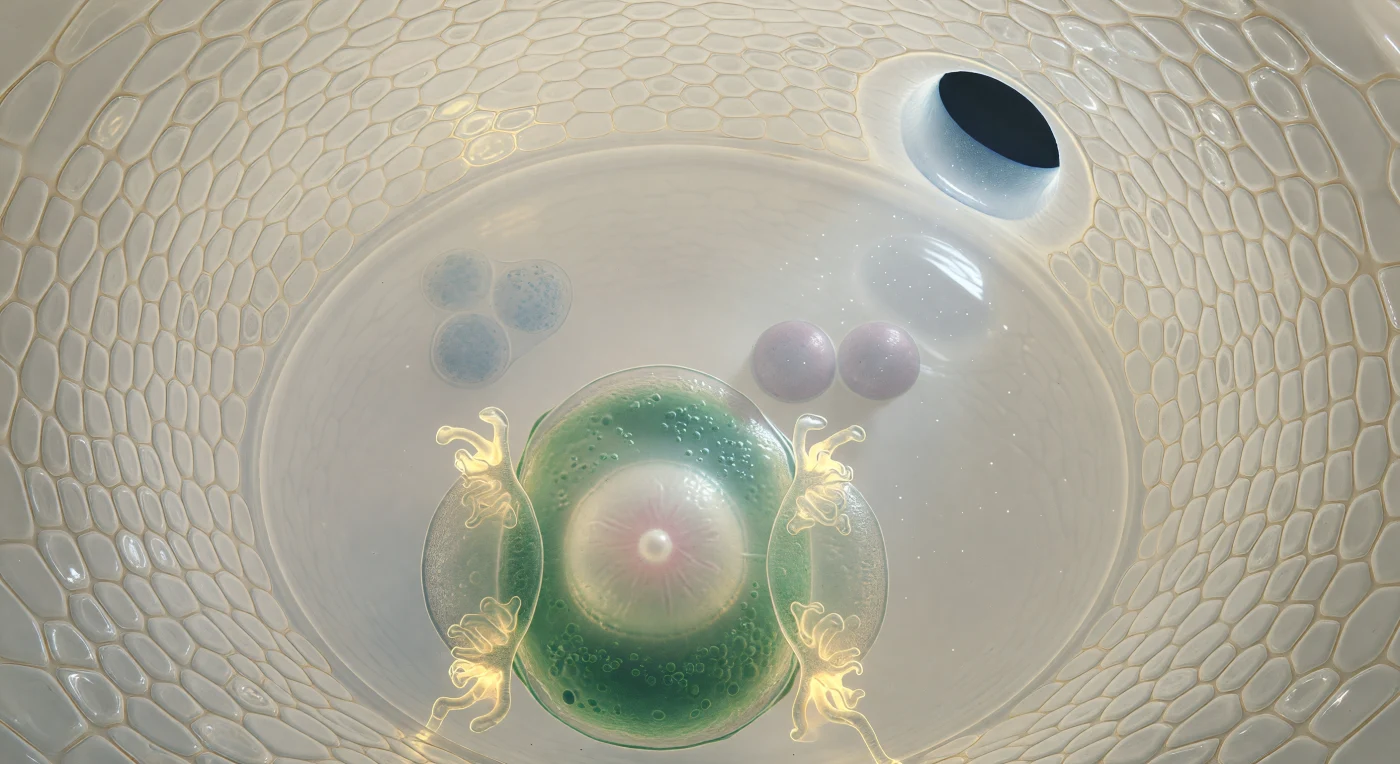
You are suspended within the embryo sac of a flowering plant's ovule, drifting perhaps forty micrometres from the egg cell's pale convex surface — a chamber so optically pure it reads less as a cell than as a room filled with water. The curved integument layers arch around you like the walls of an ivory grotto, their tightly pressed cells seamed with amber, the whole structure suffused with a warm translucency that admits a single soft shaft of light filtering through the micropyle at upper right — a passage barely fifteen micrometres across, the narrowest gateway through which a pollen tube will one day force its way to deliver the male gametes that trigger double fertilization. In the foreground the egg cell bulges close, jade-green and organelle-dense at its periphery, cradling a nucleus like alabaster veined with rose, flanked by the two synergid cells whose filiform wall ingrowths glow amber-gold at their tips — specialized membrane folds whose enormous surface area is thought to facilitate the uptake and guidance of the arriving pollen tube, charged with signaling molecules that the synergids must intercept and relay. Behind the egg, the vast central cell opens into translucent stillness, and near its center two lilac-grey polar nuclei float like paired moons awaiting the second sperm cell that will fuse with them to form the triploid endosperm — the nutritive tissue destined to feed an embryo not yet conceived — while far at the chalazal pole three antipodal cells dissolve into dim granular shadow, their function in most flowering plants still incompletely understood, the furthest reaches of a living architecture poised at the threshold of an entirely new organism.

You are suspended at the threshold between two utterly different architectures — to your left, enormous vessel lumens gape open like the mouths of sea caves, each hollow nearly 250 micrometers across, their bordered-pit walls catching polarized light as faint amber rims etched into lignified tawny walls; to your right, the latewood closes without gradation into near-solid matter, thick-walled libriform fibers packed so densely that only slit-like apertures 15 micrometers wide survive, their cellulosic secondary walls exploding under polarized illumination into electric cobalt and burnished gold — the optical signature of helically wound crystalline microfibrils storing mechanical energy in compression. This abrupt boundary marks the annual rhythm of the vascular cambium: earlywood formed in spring under hormonal signals favoring wide, low-resistance conduits for rapid water flux, latewood assembled in summer as auxin levels fall and cell expansion is curtailed, each regime written permanently into the wood as a record of seasonal physiology. Threading horizontally across the entire boundary, ribbons of ray parenchyma — pale honey-gold, their brick-shaped cells two to four cells wide — run in continuous radial bands that ignore the earlywood-latewood divide, carrying sugars and signals laterally through a tissue otherwise dedicated to vertical transport. The scene reads simultaneously as living geology and stained-glass architecture: crystalline, monumental, and encoded with decades of atmospheric memory pressed into amber and iron.
