
You are standing inside the wall of a living bacterium, looking outward through one of the densest biological architectures that life has ever built. What surrounds you in every direction is the peptidoglycan layer of *Bacillus subtilis* — a Gram-positive species that constructs its wall not as a single membrane but as a compressed, multi-layered fortress twenty to eighty nanometers thick, a structure whose depth, at this scale, feels like the interior of a cathedral made from interlocking organic masonry. The glycan strands stretch as long, warm amber rods cross-linked at irregular intervals by shorter peptide bridges, each junction a covalent bond holding compressive turgor pressure that could rupture the cell in milliseconds if the architecture failed — inside *B. subtilis*, internal osmotic pressure reaches several atmospheres, and this amber lattice is the only thing resisting it. Threading between the glycan rungs, the teichoic acid chains hang as dark sinuous polymers, their dense anionic charge shaping the wall's electrostatic environment, regulating divalent cation binding and controlling access by antimicrobial peptides that approach from the pale-blue extracellular world dissolving above. Deep below, the phospholipid inner membrane shimmers in amber warmth, studded with protein complexes that remain invisible from this vantage — and beyond the ragged outer frontier of the wall, where the last glycan threads fray into nothing, the cool aqueous medium opens into a diffuse, luminous void where the bacterium meets its world.
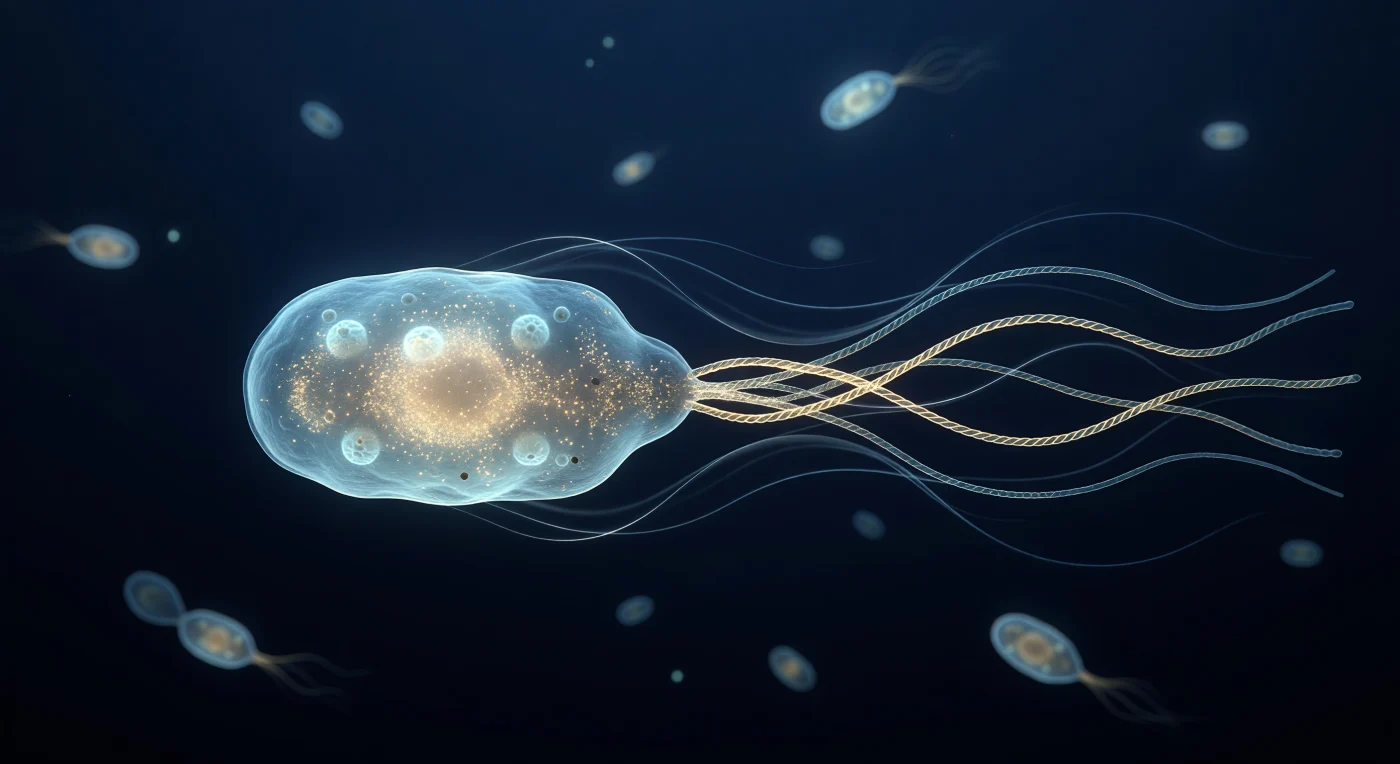
You are suspended just behind the stern of a living *E. coli* cell as it drives itself forward through a world where viscosity rules absolutely and inertia is a concept without meaning. The pale-cyan translucent body fills your field of view like the hull of a slow vessel, its outer membrane shimmering with the faint corrugation of porin rosettes and edged in a thin silver-blue caustic, while the dense amber interior resolves at the poles into the granular scatter of tens of thousands of ribosomes packed into a cytoplasm closer to gel than to water. Behind the cell, four pale-gold helical flagellar filaments have braided into a single superhelical bundle rotating at hundreds of revolutions per second, yet the surrounding medium betrays none of the turbulence that speed would imply at any larger scale — instead, delicate silver-blue Stokes-flow streamlines curve forward in perfectly symmetric arcs, silk-smooth and unhurried, the visual signature of a Reynolds number so vanishingly small that every stroke must be non-reciprocal to produce motion at all. The deep navy water is not empty: a dozen out-of-focus bacterial silhouettes drift in the distance like faintly glowing embers, each already dissolved into soft cyan bokeh, compressing depth until the space feels simultaneously intimate and oceanic — a thermally jittering, charge-saturated immensity in which the only light is the cold phosphorescent self-glow of molecular machinery that has been perfecting this particular solution to locomotion for nearly four billion years.

