
A vast, desiccated terrain fills the field of view — the collapsed cellular architecture of a single moss leaf transformed by drought into a crinkled, honey-gold continent, its buckled cell walls casting long ochre shadows under a raking amber sidelight that makes the entire microscopic world feel ancient and scorched. At the center of this parched landscape rests the tun: a tardigrade fully committed to anhydrobiosis, its 200-µm body contracted into a dense barrel of concentrically folded cuticle, the chitin compressed into concentric rings like a dried prune, opaque rust-amber at the deepest creases and faintly glowing where the oblique light catches the outermost ridges. Beside it, a second individual is mid-contraction — posterior legs already retracted into dimpled sockets, flanks beginning to crease, the cuticle still marginally translucent where residual hydration has not yet been surrendered, the paired ocelli visible as faint rust-red pinpoints flanking a contracting cerebral ganglion. This transition represents one of biology's most extraordinary survival mechanisms: anhydrobiosis, during which the tardigrade expels nearly all free water, synthesizes vitrifying sugars and intrinsically disordered proteins, and suspends metabolism into a glass-like stasis capable of enduring decades of desiccation. Fungal spore beads drift slowly through the thick amber air column, and crystallized solute rings mark where the last water films evaporated, leaving these two animals — one already sealed, one in the final seconds of becoming — suspended in a silence that could last a century.

You hover just above the waxy surface of a moss leaf, your entire field of view taken up by a split-open tardigrade exuvia draped across the green terrain like the collapsed hull of a glass vessel — its translucent silver-grey walls catching the soft, jade-filtered light in iridescent interference colors that fade to near-invisibility at the thinnest edges. Inside the gaping cuticle, which has split along its dorsal seam during a recent molt, eight cream-yellow Macrobiotus eggs rest in a loose clutch on a thin amber biofilm of flattened bacteria, each sphere roughly the size of a boulder from your vantage point, their surfaces densely armored with hundreds of mushroom-shaped cuticular processes that scatter ambient light and give each egg a frosted, lantern-like glow. Deep within each shell, barely resolved through two layers of membrane, the earliest rounds of cell division have thickened the interior into a granular mass that transmits light with a warm amber core, evidence that embryogenesis is already underway inside these architecturally intricate, species-diagnostic capsules whose process ornamentation will eventually serve as taxonomic evidence of their identity. The surrounding moss leaf extends away in soft focus, its epidermal cells enormous hexagonal tiles of pale aquamarine, water pooling into meniscus lenses at their raised junction walls — a world where surface tension outweighs gravity, and time is measured in the slow accumulation of cell divisions within these quietly glowing spheres.

You stand enclosed within a colonnade of *Bryum argenteum* stems rising like jade pillars into a canopy so layered and deep it dissolves into luminous green infinity above — each cylindrical column perhaps ten body-lengths across, its surface resolved into overlapping hexagonal cells whose walls glow translucent emerald under diffuse daylight filtered through stacked leaf after leaf, arriving not as sunlight but as something closer to an underwater glow, ancient and saturated. Between the stems, menisci of surface water hang taut and mercury-bright, curved like optical lenses — at this scale, surface tension dominates gravity entirely, and these water bridges are not droplets but walls, refracting the ambient light into pale prismatic arcs where they contact the cuticle of the stems. Across the rhizoid substrate below, pale threads 2–3 microns thick drape and spiral like anchor ropes of frosted glass, rooted into mineral grains that loom as boulders, while diatom frustules scatter across the sediment as monuments of silicate geometry, their striated pores catching light in cold amber and silver. You move through this space on eight stubby lobopodial legs, clawed pads gripping each rhizoid fiber with deliberate precision, your amber cuticle glowing faintly translucent in the green-shifted light — within you, the pharyngeal bulb is visible as a ghostly interior sphere, a muscular pump evolved to pierce and drain individual plant cells, the fundamental feeding apparatus of an animal that has survived five mass extinctions by retreating, when the world fails, into cryptobiotic stillness.

You are suspended just beneath the vaulted underside of a diatom frustule, its parallel silica ribs and hexagonal pore arrays refracting ambient water-light into slow prismatic ribbons of gold, amber, and pale aquamarine that ripple across every wet surface — a cathedral of biosilica geometry stretching across the entire upper field of view, translucent as sea-glass, thick as a structural wall. Bearing down from directly above, the circular mouth of a tardigrade presses seal-tight against the ornate frustule lacework, its pale lip ring forming a perfect gasket while two paired amber stylet lancets — honey-gold, slightly curved, each no thicker than a single frustule rib — have already punched through the silica with hairline precision, micro-fractures spidering outward from each entry point in starburst halos of violet, copper, and electric blue interference color. The stylets are hollow piercing tools, evolutionary solutions to the engineering problem of breaching one of biology's most mechanically resilient natural materials — hydrated amorphous silica — allowing the pharyngeal bulb above, a dark garnet sphere pulsing with muscular rhythm, to generate the suction pressure needed to extract cell contents through the breach. Around the contact zone, the shallow water film catches every shaft of oblique light in curved silver menisci along the silica ridges, bacterial rods drifting in slow Brownian suspension at the periphery, the whole scene a pressurized, crystalline world where predation and architecture are rendered at the same intimate scale.

