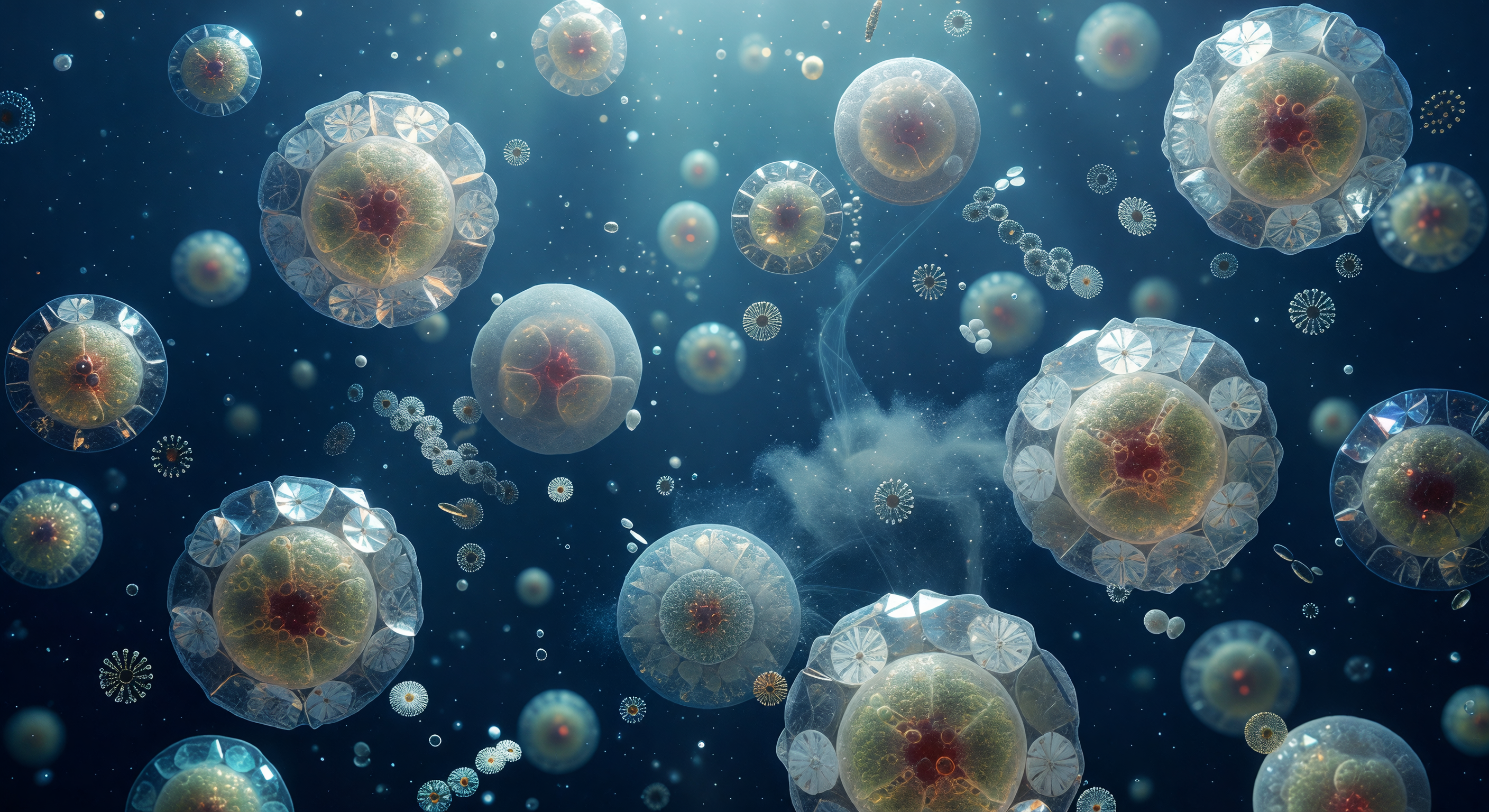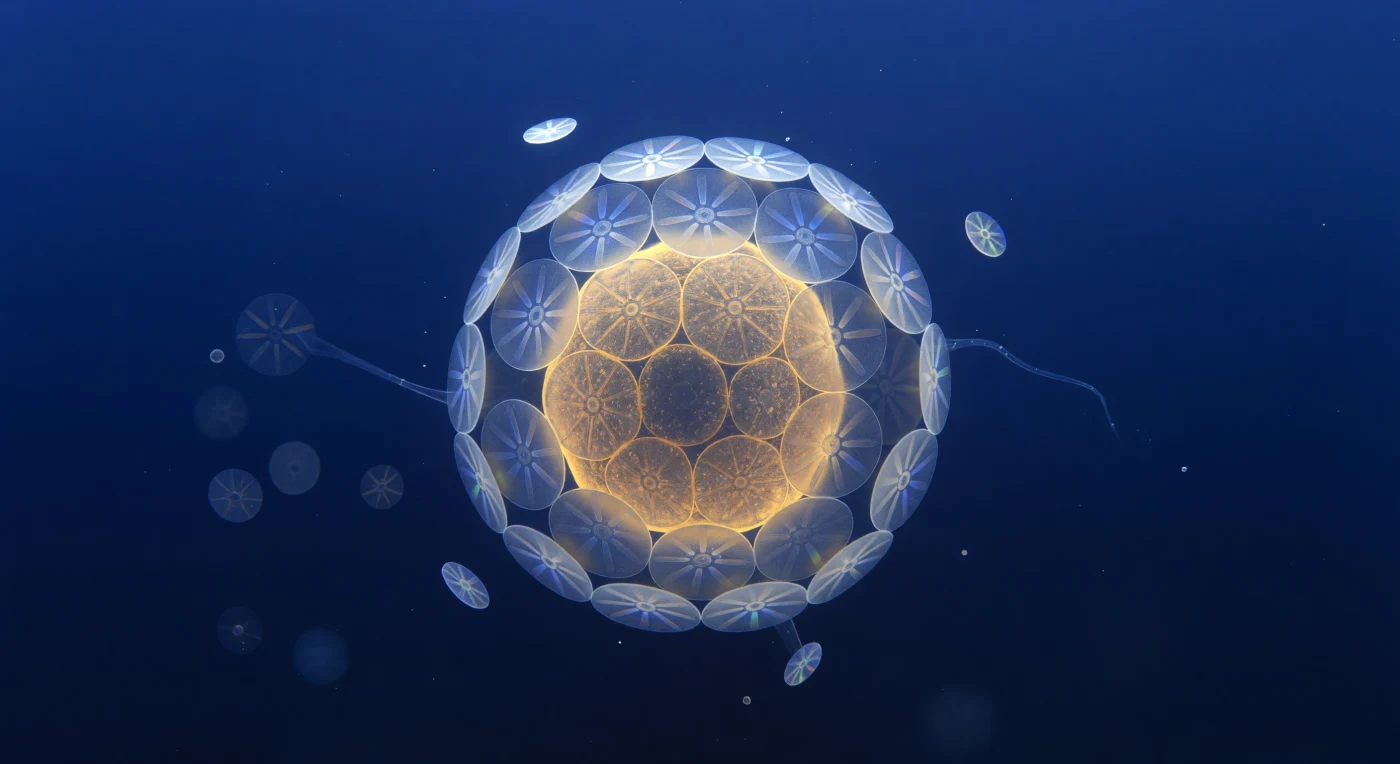
Suspended at the threshold of visibility, you face an eight-micron sphere of amber-gold radiance that fills your entire field of view like a miniature sun — the single-celled alga *Emiliania huxleyi*, its interior chloroplasts burning warm saffron through a translucent membrane, encased in twenty interlocking calcite cartwheel plates whose crystal spokes fracture the faint downwelling blue light into fleeting prismatic shivers of violet and mint before snuffing them out again. Each coccolith is a precision-engineered disc of biogenic calcium carbonate, its proximal and distal shields stacked in shallow relief, the whole coccosphere assembled by the cell itself through a choreography of intracellular calcification and exocytosis that deposits one plate at a time into the armor. Around you, the ultramarine water column is not empty but crowded with near-invisible architecture — ghost coccospheres receding into indigo fog, free-drifting detached plates tumbling in Brownian thermal noise, a filament of transparent exopolymer stretched diagonally across the mid-field like spun glass — all of it bathed in directionless volumetric light that carries no hard shadow, only the soft compression of photons that have already traveled meters of open ocean to reach this quiet, impossible scene. What you are witnessing is simultaneously structural engineering and geochemistry in miniature: these calcite shells, when the cell dies, will sink through thousands of meters of water column and, over geological time, accumulate on the seafloor as the compressed chalk that now forms the white cliffs of Dover.