Floating at the geometric center of a living *E. coli* cell, you are engulfed by a fractal storm of plectonemically supercoiled DNA that loops and re-loops in every direction, filling the entire visual sphere with dense navy-blue braided ropes no wider than a few nanometers yet collectively occupying a compressed cubic micron of space — the bacterium's nucleoid, holding roughly 4.6 million base pairs of chromosome folded into a volume smaller than the wavelength of visible light. The fibers radiate a sourceless cobalt luminance outward, bathing the environment in cold indigo gradients that deepen toward violet in the most entangled depths, while warm yellow-white beads of nucleoid-associated proteins — HU and Fis — clasp every sharp plectonemic bend, kinking the DNA into organized pleats and returning the blue glow as amber-gold constellations distributed through the cloud like embedded stars in nebular gas. The intervening medium is no empty solvent but a crowded, gel-like cytoplasm at roughly 300 mg/mL protein concentration, where molecular crowding and Brownian forces utterly replace gravity as the dominant physics, and the aquamarine haze between adjacent supercoil loops — separated by mere tens of nanometers — carries the refractive quality of smoky glass. Only at the nucleoid's outermost margin, where the DNA thins and cytoplasm reasserts itself near the inner membrane, do the dark cinder-brown spheres of ribosomes appear, soft and slightly out of focus, hovering in the transitional zone between organized genetic architecture and the wider cellular machinery beyond.

You hover over a living landscape that defies easy comprehension — the outer membrane of an *E. coli* cell, its lipopolysaccharide surface rolling in slow thermal waves driven by the constant agitation of molecular-scale energy, a bilayer so chemically asymmetric that its outer leaflet is armored with Lipid A and long polysaccharide chains rather than the phospholipids that line its inner face. Every twenty nanometers or so, OmpF and OmpC porin trimers pierce the membrane as rigid beta-barrel cylinders, their hollow interiors forming aqueous channels just wide enough to admit small solutes while excluding larger molecules, and their protein surfaces darkly iridescent where cold ambient light catches the geometric fluting of their rims. At this scale, Brownian motion is not a background abstraction but a felt reality — the entire plain shudders with thermal fluctuations that, at larger scales, would average to stillness, and the dense ionic atmosphere pressing against the membrane surface represents the Debye screening layer, a diffuse electrostatic haze a few nanometers thick where counterions crowd against the membrane's net negative charge. On the far horizon, the flagellar basal body rises as a multi-ringed protein machine spanning the full thickness of the cell envelope, its L-ring anchored in the outer membrane, its MS-ring embedded in the inner membrane far below, and its hook already curving upward into the viscous aqueous medium — the coupling interface for a rotary motor that, when running, spins at hundreds of revolutions per second and drives the cell through a fluid that, at this scale, behaves more like cold syrup than open water.

You are submerged inside a living bacterial cell, hovering just beyond the pale fibrous boundary of the nucleoid, and the world around you offers no open space whatsoever — only a warm, amber-resinous gel packed so densely with 70S ribosomes that the gaps between their dark, umber-gray surfaces read less as distance than as mortar between ancient stones, each sphere separated from its neighbor by a mere five to ten nanometers in a medium running at roughly 300 milligrams of protein per milliliter. Threading through this granular press, polysome chains sag across the middle distance like irregular pearl necklaces — six ribosomes strung along barely visible mRNA filaments that glow a faint cream-white before dissolving into deeper brown opacity — actively translating proteins at a rate of fifteen to twenty amino acids per second per ribosome, multiplied across tens of thousands of such machines simultaneously active in this single cell. To the left, a GroEL chaperonin barrel dominates a full quadrant of the scene like a petrified tree stump, its double-ring architecture rendered in deep slate-gray, its hollow central cavity offering a slightly cooler interior where misfolded proteins are sequestered and refolded through ATP-driven conformational cycling. The only color relief in this warm monochrome crush comes from occasional GFP-labeled proteins flaring briefly as small emerald embers before being occluded again by another passing ribosome — transient beacons in a cytoplasm so molecularly crowded that even diffusion is slowed, and the boundary between structured matter and empty solvent has effectively ceased to exist.

Looking straight down at an oblique angle from just above the substrate, the eye takes in a silent metropolis of GFP-jade mushroom towers — dense microcolonies of *Pseudomonas* cells rising twenty to forty micrometers from the base plane, their domed crowns packed with individual rod-shaped cells arranged in tight rosettes, each bacterial body a faint olive sliver catching oblique light like wet ceramic against the cooler interior glow. Between the spires, a network of ink-black water channels carves clean hydraulic lanes through the landscape, their smooth walls evidence of advective flow that has scoured the matrix free of cells, while the interstitial spaces fill with a warm amber extracellular polysaccharide gel — semi-translucent, slightly hazy, sagging between towers like stretched caramel and emitting faint orange autofluorescence from deep within its layered bulk. At the base of the tallest, most mature towers, propidium-iodide crimson bleeds through the semi-transparent canopy in blurred coral patches, marking cells whose membranes have failed in the oxygen-starved shadow of their own colony — a hypoxic zone hidden beneath the green and revealed only by the confocal-style optical sectioning that allows the eye to read through living matter. The entire scene stretches to a horizon measured in mere tens of micrometers, yet registers as vast: a self-constructed city of biological architecture, built from secreted polymers and cooperative cellular behavior, utterly moist, utterly inhabited, and operating at a scale where viscosity rules and every surface exerts its pull.
