Scientific confidence: High

Looking straight up from the seafloor, you see a forest of slender jade columns rising from bleached white limestone, each one a single living cell no taller than your finger, crowned by a luminous disc that glows chartreuse and straw-yellow as Mediterranean morning light passes clean through its radial segments like sunlight through a rose window. These are *Acetabularia acetabulum*, among the largest single cells on Earth — each entire stalk, holdfast, and umbrella cap a solitary nucleus commanding centimetres of organised living architecture, the chloroplasts massed along the sun-facing flank in a slow migration invisible except as a barely perceptible deepening of green. The caps are gametangial structures, their precise wedge-shaped rays packed with nuclei awaiting the signal to release gametes, the whole disc a reproductive organ built and governed by one enormous nucleus tucked far below in the rhizoid, anchored in pale crystalline rock by finger-thin holdfasts a few millimetres wide. Above the colony, shafts of caustic light ripple through a turquoise water column alive with suspended particles, the ocean surface glittering overhead like hammered silver, while between the stalks cool shadow is cut by transmitted slivers of warm green wherever overlapping caps pool their glow into something almost golden. The scene reads as cathedral and tide pool simultaneously — an architecture of breathtaking geometric precision assembled, without division of labour, by a single cell.

You are gazing down through four centimeters of mirror-still tidepool water at a living landscape that belongs, impossibly, to a single cell — or rather, to thousands of them, each one the size of a small seed yet here reading as boulders in a cobblestone terrain that stretches across coal-black basalt toward an amber fringe of encrusting sponge. The carpet below is *Caulerpa racemosa*, a siphonous green alga whose entire sprawling thallus — holdfast, stolon, and every one of these tightly packed spherical ramuli — exists as one continuous coenocytic cytoplasm enclosed by a single plasma membrane, no internal walls dividing the flowing greenness within. Noon caustics shiver and lock and dissolve across the glaucous blue-green surface, threads of refracted light tracing the microstructure of the water's skin before dissolving into the ambient teal luminescence of the pool, while each sphere catches its own hard white specular point so that the whole colony glitters like a field of frosted glass beads. What the eye receives as topography — hills and shadow crevices, the matte powder of the bloom against the wet gleam of the inner curve, the deep cobalt shadows where ramuli press against one another — is cytoplasm and chloroplast and turgor pressure made architectural, a single organism wearing the convincing costume of a landscape.

Hovering centimeters above the Mediterranean seafloor at twelve meters depth, you find yourself eye-level with one of biology's most radical architectures: an unbroken emerald carpet of *Caulerpa taxifolia*, each feathered frond a single coenocytic organism — no internal walls, no compartments, just one continuous cytoplasm enclosed within a membrane that stretches across the entire thallus, from stolon to pinnule tip. Each rachis, no thicker than a knitting needle, branches into dozens of paddle-flat pinnules arrayed with the geometric precision of a living tessellation, their chlorophyll-dense cortex so concentrated that the thinnest edges glow translucent lime-yellow where diffuse, caustic-scattered sunlight rakes diagonally down through the water column above. Within these glassy walls, cytoplasmic streaming moves silently — rivers of granular green cytoplasm circulating at a few micrometers per second, the cell's internal logistics operating at a scale you could almost perceive if you held still long enough. The sole interruption to this alien monoculture is a single bleached gastropod shell resting at the boundary between sand and stolon mat, its ivory calcite surface an incongruous relic of animal architecture against the relentless, vegetative intelligence of a world built from a single cell, repeated to the horizon.

You hover motionless just above a limestone crevice fracture, close enough that the trembling mercury sheet of the Caribbean surface fills your entire peripheral vision above, while fifteen *Valonia ventricosa* spheres occupy the crevice floor like pressurized glass baubles — each one a single cell two to four centimeters across, their taut walls pulled so tight they read less as biology than as blown borosilicate, shading from deep saturated emerald at the flanks to chartreuse where caustic threads of focused sunlight snake and bloom across their surfaces in continuous fluid reorganization. Each sphere is an individual coenocytic cell under extraordinary turgor pressure — its multilayered cellulose wall wound in opposing helical fiber arrays that lend the surface a faint silky crosshatch rather than a true mirror finish — and where the light strikes at near-normal incidence, the translucency of that wall reveals the interior as a single luminous jade volume, the massive central vacuole diffusing chlorophyll light outward so that each sphere glows as if self-illuminated, a dense green lantern with a darker ectoplasmic chloroplast rind pressed millimeter-thin against the inside of the glass. A bristle worm threads between two of the larger spheres in copper and rust iridescence, its body dwarfed by the cell mass it navigates — an animal composed of billions of cells picking its way through the architecture of just fifteen — while behind the cluster the crevice walls bear their crustose coralline algae in chalky pink granularity, the textural opposite of the glassy perfection pressing against them, and the entire scene holds suspended in a single sharp instant, every caustic filament crisp, every bristle a hair of light.