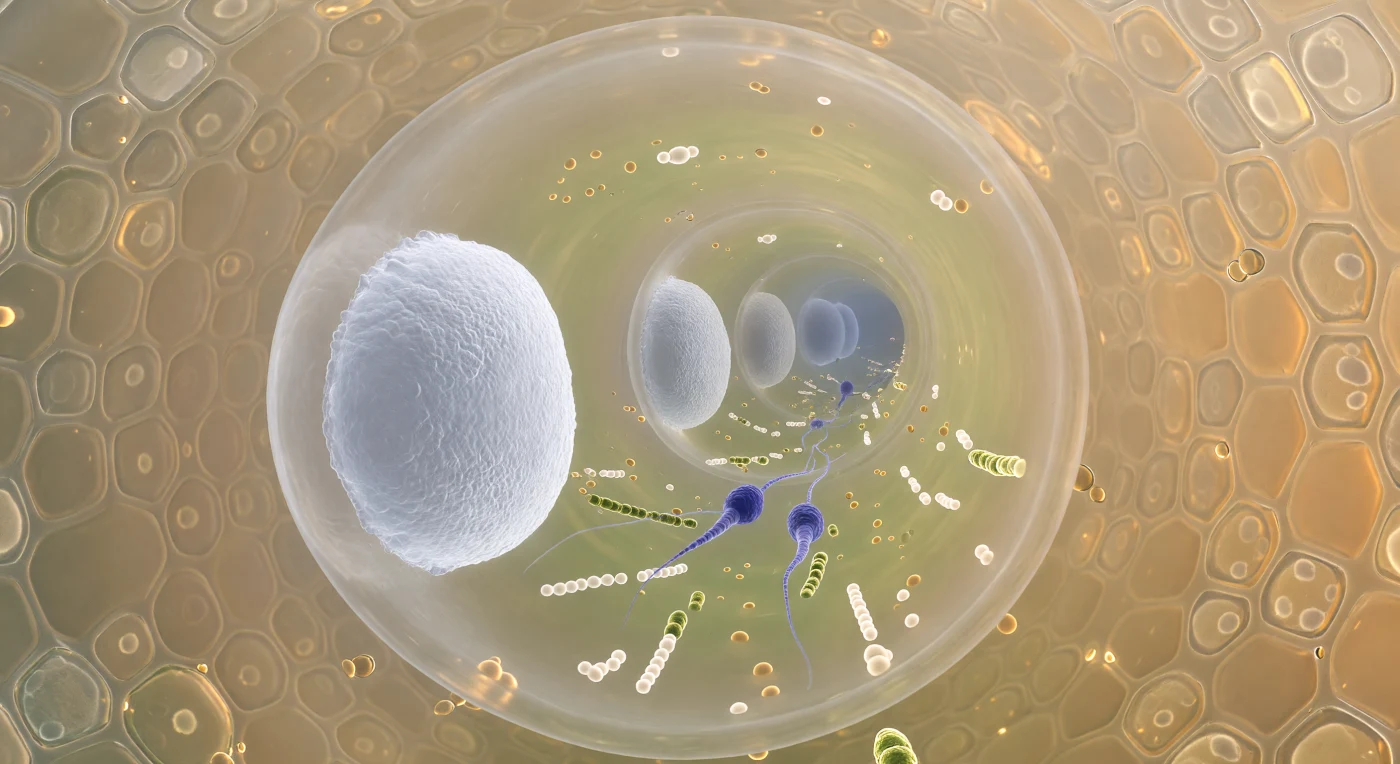
The view plunges forward through a living corridor barely twelve micrometres wide, its curved amber-tinted wall enclosing a world compressed beyond ordinary intuition — ahead, the nearest callose bulkhead rises like a frosted porthole of compacted polysaccharide gel, its surface resolving into braided microfibrils that glow a cool porcelain white, while beyond it the next plug hangs as a dimmer disc, and beyond that another, the whole colonnade receding toward the invisible growing tip in diminishing rings of pale light. Between the nearest two septa, the cytoplasm is dense with traffic: lipid droplets, mitochondria, and chains of pale vesicles crowd an axial river of streaming protoplasm, and at the centre of this flow two sperm cells — fusiform, indigo-violet, their chromatin condensed to near-opacity — ride forward without effort, trailing wisps of endoplasmic reticulum like silk ribbons caught in a current. The callose plugs themselves are not passive architecture but active seals, deposited in rhythmic succession as the tube elongates, each one isolating the older compartments behind it while the living machinery of fertilisation presses on ahead. Beyond the tube wall, the stylar transmitting tissue crowds close as a mosaic of pale gold secretory cells embedded in an adhesive polysaccharide matrix, their refractive secretions diffusing a warm amber glow through the wall and transforming this microscopic passage into something that feels simultaneously intimate and purposeful — a pressurised corridor navigated entirely by chemistry, turgor, and the directional urgency of reproduction.