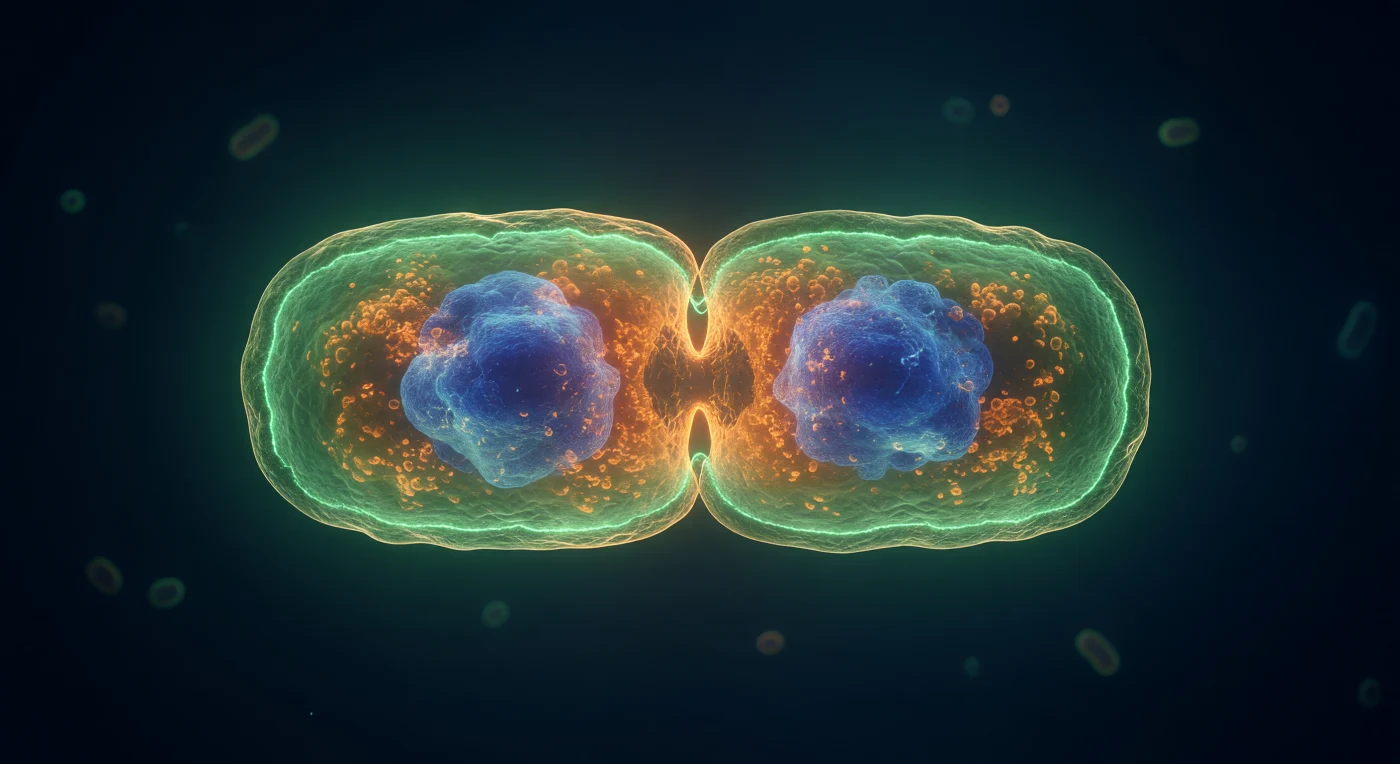
You orbit the equatorial belt of an *E. coli* cell caught in the most decisive instant of its existence — the moment of cytokinesis, where a single organism becomes two. Ringing the deepest point of the hourglass constriction, the FtsZ Z-ring blazes as a continuous crown of electric GFP-green: a self-assembled polymer scaffold of tubulin-like FtsZ proteins, hydrolyzing GTP to generate the constrictive force that pinches the membrane inward, its luminous band no wider than a few protein filaments yet encircling the entire 1-μm circumference with extraordinary precision. The FM4-64-stained inner membrane traces that constriction as a warm amber double-curve, bowing inward under molecular tension while the peptidoglycan remodeling machinery — amidases, synthases, the divisome complex anchored to the Z-ring — actively remodels the cell wall in coordinated waves that propagate around the ring faster than any mechanical tool could manage. Deep within each nascent daughter half, DAPI-saturated nucleoid masses glow as dense cobalt nebulae, having already completed their segregation driven by the MukBEF condensin complex and the entropic spring forces of chromosome elasticity, while the surrounding cytoplasm — packed to ~300 mg/mL with ribosomes, metabolic enzymes, and signaling molecules — glows with the warm amber density of a living gel rather than any simple aqueous solution. In the near-lightless navy medium beyond the cell surface, where visible-light wavelengths approach the diameter of the cell itself and diffraction softens every boundary into a jade aureole, the physical world operates entirely without gravity or inertia — only thermal noise, viscous drag, and the relentless chemical potential gradients that drive this nanoscale drama toward its inevitable, irreversible conclusion.

Before you stretches a chain of fifteen magnetite crystals arranged with an almost architectural exactness that seems impossible for something assembled by a living cell, each black faceted body rising roughly four times your own height and throwing blue-violet metallic gleams across the amber haze of the crowded cytoplasm. These are magnetosomes — organelles unique to magnetotactic bacteria like *Magnetospirillum* — in which the cell biomineralizes iron oxide into crystallographically precise cuboctahedral magnetite (Fe₃O₄), each crystal enclosed within a dedicated lipid membrane vesicle whose bilayer surface trembles visibly with thermal undulation, lending every dark mineral body a faint pearl-white halo. The entire array is anchored to a MamK filament running beneath the chain like a taut cobalt-blue rail, a prokaryotic actin homolog whose polymerization actively positions and spaces each crystal so that their magnetic moments sum into a single cellular compass needle aligned with geomagnetic field lines. Around you, the cytoplasm is no transparent solution but a dense, semi-opaque amber gel — ribosomes packed at concentrations approaching three hundred milligrams of protein per milliliter — through which Brownian motion continuously jostles the nearest granules in slow, stuttering drifts that remind you gravity has no authority here, only thermal noise and viscous drag. The inner membrane curves away at the periphery like the wall of a vast humid cavern, its surface studded with respiratory complexes catching the diffuse bioluminescent glow of the interior, before the world thins into the cooler blue-gray corridor of the periplasm and then absolute darkness beyond the cell wall.