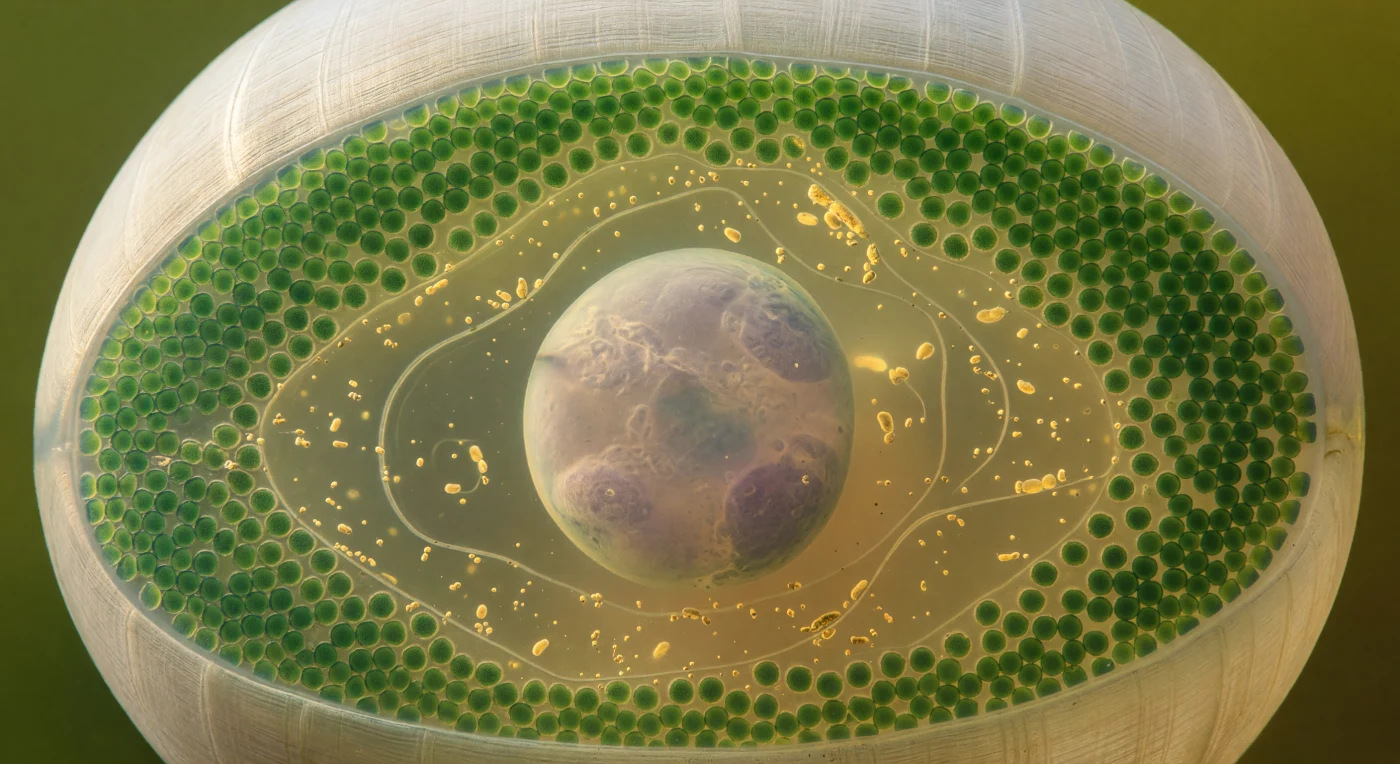
You are suspended inside the rhizoid chamber of a living *Acetabularia*, a green alga capable of reaching ten centimeters in height while remaining a single cell — and the vast pale sphere dominating your field of view is its one and only nucleus, an 80-micron structure that must coordinate the entire organism's growth, regeneration, and reproduction alone. Pressed against the surrounding cell wall in an unbroken mosaic, hundreds of chloroplasts form a stained-glass pavement of deep jade, each disc glowing with its own emerald saturation as sunlit seawater transmits through the cellulose-microfibril wall from outside, filling the enclosed space with warm amber-green light that seems to emanate from every direction at once. The nucleus itself is not a hard object but a softly luminous membrane-bounded world, its pearl-grey surface shading into lavender where internal chromatin condenses, and at its heart the nucleolus sits as a denser indistinct mass — a factory of ribosomal RNA production felt more than resolved through the translucent nuclear envelope. Between wall and nucleus, the cytoplasm moves in the slow looping arcs of cyclosis, golden granules and slivers of endoplasmic reticulum drifting at geological pace around the nucleus and back, the whole interior a warm, slightly viscous medium whose unhurried motion belies the fact that this streaming is the cell's only means of distributing materials across a body that, by any conventional measure, should be far too large to function without a circulatory system.