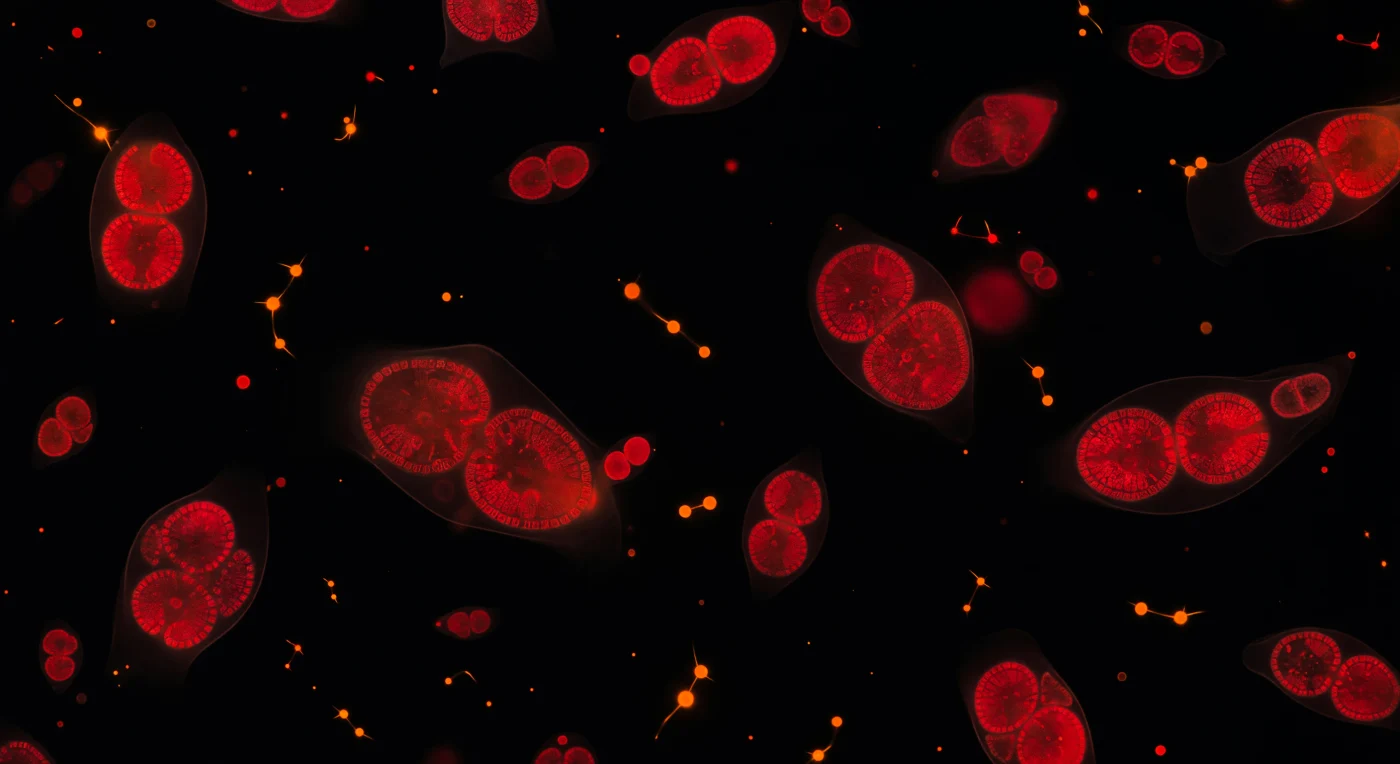
Suspended in absolute darkness, you find yourself adrift in a world measured in millionths of a meter, surrounded by dozens of phytoplankton cells burning as deep crimson embers — their chlorophyll molecules fluorescing at 680 nm, a wavelength they normally harvest to drive photosynthesis, here radiating outward as self-generated scarlet light with no external source to supplement them. Inside the nearest cells, double-lobed chloroplasts resolve as paired kidney forms dense with stacked thylakoid membranes, the molecular machinery of oxygenic photosynthesis compressed into structures just a micron or two across, their pigment concentration so high that the innermost folds deepen toward burgundy. Scattered among the crimson masses, cyanobacteria punctuate the void as sharp tangerine-orange sparks — prokaryotic cells carrying phycoerythrin rather than chlorophyll, evolutionarily ancient organisms whose ancestors first oxygenated Earth's atmosphere some 2.7 billion years ago, now burning a visibly different color in the same dark medium. The fluid itself is invisible yet present, its viscosity absolute at this scale — inertia here is essentially meaningless, Brownian thermal drift the only motion — and faint aureoles of scattered light around the nearest cells confirm you are suspended in ocean water rather than vacuum, a single microliter of sea surface that, multiplied across hemispheres, constitutes the base of nearly all marine food webs and produces roughly half of Earth's atmospheric oxygen.