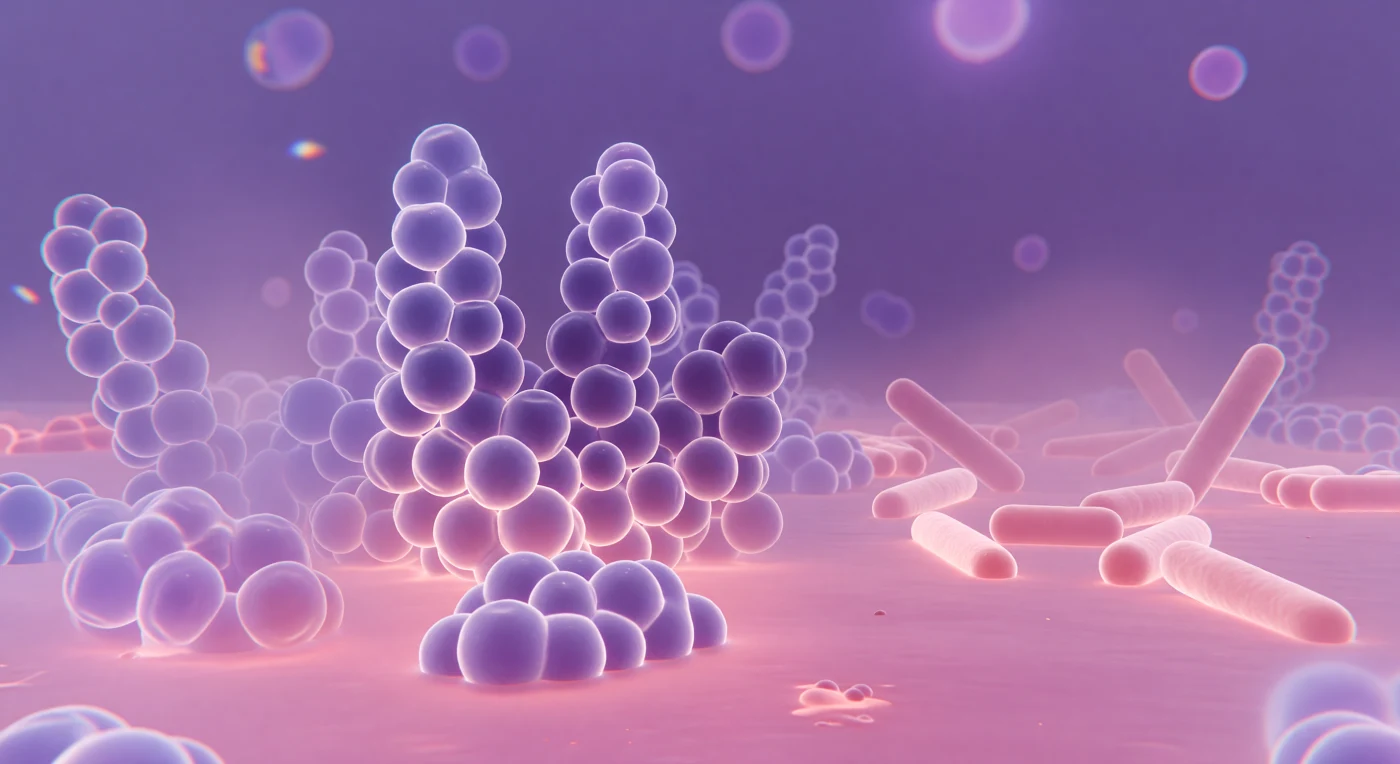
You stand inside a world saturated in deep violet and warm rose, surrounded by towering spherical masses that cluster overhead and on all sides like enormous soap bubbles compressed by their own abundance, each one a *Staphylococcus aureus* coccus whose thick Gram-positive peptidoglycan wall has greedily bound crystal violet dye, staining the entire landscape a bruised, luminous purple. The light arrives from everywhere at once, transmitted through the specimen like illumination through stained glass, casting no shadows but instead giving every curved surface an inner glow, while around each sphere's widest girth a faint diffraction corona shimmers — a physical consequence of visible light bending around objects barely larger than its own wavelength, ~400–700 nm meeting a 1-μm cell, the optics of glass and biology negotiating at their shared limit. Depth collapses abruptly: cells displaced even slightly above or below the focal plane dissolve into translucent lavender halos, their membranes bleeding into the warm safranin-pink counterstain that pools across the ground like rose-tinted water between the cluster's bases, marking the Gram-negative *E. coli* rods in the far quadrant — pale dusty pink, three times your length, their thinner peptidoglycan unable to retain the primary stain. At the visual periphery, chromatic fringes of blue and amber rim the ghost outlines of distant cells, honest artifacts of glass optics that make this single luminous focal plane feel like a window cut through an infinite layered city of living architecture extending upward and downward, purple and pink, far beyond sight.