You are standing inside a single living cell, though nothing about the space suggests smallness — the hollow corridor of a *Caulerpa* stolon lumen stretches away before you like the nave of a Gothic cathedral, its curved walls radiant with the packed emerald and viridian luminescence of thousands of chloroplasts glowing in diffuse transmitted light, the cellulose matrix between them catching the green as a faint silver-gold textile weave pressed against translucent glass. This is a coenocytic organism: the entire *Caulerpa* plant — holdfast, stolon, upright fronds — constitutes a single continuous cell, its cytoplasm undivided by any internal wall, held open and pressurized by the vacuolar fluid that now fills the dark bottle-green space around you. The trabeculae crossing the lumen are cytoplasmic strands, tensile structures of condensed ectoplasm that brace the cell wall against collapse under its own turgor, and they recede in ranked perspective — nearest ones thick and ivory-warm, catching the chloroplast glow, the furthest reduced to cold hairline threads fading into luminous obscurity — their pointed junctions with the curved wall forming arches no architect designed. Along these strands and drifting free through the vacuolar fluid move clusters of amber granules, organelles and starch bodies carried by cytoplasmic streaming at speeds too slow to register except as the faintest suggestion of current, golden motes suspended in a cathedral that has lit and held itself open, without a single wall between its rooms, for the entire duration of its life.

You are hovering centimeters above the floor of the world, weightless in water cold enough and pressurized enough to feel solid, while directly ahead the organism fills your lower field of vision entirely — a hunched, charcoal-black mound of extraordinary textural complexity, its surface assembled grain by grain and shell by shell into something that reads less as biology than as geology, a miniature karst landscape of pits and ridges where the ROV's harsh blue-white LED cone carves razor-edged shadows into every crevice. This is *Syringammina fragilissima*, a xenophyophore and one of the largest single cells on Earth, a solitary foraminifer that has spent years or decades extending cytoplasmic tubes outward through abyssal sediment, agglutinating mineral grains and the calcareous shells of smaller foraminifera into a rigid protective test now fifteen centimeters across — an architectural feat accomplished without a single internal cell wall, all of it one continuous cytoplasm housing millions of nuclei in a branching tube network beneath that dark mineral crust. Individual shell fragments catch the LED wash as specular highlights bright as broken porcelain against the surrounding matte matrix, while the mound's boundary dissolves into the pale ashen sediment in lobes and peninsulas where the organism continues its lateral expansion, colonizing new substrate with the patience of stone. Three body-lengths away the light fails absolutely — not ordinary darkness but the physical blackness of five kilometers of ocean pressing down from above — and through the lit water column above the mound, marine snow particles drift in near-perfect stillness, witnesses to a moment of borrowed illumination in a place that has otherwise known only dark for geological time.