You stand chest-deep in water that is no longer quite water: the North Atlantic at the height of an *Emiliania huxleyi* bloom has become a living mineral suspension, opaque as diluted chalk, its jade-white and turquoise-cream surface stretching to the horizon in every direction under a noon sun that seems to burn upward from below as much as down from above. At ten million calcite-plated cells per milliliter, each microscopic organism armored in interlocking geometric wheels of crystalline calcite, light cannot penetrate more than a hand's depth before being scattered back skyward, transforming the entire sea surface into a diffuse, self-luminous mirror that reflects over forty percent of incoming solar radiation. Langmuir windrows rake the surface in long parallel ribbons of condensed-milk cream, each streak the visible fingerprint of paired counter-rotating vortices a few meters deep, concentrating buoyant coccoliths into lines spaced like crop rows across an impossible field. At the horizon, the bloom boundary draws an almost surgical line between chalk-white opacity and the sudden extraordinary darkness of open cobalt ocean, a boundary that from orbit would appear as a distinct edge across hundreds of kilometers of North Atlantic surface, marking the outer frontier of what is effectively a biogenic mineral weather system lying flat on the sea. Glancing down through the luminous white-green curtain swallowing your own submerged torso, you register the faint sulfurous sweetness of dimethyl sulfide rising where senescent cells lyse at the bloom's edge — a chemical signal detectable by seabirds from kilometers away, a smell that means life, and death, and the slow transfer of carbon downward into the deep.
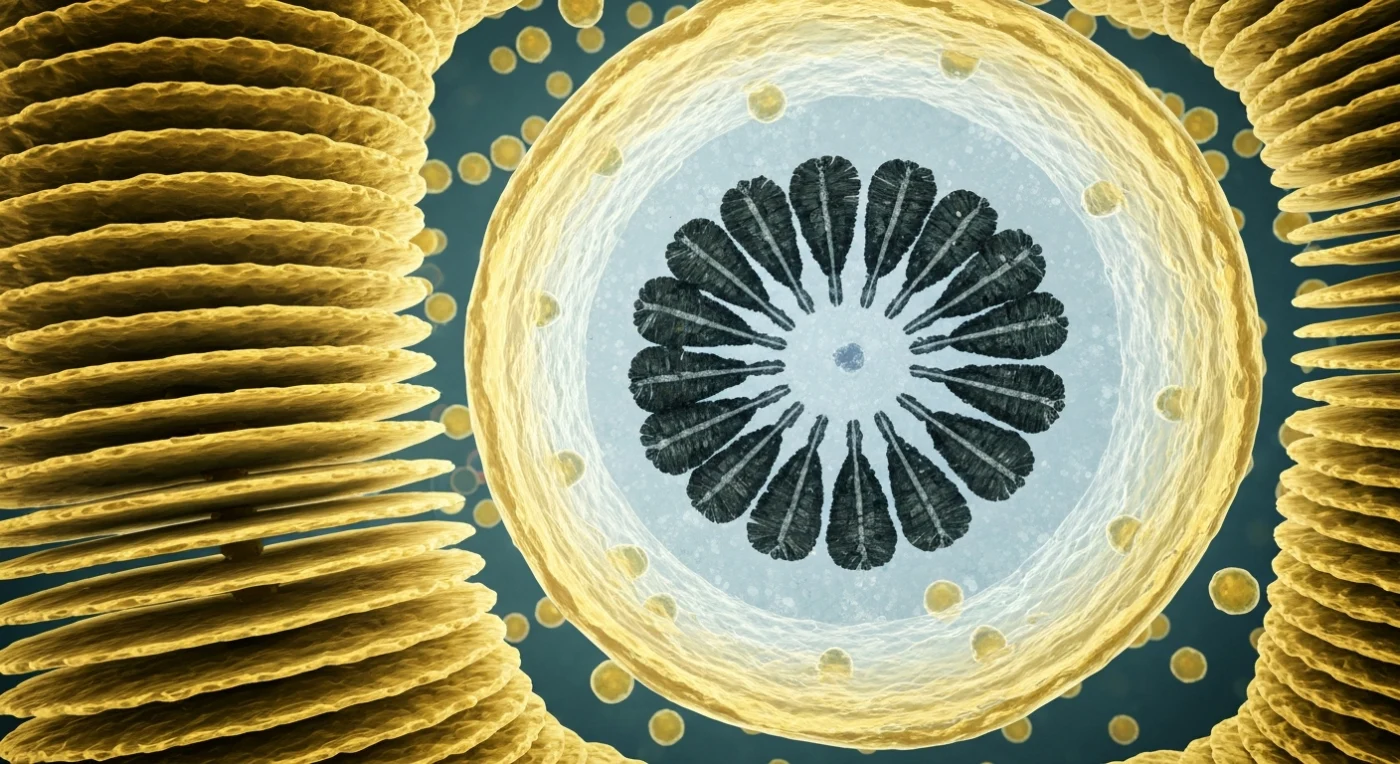
You are standing inside a living cell, surrounded by stacked sheets of membrane that rise around you like tiers of hammered amber glass — the Golgi cisternae, each bilayer only a few tens of nanometers thick, curving at their rims into swollen vesicular buds that pinch free and drift through the cytoplasm like warm lanterns, each one ferrying calcium and carbonate precursors toward the great assembly chamber ahead. Every surface in this crowded interior is studded with ribosomes, tiny dark granules arrayed in dense constellations across the pale gray-green skin of the endoplasmic reticulum, giving each membrane a velvety roughness as though the organelles themselves are breathing. Dominating the view like an industrial dome, the coccolith vesicle swells — its translucent amber wall enclosing a dim aqueous interior suffused with cold blue-gray light, where a nascent calcite ring assembles against a ghostly polysaccharide base plate, thirty-odd dark charcoal prisms interlocking in a radial wheel with a precision that feels engineered, one quadrant still open, its terminal crystals jagged where mineralization is actively proceeding, calcium ions arriving in invisible pulses through the membrane. This is *Emiliania huxleyi* building its armor from seawater chemistry alone — a molecular construction project whose product, one calcite plate just a few micrometers across, will eventually drift to the seafloor and, across geological time, become chalk.

Before you, suspended in a void of absolute black, a coccosphere blazes like a captured star — fifteen interlocking calcite wheels arranged into a near-perfect sphere, each coccolith erupting with birefringent fire under cross-polarized light, the classic Maltese cross extinction pattern sweeping across every plate in velvety black while the surrounding crystal domains burn in first-order white of almost physical density. At the thickened spoke rims and raised distal shield edges, thin-film interference lifts the palette fractionally into pale champagne and warm ivory-gold, the only concession to color in an otherwise achromatic spectacle. Drifting outward in every direction, shed coccoliths tumble in imperceptible Brownian drift — each an elliptical calcite wheel just a few micrometers across, architecturally ornate, some edge-on as luminous slivers, others face-on to reveal the full crystallographic symmetry of their spoke geometry, their open central area framing what reads as a window into a microscopic cathedral. These plates are not passive shells but biologically precipitated calcite, grown one calcium carbonate ion at a time inside specialized vesicles within the living cell, the crystal orientation of each radial unit genetically controlled to produce precisely this optical signature. You float among them with no body and no instrument — only attention — adrift inside a chandelier assembled by a single-celled organism in cold, dark water, its geometry encoding both a life strategy and, grain by grain across geological time, the raw material of chalk cliffs.