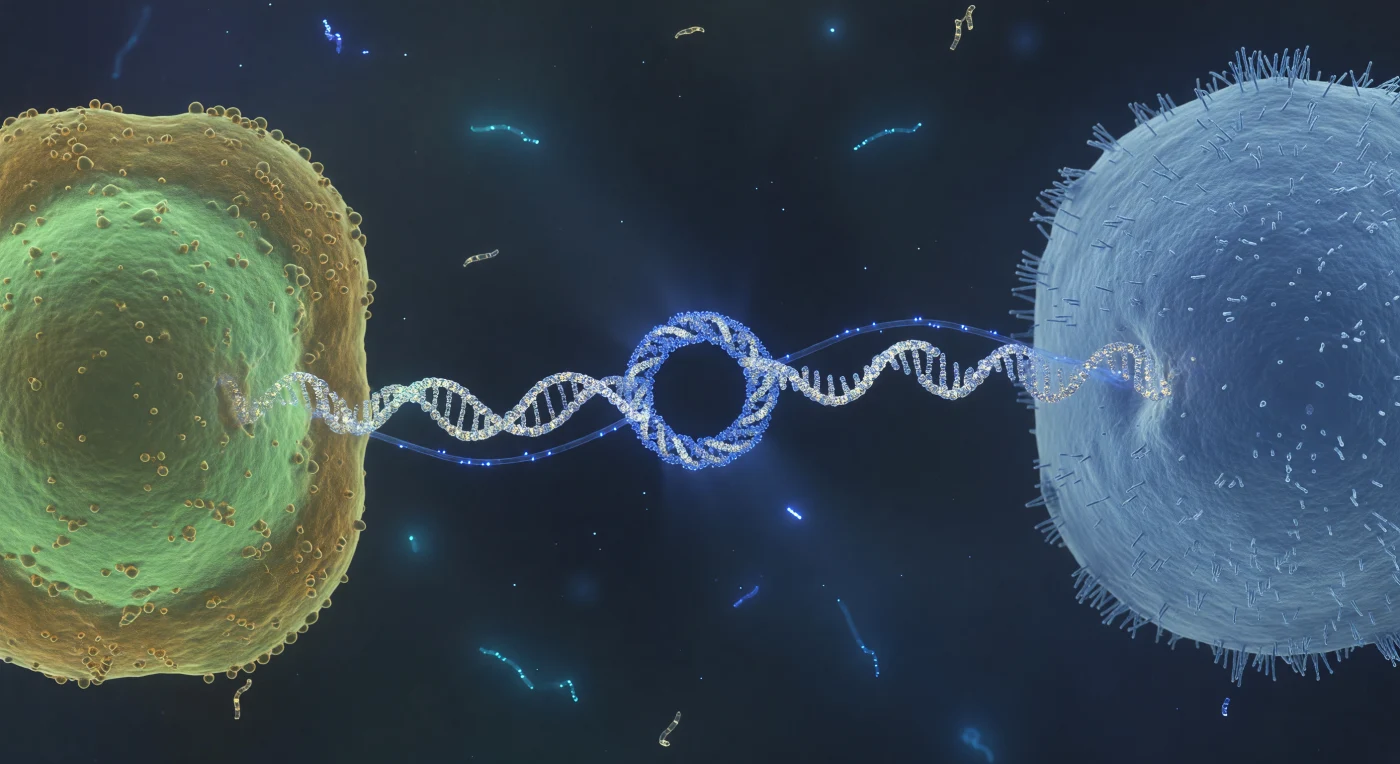
You hover at the midpoint of something that should not exist at this scale — a taut, luminous cable no wider than eight protein subunits, stretched across the gulf between two living worlds. The pilus is a conjugative type IV structure, a helical polymer of pilin subunits assembled and extended through the donor's inner and outer membranes, capable of retraction as well as extension, its architecture evolved not for locomotion but for molecular intimacy: it draws recipient and donor into direct membrane contact, collapsing the distance until the conjugative pore — a 20-nanometer docking annulus of multiprotein relaxosome and mating-pair stabilization complexes — aligns across both envelopes and opens a channel through which a single-stranded DNA copy of the plasmid threads, nicked at its origin of transfer and piloted by a relaxase enzyme covalently bound to its leading end. What moves through that channel now is not passive diffusion but a directed, ATP-powered translocation event — the electric-blue filament pulsing from donor to recipient is a strand of mobilizable DNA crossing a biological border in real time, carrying cargo that may include antibiotic resistance genes, virulence factors, or metabolic innovations, a horizontal transfer event of the kind that has reshuffled bacterial genomes across geological time. In the surrounding fluid, the low Reynolds number regime makes the aqueous medium feel viscous and absolute, thermal fluctuations registering as a ceaseless molecular tremor at every surface, while the ghostly cyan wisps drifting in the far background — free plasmid fragments or degraded nucleic acids tumbling under Brownian bombardment — remind us that this intimate handshake unfolds inside a molecular ocean where information and material are always in motion, always available to be captured.