You stand at the exact center of a living sphere, and every direction you look curves away from you in an unbroken arc — a sealed cathedral three centimeters across whose walls are the cell itself, a single *Valonia ventricosa* algal cell maintaining its architectural integrity through turgor pressure as precise and rigid as blown glass. The entire enclosing surface blazes with a continuous mosaic of chloroplasts pressed flat into the cortical ectoplasm, their collective photosynthetic pigment painting the spherical horizon in saturated emerald, shifting to forest shadow at the poles where light from the surrounding sea diminishes, the whole inner dome functioning as a self-illuminating lamp whose green radiance reaches you from every direction simultaneously with almost no attenuation through the extraordinarily still, straw-yellow vacuolar sap — a fluid chemically alien to seawater, its elevated potassium concentration invisible but somehow felt in the density of the medium. Through the living green mantle, barely resolved, the cellulose microfibril layers of the wall itself form a cream-white herringbone lattice — opposing diagonal bands laid down in successive laminations, a woven structural textile holding the sphere rigid against the osmotic pressure differential that would otherwise collapse or rupture this improbable mononuclear architecture. Occasionally, a vesicle or suspended crystal drifts through the sap at speeds so glacial it appears stationary, catching the ambient green light as a momentary fleck of gold — the only visible evidence of the cytoplasmic dynamics and ionic chemistry quietly sustaining, within a single membrane-bound cell, the entire biology of this organism.

Peering laterally into this cleanly sheared wall of coastal sediment, you find yourself suspended in warm, particulate darkness beside individual sand grains the size of boulders — frosted quartz spheroids and feldspar plates that dwarf the translucent amber rootlets threading between them with extraordinary precision. Above you, the stolon of a *Caulerpa* plant arches like a pressurized cylinder of green glass, its lacquered emerald surface catching a thin meniscus of coastal light along its upper edge — and yet this entire structure, from the glistening stolon overhead to the finest filament disappearing into brown-black sediment two centimeters below, is a single cell, one continuous cytoplasm bounded by a single membrane, containing no internal walls despite encompassing a volume that stretches meters across the seafloor. The rhizoid network descending from the stolon's underside branches with fractal regularity, primary cylinders a hundred microns wide splitting into secondary and tertiary rootlets that conform precisely to the curved topography of individual mineral grains, anchoring the organism physically while also absorbing dissolved nutrients from interstitial porewater — a task accomplished without the division of labor that multicellular plants require. The light gradient tells the full story of this buried world: bright, blue-green diffuse radiance at the sediment surface deepening through amber-tan to near-total darkness below, the finest rhizoid tips visible only as faint warm outlines before vanishing into granular obscurity.

Three individuals of *Acetabularia* rise from the bleached aragonite floor like architectural monuments — a juvenile trailing feathery hair whorls of radiating cytoplasm, an adolescent swelling into an unresolved proto-cap of fusing ray primordia, and a mature organism spreading its perfectly tessellated reproductive disc like a green parasol — each one a single cell, each one centimeters tall, each stage of development separated in space as clearly as illustrations in a textbook. Warm raking light drives in from the right, turning the cap margins to stained-glass emerald while the open water beyond holds everything in deep oceanic blue, and on the chalk-pale substrate beneath the mature cap a crisp circular shadow marks where living tissue refracts the sun into a thin caustic ring. *Acetabularia* is among the most extraordinary organisms in biology: despite reaching up to ten centimeters, each individual is a single uninucleate cell — its one enormous nucleus sequestered far below in the rhizoid, while morphogenetic signals encoded in long-lived messenger RNAs travel the length of the stalk to orchestrate cap formation entirely without cell division. The calcium carbonate impregnating the stalk walls, the chloroplast-carpeted cortex glowing faintly green, the slow rivers of cytoplasmic streaming invisible at this scale but coursing within — every structure visible here is the architecture of one cell deciding, slowly and over weeks, what shape it will become.

You hover beside the uppermost fork of a *Halimeda* bush, eye-level with its topmost segments, looking along an architecture that reads less like a living thing than like a mineral jeweller's construction — a branching chain of flat, coin-shaped lozenges in chalky aragonite grey, each one articulating with the next through a pinched, intensely vivid-green node where living, unchristallised cytoplasm holds the calcified discs in supple articulation, the whole structure swaying in the residual surge with the slow, weighted rhythm of a necklace. What you are witnessing is one of biology's most radical paradoxes: this entire branching bush, eight centimetres tall, is a single cell — a coenocytic giant unicell whose cytoplasm flows continuously through every segment and node without internal walls, progressively mineralising its own outer cortex into a crust of crystalline aragonite that serves simultaneously as skeleton, grazing deterrent, and carbonate sediment factory for the seafloor below. Across the upper faces of each disc, constellations of ochre-and-gold diatom epiphytes catch the eight-metre-filtered light as warm amber sparks against the cool matte limestone surface, while fine carbonate flour — shed from the segments themselves — drifts through the water column as brief silver motes, a slow mineral snow that will eventually accumulate into the pale buff sediment visible in fragments beneath the lower branches. Behind and beyond, the seagrass blades dissolve entirely into a luminous, shifting bokeh of jade, khaki, and amber-gold, a warm enclosing wall that throws the rigid, fractal precision of this single cell's stone architecture into extraordinary relief.