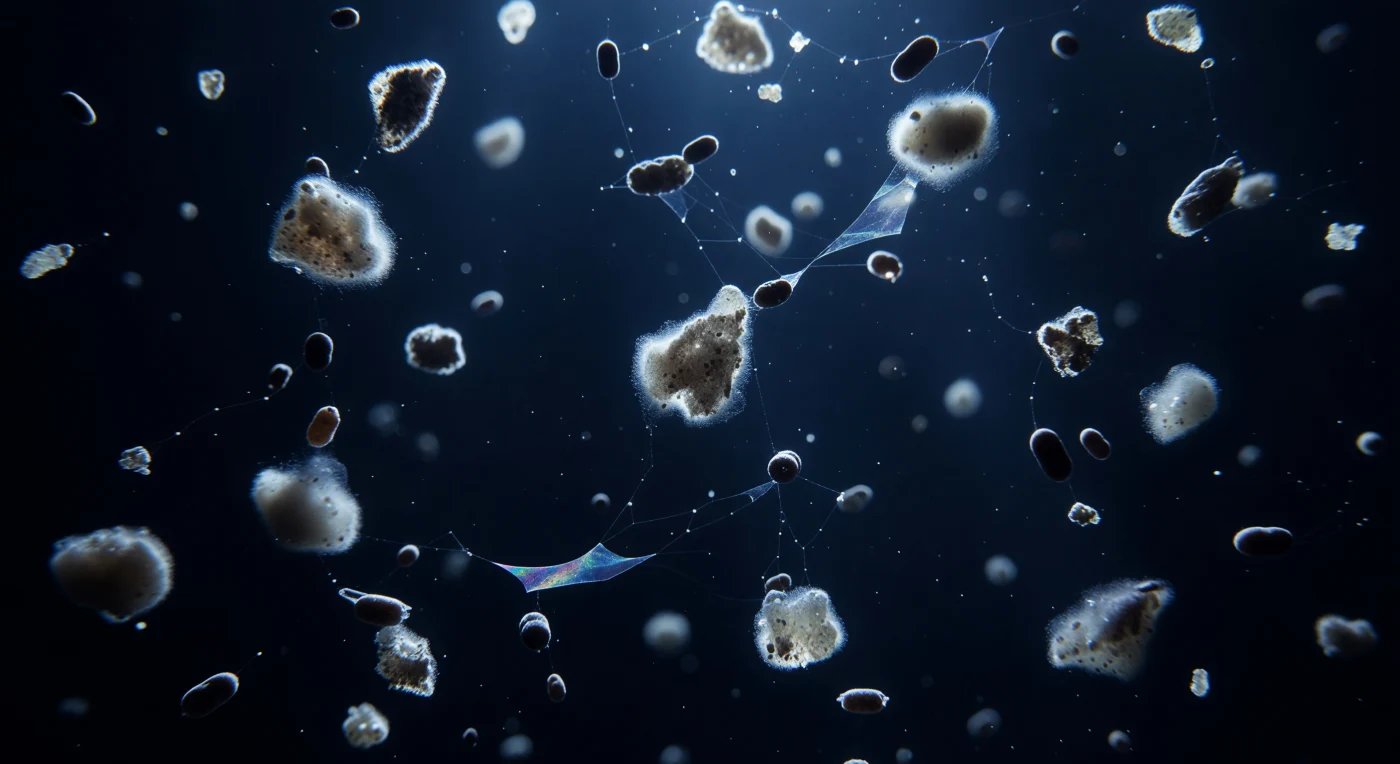
You are suspended in the mesopelagic twilight at 200 meters depth, immersed in an indigo-black water column where the last attenuated photons from the sunlit zone above barely register — and yet they are enough to illuminate a slow, ceaseless blizzard descending around you. Marine snow aggregates, each a loose confederation of coccolithophore armor plates, diatom frustules, mucus strands, and digested cellular debris compacted over days of aggregation, drift downward as cottony gray-white masses ranging from a few millimeters to the size of a small plum, their embedded calcite fragments catching residual blue-white light at 450–490 nanometers and igniting into a faint, ghostly halo at each floc's margins. Between them, compact dark-brown fecal pellets — dense cylinders ejected by copepod grazers in the waters above — fall three to ten times faster, punching silently through the aggregate field, while nearly invisible sheets of transparent exopolymer particles (TEP), secreted by phytoplankton as overflow products of carbon fixation, trembles in gossamer webs between the flocs, catching the sparse light only at their thinnest edges. This is the biological pump made visceral: each descending aggregate is a carbon archive, a compressed record of photosynthesis, grazing, death, and mineral ballasting, obeying gravity in a procession so slow it reads as geological — yet in aggregate, across the world ocean, this blizzard transfers hundreds of millions of tonnes of organic carbon to the deep seafloor every year, regulating atmospheric CO₂ on timescales that dwarf human civilization.
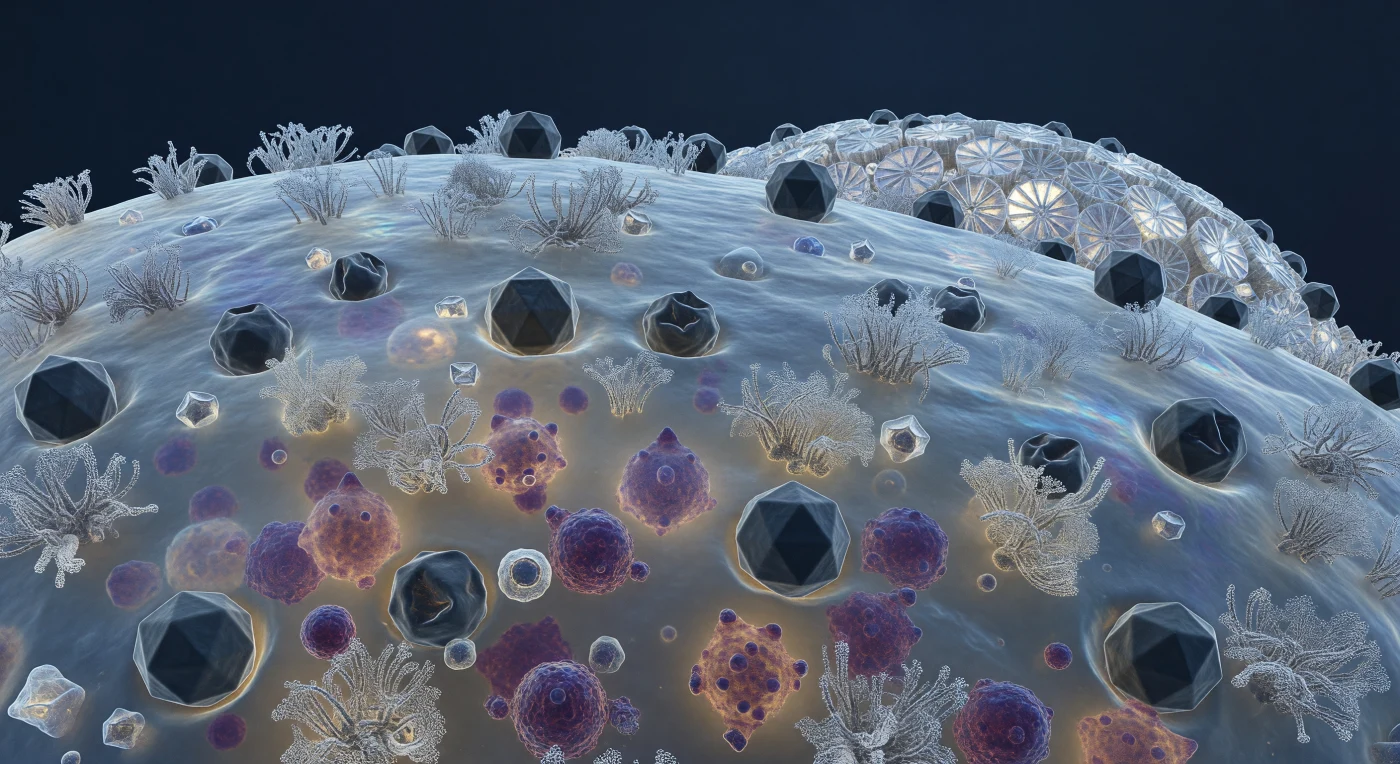
The viewer hangs motionless above a gently curving biological horizon, close enough that the coccolithophore's outer membrane resolves into a trembling, semi-translucent skin — a landscape of glycoprotein clusters rising like coral formations from a blue-gray surface shimmering with thin-film interference where lipid raft domains concentrate. Pressed into this terrain at dozens of sites, dark icosahedral capsids of *Emiliania huxleyi* virus sit in various states of predatory engagement: some geometrically pristine, their twenty facets catching cold diffuse light along sharp ridges, others visibly collapsed inward — capsid shells crumpled like punctured geodesic domes after successful genome injection, the membrane dimpled beneath each one in a shallow hollow of biological surrender. Through the translucent membrane wall, the cytoplasm glows like fogged amber glass, crowded with dense irregular masses of deep purple and magenta — viral progeny assembling by the hundreds on nucleoprotein scaffolds in a factory-scale replication event that presses outward against the cell wall from below, bulging the membrane like overfilled sacking. Along the cell's sweeping curvature toward the distant coccosphere horizon, interlocking calcite coccolith plates rise as white ceramic buttresses, their spoke-and-rim geometry scattering what little deep-water light reaches them into brief prismatic flashes — a world of extraordinary crystalline precision and molecular beauty quietly unraveling, every contested surface a boundary between the cell's remaining chemistry and the viral program steadily overwriting it.

You hover in a world that is neither dark nor lit in any ordinary sense — the water around you glows a precise, almost architectural cobalt-blue, the residual 475-nanometer light that survives a hundred meters of ocean column arriving as a diffuse luminance that seems to originate everywhere and nowhere at once. This is the deep chlorophyll maximum, a layer where photosynthetic cells accumulate at the exact depth where light energy and nutrient supply reach their most productive compromise, and at the scale of a single dinoflagellate cell the community around you resolves into dozens of warm olive-gold organisms drifting in loose constellation — their chloroplasts burning amber against the cold indigo, their silica-walled diatom neighbors extending in spiral chains that catch stray photons and scatter them like compressed embers. A marine snow aggregate the size of a building