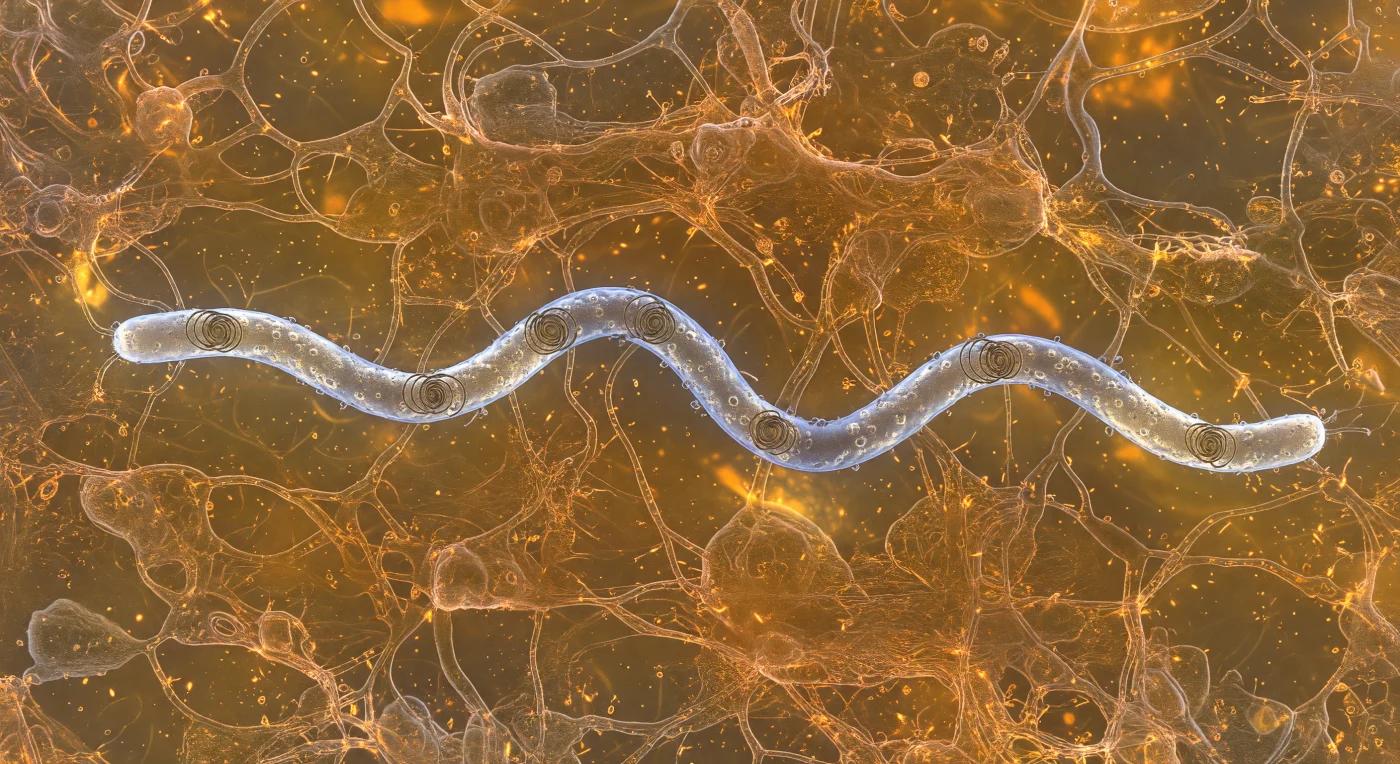
You find yourself suspended in a viscous amber world, immersed in the mucin gel lining a biological passage — a three-dimensional lacework of glycoprotein strands glowing with diffuse, sourceless warmth, pressing close on all sides like copper rigging in slow suspension. Five micrometers to your left, a *Borrelia burgdorferi* spirochete slices through this matrix with mechanical precision: its eighteen-micrometer helical body gleams with a cold silver-blue iridescence, outer-membrane proteins dimpling the surface like hammered platinum foil, while periplasmic flagella — entirely enclosed within the cell's outer sheath — are just visible as shadow-dark internal cables, their coordinated rotation bending the whole cell into a traveling flat wave that drives it forward without a single exposed appendage contacting the surrounding gel. This is a profoundly low Reynolds number world, where inertia means nothing and viscosity is absolute — the moment that internal flagellar machinery would cease its rotation, motion would stop instantaneously, the amber medium clamping down like cooling syrup. Where the spirochete's tapered ends push through the polymer network, glycoprotein strands bow outward in a slow V-wake before reforming behind the cell, individual filaments catching new angles of the omnidirectional bioluminescent glow and flashing brief copper-gold highlights as the pathogen navigates, molecule by molecule, through the dense biological architecture of its host.

You are standing inside a living cell, suspended within a cathedral of stacked membrane sheets that curve away in every direction, their surfaces burning with a deep wine-red luminescence born of chlorophyll molecules embedded in the bilayer itself. Each membrane is encrusted with hemispheric phycobilisome antenna complexes — coral-orange and saffron domes crowding together like baroque ornaments — that harvest light energy and funnel it into the photosynthetic machinery below, while the narrow lumenal corridors between membrane sheets glow amber-gold with electrochemical gradients driving ATP synthesis. Drifting in the surrounding cytoplasm, icosahedral carboxysome shells hover like frosted lanterns, their pale faceted walls enclosing dense clusters of RuBisCO enzymes that fix carbon dioxide into organic matter, a chemistry that oxygenated Earth's atmosphere over two billion years ago and made complex life possible. The cytoplasm itself is not a clear fluid but a crowded amber gel — ribosomes, proteins, and nucleic acids packed so densely that molecules diffuse as if through warm resin, every cubic nanometer contested. At the cell's far boundary, a faint teal luminescence bleeds through the plasma membrane from the aquatic world outside, the entire scene self-lit from within, a photosynthetic engine no more than a few micrometers across that converts sunlight into the biochemical currency sustaining nearly every food web on the planet.