You are suspended in water so clear it functions as blue-tinted optical glass, eye-level with a catastrophe still unfolding in slow motion — ten centimetres above a sunlit reef floor, where the collapsed remains of a ruptured *Ventricaria ventricosa* drape across coralline rubble like a deflated lantern, its thick olive-dark walls curling inward at the torn edges, glistening with residual cell sap. Erupting outward from that collapsed interior, a dispersing constellation of daughter protoplasts — each a near-perfect sphere, ranging from sand grain to peppercorn in size — tumbles through the water column in every direction, catching tropical shafts of light so that their chloroplast-packed cores read as deep saturated emerald in shadow and blazing citrine-gold at their illuminated rims. This is *segmentation*, the organism's primary reproductive strategy: a single giant coenocytic cell, one of the largest unicells on Earth, ruptures under physiological stress or mechanical damage and releases hundreds of totipotent protoplasts, each carrying a full complement of chloroplasts, cytoplasm, and genetic material sufficient to establish a new individual if it survives to settle on suitable substrate. The nearest protoplasts are architecturally present — their membranes showing the faint quilted mosaic of chloroplast packing beneath, refractive chemical gradients from released cell sap bending the background reef into gentle lensing arcs around each sphere's equator — while further out, at five to fifteen centimetres, they dissolve progressively into glowing smears of emerald-gold light against open water, the whole dispersing cloud acquiring the character of a slow stellar detonation, a green-gold nebula blooming in geologic silence above the chalky pink and purple reef below.
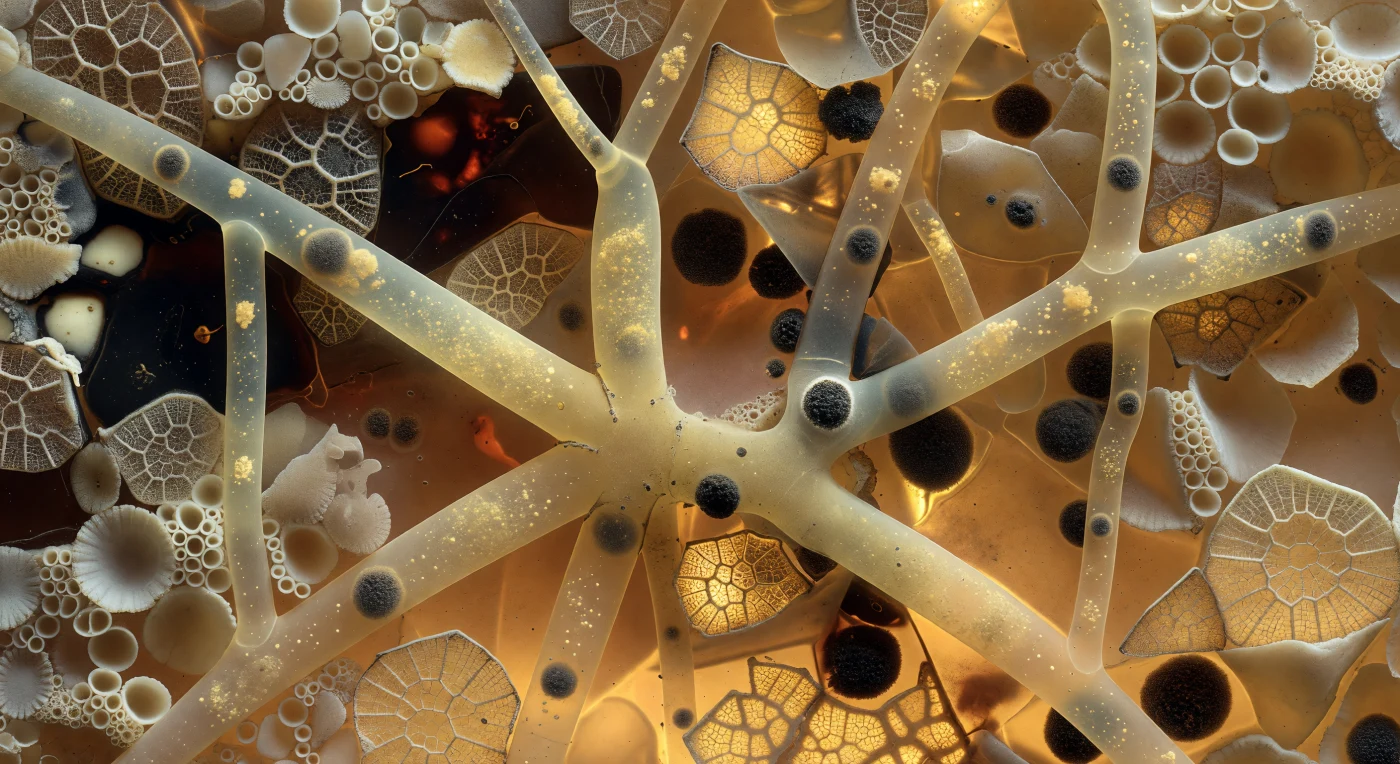