in your reference frame descends past you in absolute silence, a translucent architecture of exopolymer threads, interlocked coccolith plates, and half-living cells wrapped in mucus, its thinnest membranes backlit to faint luminosity while its denser core remains nearly opaque — the biological pump made visible, carbon and calcium packaged for the long fall into the aphotic zone below. Farther down, the olive warmth of living cells grows sparse and then absent, the gradient between the chlorophyll maximum and true darkness unfolding not as a boundary but as a slow extinction of color, until only the cold blue remains, pierced occasionally by the white sparks of distant marine snow catching the last available photons on their spiral descent toward the sediment thousands of meters below.

You are suspended in the uppermost millimeter of the open ocean in the last minutes before sunrise, floating beside a structure that dwarfs you the way a city bus dwarfs a pedestrian: a single *Ceratium tripos* dinoflagellate, its amber-gold body enclosed in tessellating amphiesma plates whose seams catch the first photons filtering down through the air-water interface above as hairlines of cold blue fire. The cell's three hollow horns — one apical, two antapical — are not solid spears but transparent calcite lances braced internally by trabecular lacework, thin enough that the deepening predawn light passes through them and illuminates the brownish-gold chloroplast clusters packed within the main cell body, strobing them rose and amber as micro-capillary ripples skate the surface and bend the incoming light into slow prismatic sweeps. At the equator of the cell, seated in the cingulum groove like a thread running through a carved canyon, the transverse flagellum beats in continuous helical strokes that blur into a silver shimmer — the primary engine of this organism's diel vertical migration, which carries it toward the surface each dawn to harvest photons before descending again at dusk, a daily commute of meters that, at this scale, feels like the movement of weather systems. Beyond the Ceratium, three to four centimeters away in real space but perceived here as the distance to a glass office tower, a copepod hangs motionless in the water column, its paired antennae spread like enormous translucent oars, its segmented legs individually larger than the entire dinoflagellate before you — a reminder that this luminous microscopic cathedral exists inside a food web whose predators are, by the logic of scale, geological in their mass.

You are suspended inside what appears to be a living medium, a jade-green suspension so densely packed with *Ostreococcus tauri* cells that the fluid itself has taken on color, each cell a near-perfect sphere barely larger than the wavelength of visible light, trembling constantly on Brownian thermal currents with no surface, no floor, no orientation to anchor you. These are among the smallest known eukaryotes on Earth — 0.8 micrometers across, yet fully equipped with a nucleus, a single chloroplast that nearly fills the entire cell body, and a lipid membrane just 7 nanometers thick that nonetheless catches light as a discrete optical surface, producing a faint iridescent halo before rotating away into the fog. The suspension is so optically dense that photons scatter before they can travel the length of a few cells, meaning depth dissolves into color saturation rather than perspective — nearby cells resolve as distinct viridian spheres with dark chloroplast inclusions, while those two or three micrometers distant blur into the collective jade luminescence, their individuality surrendered to the aggregate. In ocean surface waters, blooms of picophytoplankton like *Ostreococcus* contribute disproportionately to global primary production precisely because their extraordinary surface-area-to-volume ratio makes them ruthlessly efficient at harvesting diffuse photons and assimilating dissolved nutrients in oligotrophic waters where larger cells starve. Here, immersed in their billions, the boundary between organism and medium has effectively ceased to exist — the water is the life, and the life is the water.