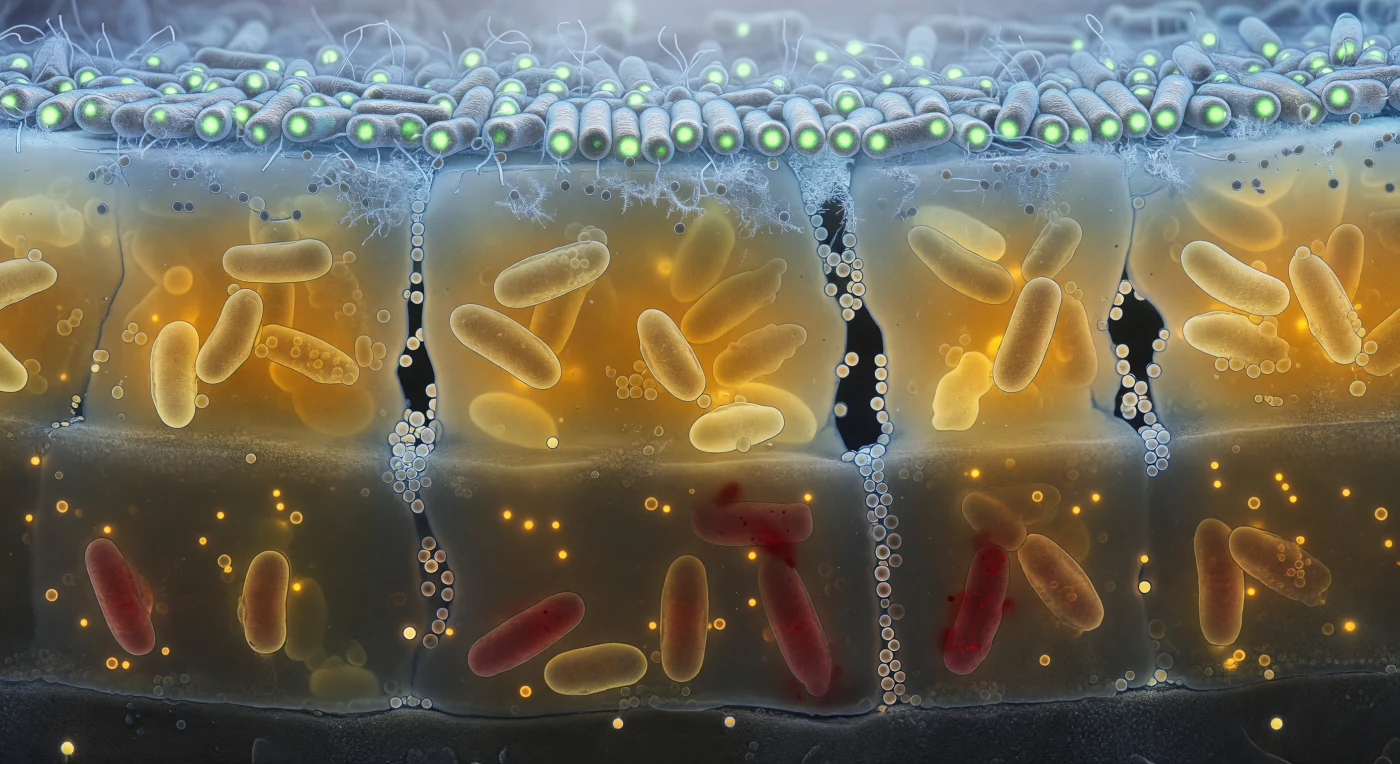
The viewer stands inside a living wall — a vertical face cut through a 50-micrometre-thick biofilm that reads like geological strata, each layer glowing with its own chemistry and gradual extinction of light. At the upper canopy, rod-shaped bacterial cells press tightly together in dense, ordered ranks, their lipid bilayers catching the cold blue luminescence of oxygenated fluid bleeding down from above, while GFP fluorescence turns the entire stratum into a field of cold green lanterns rimmed with frost-fine flagella and extracellular fibrils. Descending into the amber mid-zone, the blue light is rapidly absorbed by thickening extracellular polymeric substance — a translucent, honey-coloured resin of polysaccharides, proteins, and eDNA filling every interstitial space — while elongated, metabolically stressed cells cluster around dark, vertically oriented water channels whose walls are studded with membrane vesicles glittering like soap-bubble spheres: these channels are not incidental voids but engineered mass-transport conduits that sustain the colony's interior against diffusion limitations. Deeper still, in the hypoxic foundation, propidium iodide bleeds red through the compromised membranes of dead and dying cells whose outlines have blurred and swollen into the surrounding matrix, the oxygen gradient that structures this entire vertical world now collapsed to near zero, registered only as an absence of blue light and a cold darkness anchored against the rough mineral substrate below. The scene encodes, in living fluorescence, one of the fundamental truths of biofilm ecology: that spatial heterogeneity — of oxygen, of metabolic state, of cell viability — is not a failure of homeostasis but the colony's primary survival architecture.

You are suspended inside absolute darkness that is not empty — it is the reconstructed interior of a single dividing bacterium, rendered point by point from thousands of individual fluorophore detection events captured by Stochastic Optical Reconstruction Microscopy, a technique that circumvents the diffraction limit of light by localizing single molecules to ~20-nanometer precision. The warm golden arc curving across your horizon is the FtsZ contractile ring, a discontinuous polymer scaffold assembled from tubulin-like GTPase subunits that will physically constrict the cell at its equatorial waist, driving binary fission — its gaps are not artistic license but biological truth, reflecting the dynamic treadmilling of oligomeric patches rather than a continuous filament. Flanking you in cool cyan, the MreB actin homolog traces its helical cytoskeletal array, a scaffold that couples peptidoglycan synthesis to cell elongation and maintains the rod shape against turgor pressure exceeding several atmospheres. The membrane boundary glowing in dense red-orange above and below you is not a surface you could touch in any conventional sense — it is a 7-nanometer lipid bilayer crowded with porins, respiratory complexes, and chemoreceptor arrays, and the depth-coded thermal gradient shifting from amber warmth to cold violet across the cell's axial length encodes the genuine three-dimensional geometry recovered from thousands of axial focal planes, collapsing a living cell's molecular architecture into a constellation of hard-won photon events that together constitute, with extraordinary sparse precision, the spatial truth of a cell choosing to divide.