You stand suspended inside the living architecture of a Xenophyophore — one of the ocean's most improbable giants, a single cell that may span tens of centimetres, yet contains within it this intricate mineral fortress of its own construction. Around you, the test wall is a densely packed mosaic of scavenged debris: foraminifera shells pressed together like pale ivory flagstones, radiolarian lattice fragments glowing where warm amber light filters through their preserved silica geometry, and near-black organic cement binding everything into a structure that reads simultaneously as geology and as living tissue. Threading through this lapidary matrix, the linellae channels — branching tubes forty to eighty microns across at true scale — carry pale golden cytoplasm studded with dense charcoal-grey nuclear bodies, their translucent walls barely distinguishable from the surrounding amber field except where a slightly cooler refractive tone betrays living fluid against mineral mass. This is the paradox at the heart of the Xenophyophora: an organism so vast it incorporates the skeletal remains of countless other protists into its own body wall, yet remains throughout a single uninterrupted cytoplasm, its nuclei drifting through warm mineral corridors in unhurried procession, deep on the abyssal seafloor where no light has ever reached except the backlit amber glow of its own biology.
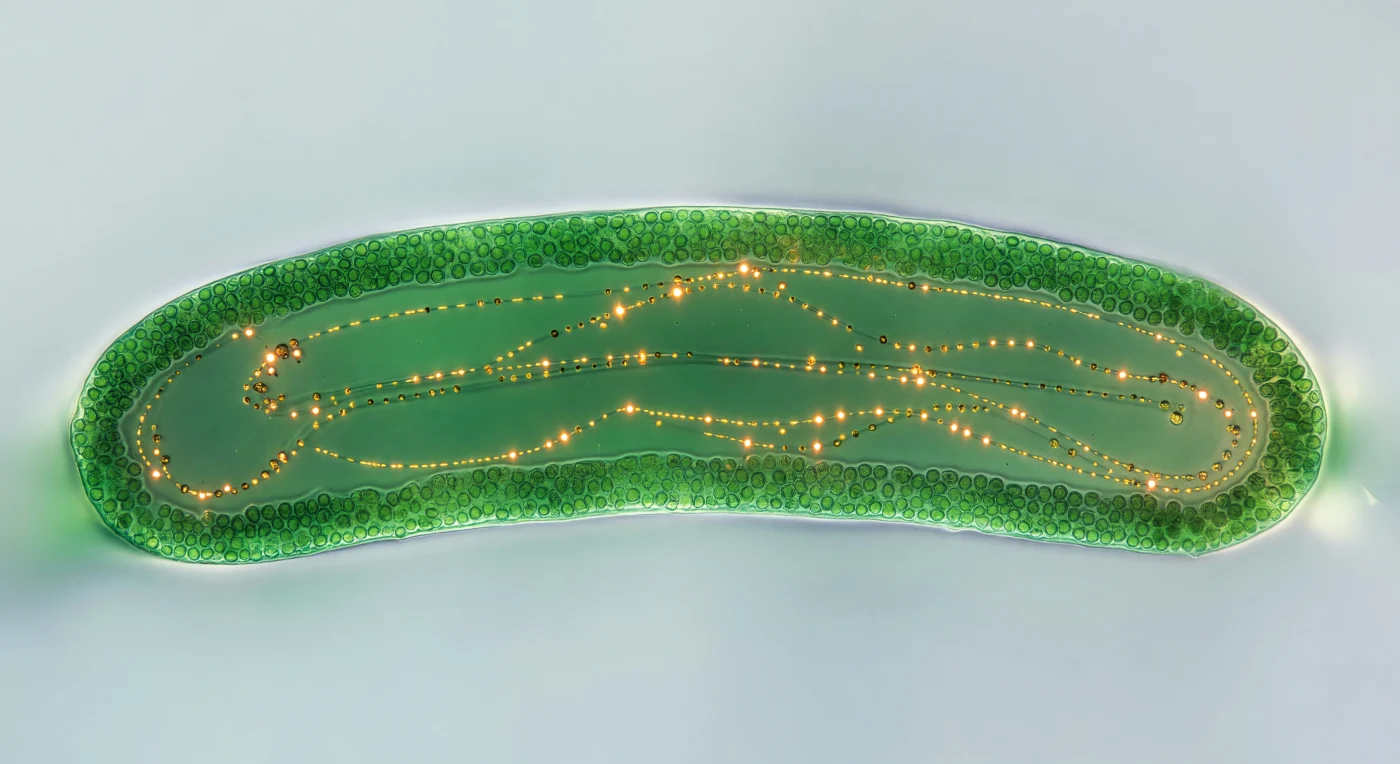
You hover directly above a curved green world that fills the entire field of view — the interior of a single *Caulerpa* stolon, two millimeters across, transformed by transmitted backlight into a self-luminous lantern of jade and malachite, its outer cortical mantle so densely packed with chloroplasts that it reads as a solid mosaic wall, while the central endoplasmic corridor opens inward like the nave of a cathedral carved from living glass. This is a coenocytic organism: no internal walls divide it, no cell boundaries interrupt the continuous cytoplasm that stretches — in the same unbroken cell — through stolons, holdfasts, and fronds that may span meters of seafloor, all of it one. Within the darker central channel, cytoplasmic streaming moves at the pace of slow breathing, amber and honey-gold granules riding invisible actin cables in long arcing trajectories, some catching the transmitted