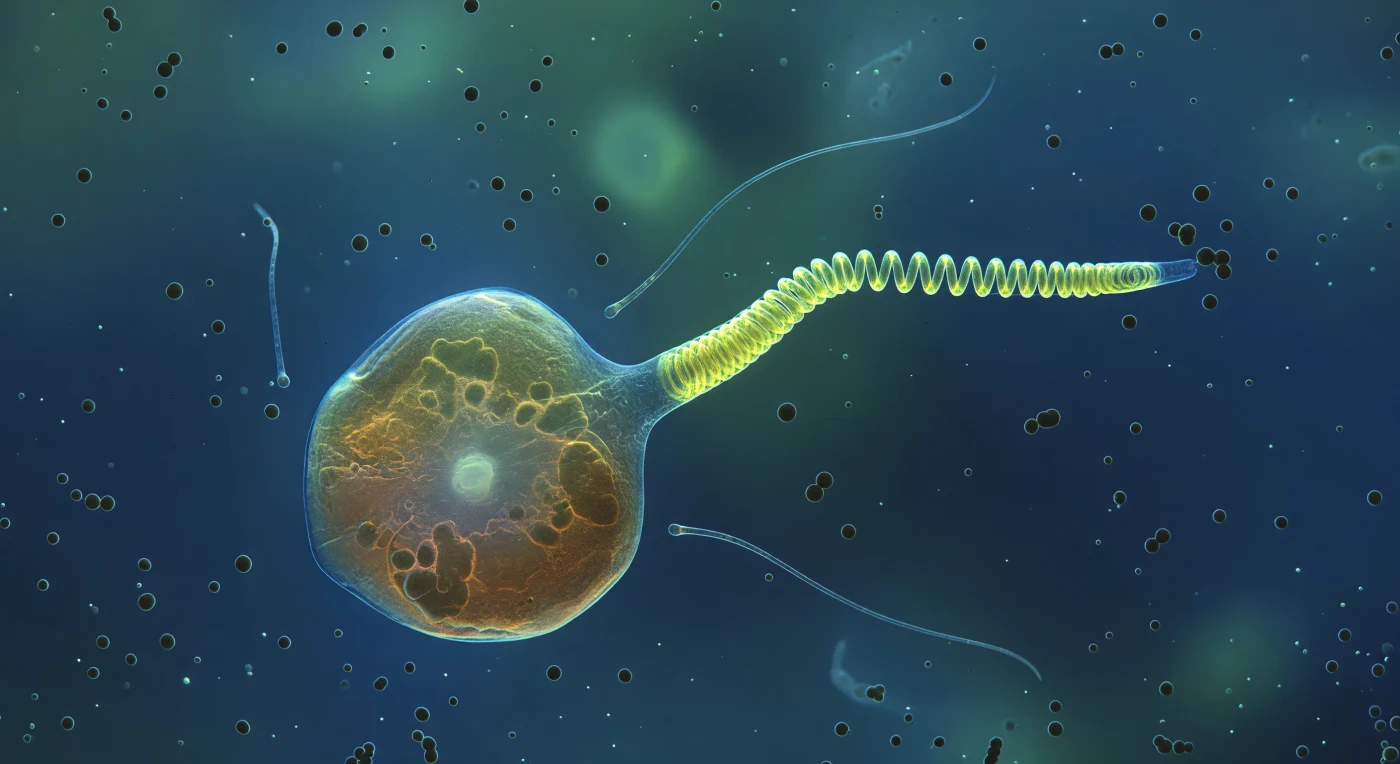
You are suspended inside a moment of microscopic violence, watching a single *Chrysochromulina* cell — roughly the size of a small house at this magnification — hold the center of a cold blue-green world as its haptonema tears itself between two geometries. The proximal coil has already snapped into a tight fluorescent helix, dense and spring-loaded, while the distal portion still trails as a stiffening rod caught mid-buckle at the transition zone, the entire structure blazing yellow-green against the deep sapphire medium like a neon filament under tension. The haptonema is not a flagellum but a separate organelle entirely — a microtubule-reinforced appendage unique to haptophyte algae, capable of recoiling in milliseconds through a calcium-triggered conformational cascade, likely used to capture bacterial prey or anchor the cell to surfaces. Dark charcoal spheres — bacteria roughly a tenth the cell's diameter — cluster near the thrashing tip, drawn into the chemical gradient radiating from the larger organism, while two ghostly flagella drift loosely to either side of the amber-translucent cell body, utterly indifferent to the snap occurring beside them. The surrounding water carries its own quiet density: particulate scatter, membrane vesicles, dissolved organics lending a faint milky luminosity to the background before it dissolves into oceanic black, the distance communicating itself not through any visible horizon but through the way the bacterial spheres simply fade and vanish into the ambient blue before the eye finds an edge.

Standing at the bow rail, you look almost straight down at one of the sharpest natural boundaries visible anywhere on Earth's surface: to port, an abyss of indigo-cobalt water so transparent that afternoon light plunges meters into it and returns nothing but depth; to starboard, a body of water that reads as something between liquid and mineral — chalky, opaque, turquoise-white, emanating a diffuse pearl-bright luminescence that seems to originate from within the water column rather than from its surface. That glow comes from tens of billions of individual *Emiliania huxleyi* cells, each no larger than a human red blood cell, armored in interlocking calcite plates just two to four microns across that tumble freely and scatter incoming photons in every direction simultaneously, turning the upper water column into a living diffusion lens. The boundary between the two water masses — one ancient and empty of this white alchemy, one dense with microscopic geometric armor — is almost surgical, perhaps two meters wide, organized along its length by Langmuir circulation into parallel foam windrows that mark where the two regimes shear against each other in slow surface eddies; a gannet punches through exactly at that line and erupts a geyser of milk-white water that catches the low-angled sun before the surface closes and the bird vanishes into the bloom. The air carries the faint sulfurous sweetness of dimethylsulfide off-gassing from stressed cells, a scent that connects what the eye sees — a pastel planetary bruise stretching to the horizon — to chemistry happening inside organisms invisible to the naked eye and to the cloud-seeding, climate-altering consequences that follow from their collective death.

You are suspended within a compressed continent of the dead, surrounded in every direction by interlocking calcite wheels — the skeletal remains of coccolithophores that once photosynthesized in sunlit surface waters tens of millions of years above you in time. Each disc measures just a few microns across, yet its geometry is obsessively precise: radial crystal spokes fused into a raised rim, the trigonal calcite units still interlocked with structural integrity despite the crushing weight of millennia of overburden, their surfaces catching a raking amber sidelight that throws micro-shadows across every raised edge and transforms the dense matrix into a landscape of miniature craters and pale archways. This is coccolith ooze — the foundational material of chalk — where species of *Emiliania*, *Gephyrocapsa*, and *Calcidiscus* are identifiable by their spoke count and rim architecture alone, their biological identity reduced entirely to crystal symmetry. Looking along the laminar horizon, alternating bands of tightly-packed assemblages and clay-intruded glacial intervals curve across the field of view like compressed geological strata, each lamina only tens of microns thick yet representing centuries of slow carbonate rain from the photic zone above, the biological pump made lithological. There is no motion here, no chemical gradient, no Brownian jostling — only the afterlife of an ancient bloom, its geometry persisting long after every lipid, chloroplast, and membrane has dissolved into pore fluid, held in sharp relief by a sidelight that refuses to soften.