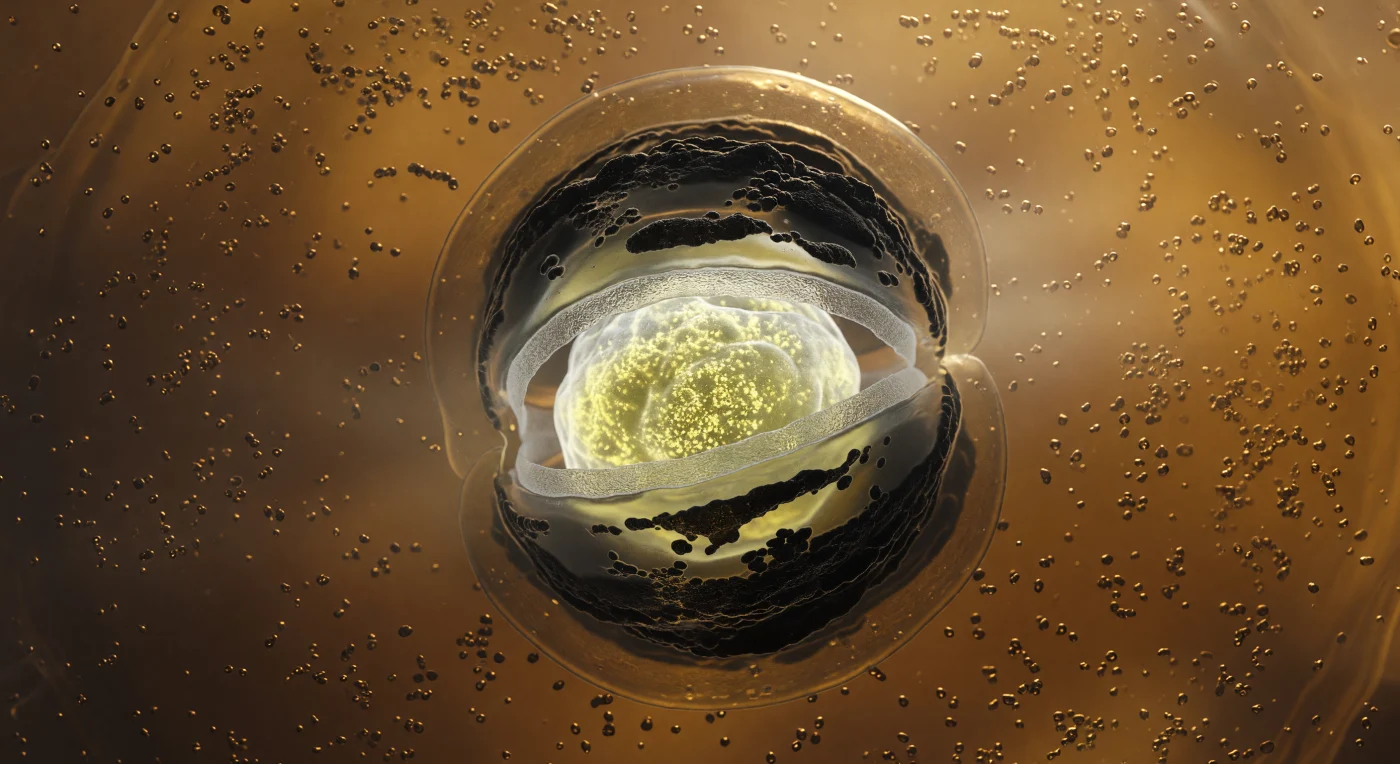
You are standing inside a living cell that is consuming itself to build an ark — the amber-gold cytoplasm surrounding you is so densely crowded with ribosomes and drifting protein granules that the medium itself feels more like warm gel than aqueous solution, every particle suspended in a low-Reynolds-number world where inertia means nothing and molecular friction governs all motion. At the center of this viscous cavern, the forespore hangs like a luminous moon: an ovoid compartment lit from within by the cold mineral glow of calcium-dipicolinic acid deposits and hyper-condensed DNA wrapped tight by small acid-soluble proteins, its interior reaching densities no normal cell ever achieves, the accumulated mass of a dormancy strategy refined across billions of years. Encircling that inner light, a pale gray peptidoglycan cortex scatters the glow diffusely outward like frosted glass, while beyond it, concentric sheets of SpoIVA and CotC coat proteins polymerize into dark, matte-black arcs that absorb light rather than reflect it — the structural armor of a spore that will outlast drought, radiation, and geological time. Most arresting is the engulfing membrane itself, two glistening lipid bilayers curving inward from the mother cell's own inner membrane in a tight phagocytic embrace, bent to their geometric limit around the forespore's equator, the periplasmic gap between them barely visible as a sliver of dark contrast against the warm amber glow — a molecular zipper closing, irreversibly, around a future.

Standing at eighty nanometers above the outer membrane of *E. coli*, you look up into the slow, silent descent of a T4 bacteriophage — its icosahedral capsid, roughly one hundred nanometers across, looms like a faceted asteroid eclipsing the upper field of view, its dark-gray polyhedral faces traced by faint gold seams where capsomere proteins lock together with crystallographic precision, the entire structure carrying the gravitational authority of a body that has crossed this threshold ten thousand times before. Below it, six jointed tail fibers splay radially across the teal-gold LPS terrain like the legs of some ancient, patient predator, their receptor-binding tips pressing into the lipopolysaccharide surface and creating shallow molecular dimples where glycan chains reorganize under localized mechanical stress. At the composition's center, the tail tube has already breached the membrane — a pore perhaps three nanometers across, its lipid rim slightly disordered, the bilayer's material character shifting from uniform teal to a warmer amber at the wound site — and through that needle-thin channel a luminous filament of single-stranded DNA streams downward into the periplasm, pale blue-white and opalescent, driven by the osmotic pressure differential between capsid interior and host cytoplasm. In the aqueous middle distance, two further phage particles hover in the diffuse blue-gray medium with tail sheaths still extended and gleaming cold silver, their approach as inevitable as gravity at any other scale — the surrounding fluid not a passive backdrop but a thermally restless, chemically saturated medium in which every surface attracts, repels, and negotiates across distances measured in bond lengths.
