light as sharp orange sparks, others trailing into short ochre dashes — a braided river of organelles conducting photosynthate and signals across a single cell's interior at one to ten micrometers per second. The surrounding seawater is almost invisible, present only as a thin prismatic highlight along the stolon's curved outer wall, beyond which the backlit glass fades to featureless pale grey, leaving the cell suspended in its own radiance like a stained-glass window held in fog.

Three pale stalks rise from the limestone crust before you, each one a single living cell standing as tall as you are wide, their umbrella caps fanning open in precise radial segments like lace cut from frosted sea-glass. Between the irregular cascades of bioluminescent streaks fired by panicked dinoflagellates sweeping through the water column above, the only light in the world is the faint grey-green phosphorescence bleeding outward from within each stalk — chloroplasts suspended in cytoplasmic gel, glowing like foxfire seen through ivory, the thinnest cap margins luminous where the membrane barely holds. When a streak arrives — a sharp, chemical-blue crack through the particulate Mediterranean darkness — raking illumination transforms the caps into architectural sculptures, their radial ribs casting hard-edged shadow-stripes down the stalks and briefly revealing the granular interior fog of a single cell's living organization before absolute darkness swallows the scene again. Each of these organisms is not a colony or a tissue but one continuous cell, its single enormous nucleus resting in the rhizoid holdfast below as a barely perceptible pale node against the encrusted rock, the entire cytoplasm streaming in slow invisible currents through a body visible to the naked eye. The water itself participates — dense with suspended microaggregates that catch each bioluminescent burst as a slow-motion galaxy of sparks — and time here is measured not by any internal clock but by the random panic of invisible organisms above, their flight becoming, briefly, your only sun.