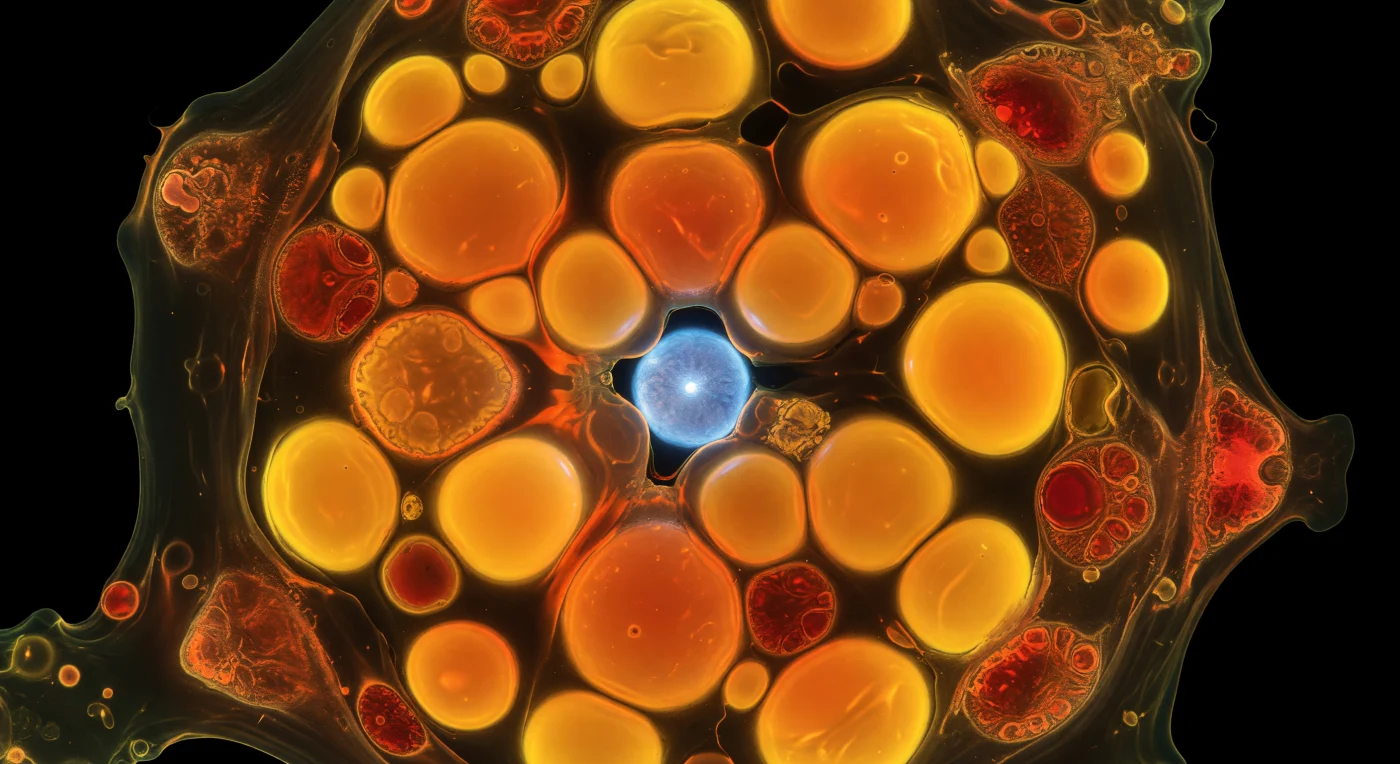
You stand inside a single *Emiliania huxleyi* cell under nitrogen starvation, surrounded on all sides by a press of enormous lipid droplets that glow with an almost violent orange-gold fluorescence — Nile Red molecules intercalated into neutral lipids and emitting light from within, so that each spherical droplet burns like an amber lantern in absolute cellular darkness. These droplets, ranging from boulders to small moons at this vantage point, crowd the cytoplasm so completely that they deform against one another at contact points into flattened lenses, throwing overlapping halos of saffron and deep amber through the viscous cytosol threading between them. This is the cell's metabolic crisis made visible: when nitrogen is withheld, *E. huxleyi* diverts carbon away from protein synthesis and chloroplast maintenance and into triacylglycerol accumulation — lipid droplets swelling until they dominate the entire cytoplasmic volume, a last-resort energy reservoir that can constitute 30–50% of cell dry weight under severe stress. At the periphery, chloroplasts have been displaced and compressed by the advancing lipid mass, their chlorophyll autofluorescence reading as a dim, cooling crimson against the orange blaze — thylakoid membranes still faintly laminated but photosynthetically throttled. Somewhere deeper, glimpsed through a gap between blazing droplets, the DAPI-stained nucleus glows a cold cerulean blue, its nuclear envelope smooth and taut against the surrounding warmth — a quiet genomic archive in the center of a cell that has reorganized nearly everything else around the imperative of storing carbon.

You are standing chest-deep in a sea that has stopped being a sea and become instead a field of dark potential, every movement of your body rewriting the darkness in cold electric blue. The light is not ambient but generated — biochemical, involuntary, erupting from *Lingulodinium polyedra* cells each measuring roughly 40 micrometers across, single-celled organisms whose scintillons, membrane-bound organelles packed with luciferase enzyme and its luciferin substrate, discharge in millisecond cascades wherever mechanical pressure disturbs the water, converting chemical energy directly into photons at precisely 460 nanometers with no heat, no combustion, only oxidation at the molecular scale made suddenly, staggeringly visible. What appears as a continuous river of light in the boat wake 50 meters distant is in truth the superimposed discharge of billions of individual cellular firing events, each organism responding to shear stress as it has for hundreds of millions of years — a defense mechanism or a startle response whose evolutionary logic is largely unresolved, but whose physical consequence is that the ocean's fluid dynamics become legible in living light, every vortex and pressure gradient mapped by the cells that inhabit it. The glow around your own torso traces the exact boundary between disturbed and undisturbed population, a continuously updated record of your heartbeat and breath rendered in organisms so small that ten thousand of them stacked end to end would reach only as high as your knee.
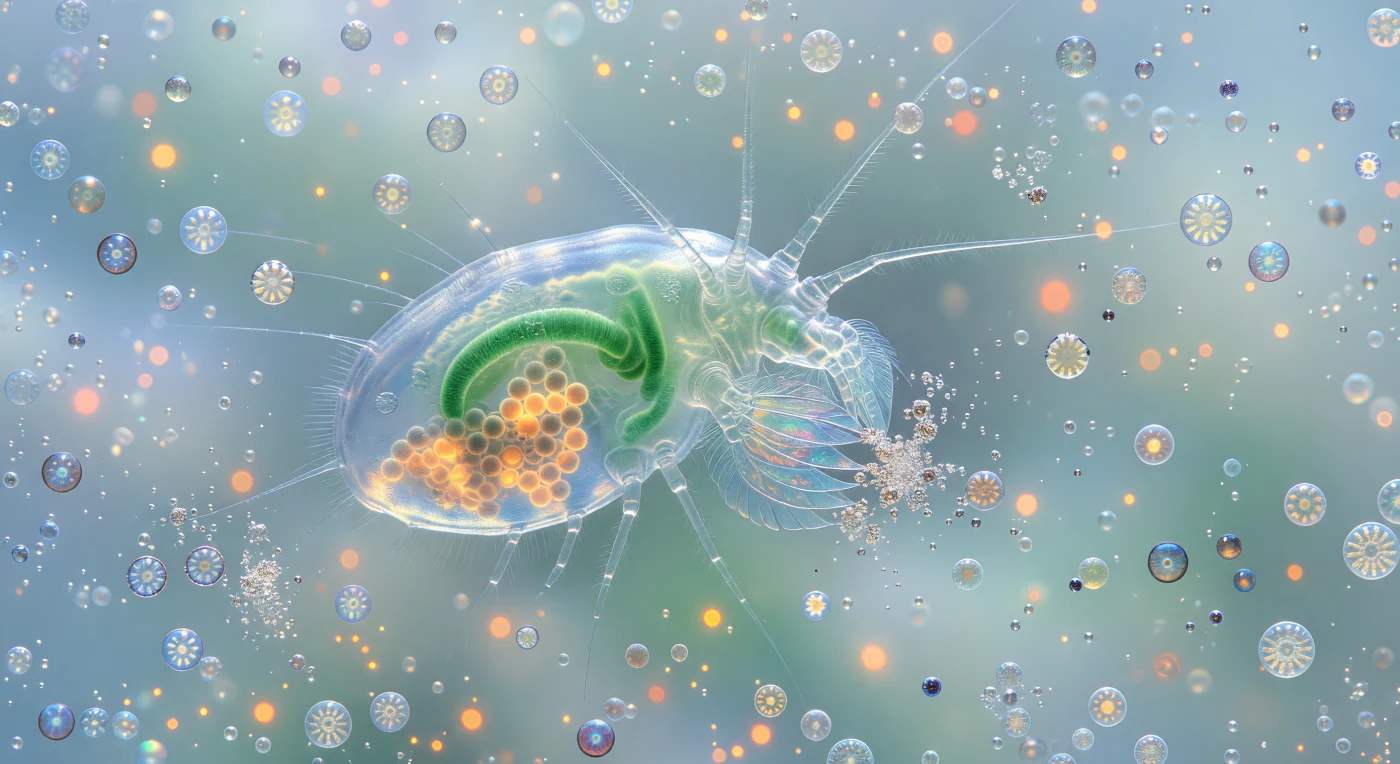
You hover two millimeters from a creature the size of a rice grain that is nonetheless an architectural presence, its chitinous carapace so transparent that jade-green gut contents and dense amber egg clusters burn through three layers of cuticle like lantern glass held to sunlight, every internal organ backlit by the diffuse blue-green luminance filtering down from the ocean surface above. The animal is a *Calanus* copepod, one of the ocean's dominant grazers, and its feathery maxillipeds — appendages arrayed like overlapping fans of blown glass, each seta hair-thin and refractive — tremble at the edge of vision as they intercept individual *Emiliania huxleyi* cells drifting inward along an invisible feeding vortex, the current legible only in the slow orbital rotation of whole coccospheres as they spiral toward the mouth, their interlocked calcite wheel-plates flashing from silver to pale gold depending on the angle of encounter. Where the maxilliped tips make contact, coccoliths detonate into white glitter bursts — each crushed two-to-four-micron calcite disc tumbling and strobing before the current sweeps it aside, releasing dissolved organic carbon that the surrounding bloom water will redistribute through chemical gradients no eye can follow. The water itself is not empty but a luminous turquoise suspension, tens of millions of detached coccoliths per milliliter scattering the available light into a soft glowing fog in every direction, so that distance collapses entirely — three body-lengths back the world dissolves into blue-white haze, intact coccospheres drifting through it like tiny armored planets trailing faint red autofluorescence from their chloroplasts, the whole living medium pressing inward from all sides as the copepod's feeding geometry organizes everything around it into vectors of approach, capture, and glittering calcite destruction.

You are suspended in a column of deep oceanic blue where the sun exists only as a trembling silver smear above, its light stripped to cool blue-green wavelengths by fifty meters of water column, and every surface you see is lit not from any direction but from the water itself. To your left, the diploid C-phase cell fills your field of vision like a baroque chandelier carved from mineral precision: a coccosphere of overlapping cream-white calcite plates, each coccolith a wheel of interlocking crystal blades whose trigonal geometry throws back the ambient light as cold prismatic glints of white and pale gold, the entire armored surface so architecturally dense — radial spokes, interlocking shields, tiny central fenestrae — that the cell appears almost self-luminous against the surrounding ultramarine. To your right, separated by a gulf of open column that your Brownian drift makes feel vast, the haploid N-phase twin offers a startling contrast in every material register: entirely unarmored, its soft translucent membrane of greenish cream reveals the amber-jade warmth of chloroplasts beneath converting even this dim filtered light, and its two flagella — biological cables a fraction of a micron across — are frozen at maximum arc, each one a graceful sinusoid catching ambient glow like wet glass filaments, while the delicate haptonema coils between them in a loose spring, betrayed only by its faint refractive edge. These two cells represent the same species caught at opposite poles of its life cycle, the calcified diploid phase building the mineral armor that will eventually sink as geological sediment and the naked flagellated haploid phase trading structural protection for motility, both strategies written in the same deep water column where Brownian jostling and viscous drift govern all motion and detached free coccoliths drift at varying distances behind them, some close enough to resolve their full geometry, others reduced to white sparks of scattered light that give this infinite blue world its only sense of depth.

You stand at the base of a vertical white world that rises until it becomes sky, the chalk face blazing under raking afternoon light so intensely that the stone seems luminous from within, each pale surface a compressed biography of ancient ocean life. What appears as smooth white rock is in fact the accumulated calcite remains of countless generations of microscopic algae — coccolithophores no larger than a fraction of a hair's width — whose calcite plates sank through Cretaceous seas over tens of millions of years, layer upon patient layer, until burial and pressure transformed biological snow into this cathedral of biogenic limestone. The dark flint bands slicing horizontally across the cliff face at irregular intervals are chemical signatures of that same seafloor, zones where silica migrated through the sediment during diagenesis, each band a compressed moment of deep time locked in glassy stone against the surrounding chalk. A single fulmar resting on a narrow ledge halfway up suddenly makes the scale vertiginous and real — the bird a centimeter of apparent size against hundreds of meters of geological record, its living warmth pressed against the cold archive of a billion dead blooms. To stand here is to be dwarfed not by rock alone but by biological time itself, the cumulative weight of organisms that photosynthesized and calcified and sank in an ocean that no longer exists, their invisible geometry dissolved by pressure yet their mass forming everything you see.