Before you, a slow catastrophe unfolds in absolute silence: the advancing front of an ice crystal sweeps across the moss substrate like a wall of optical glass, its hexagonal geometry precise and merciless, each crystallographic face terraced at the molecular scale where water molecules are surrendering one by one to the lattice. You are pressed into the shelter of a bryophyte stem base, your amber-translucent barrel body no larger than a grain of pollen, your legs already withdrawn, your cuticle folding inward in tight wrinkled pleats as cryptobiosis takes hold — the tun state, a sealed capsule of suspended biochemistry riding out the thermodynamic siege. The thinning water film ahead of the ice front trembles as a last meniscus ocean, its surface tension geometries taut against the moss cell walls, before the crystal claims it entirely. Other tuns cluster nearby in the corrugated hollows of compressed cellulose and biofilm, some already embedded within the lattice — their amber warmth glowing through clear ice like embers sealed in glass, organic heat preserved as pure color against the achromatic crystalline architecture swallowing everything around them. The diffuse blue-white Antarctic overcast filters down through a millimeter of ice above, casting the entire scene in a luminous cyanotype palette where depth is measured in micrometers yet recedes with the visual weight of something planetary.

You are suspended at the threshold of a cathedral passage between two immense quartz boulders, your eight stubby legs gripping crystal faces through a meniscus skin of capillary water, surface tension the dominant force holding you to the mineral world rather than any meaningful pull of gravity. The quartz walls rise around you like cliffs of frosted obsidian and pale smoky glass, their conchoidal fracture faces catching diffuse transmitted light from above and refracting it into cold blue-white shards and amber internal glows that pulse through ancient mineral inclusions deep within the crystal matrix. Overhead, two fungal hyphae span the passage like translucent rope bridges of ivory-gold, their cytoplasm a faint granular shadow within glassy tubular walls, while beyond them a dense plug of dark humus seals the far tunnel in near-absolute blackness, its surface a crumbled labyrinth of collapsed organic matter and tannin-stained fragments. Clay platelets jut from the quartz base in laminated ochre shelves, bacterial biofilms glinting as smooth reflective patches against matte mineral texture, and at every water contact point curved menisci catch the light as bright crescent arcs — the physics of adhesion and viscous drag ruling this world entirely. What reads subjectively as an immense cave system of staggering internal complexity is, in physical reality, a soil pore space measured in tens to hundreds of microns, close and vast at once, a universe compressed into the dark architecture between grains.
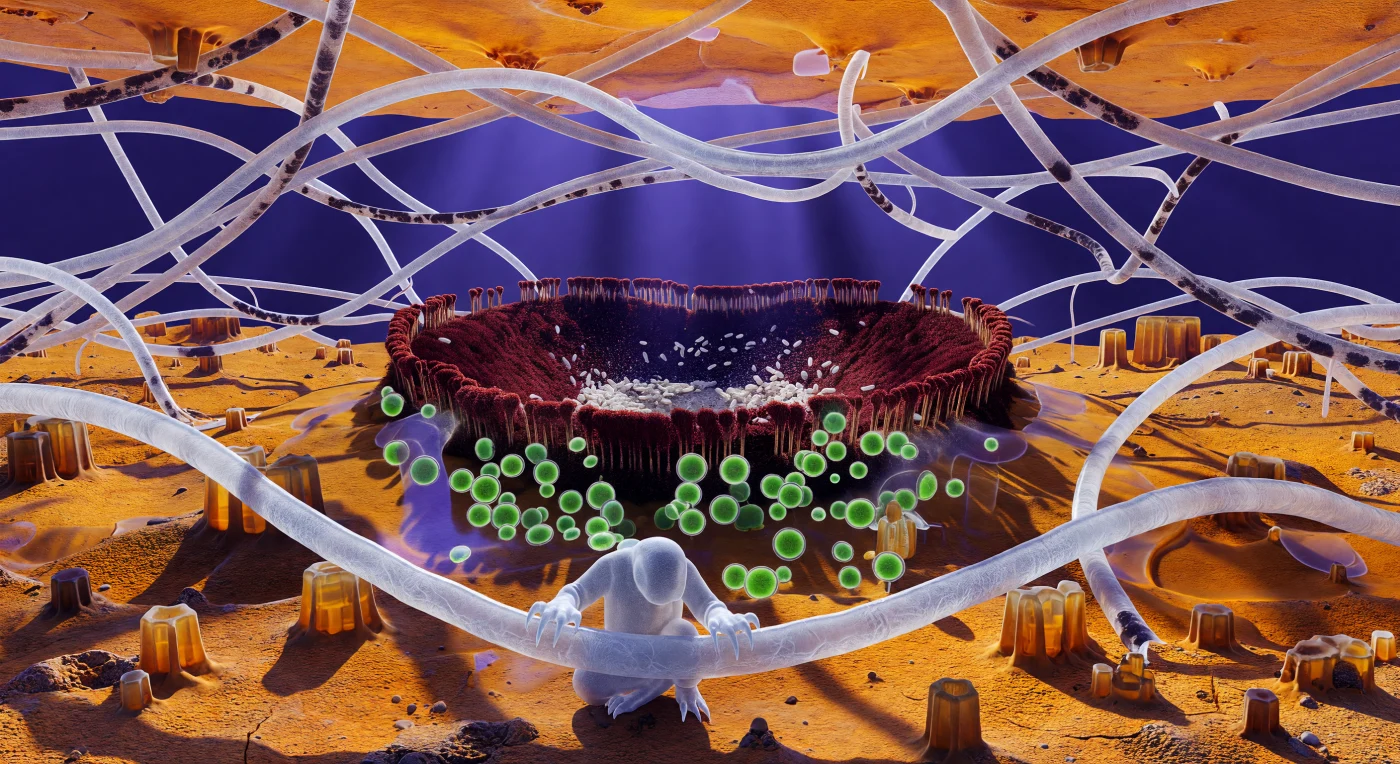
The ground beneath you is not ground — it is a living plateau of compressed fungal and algal tissue, stretching orange and amber in every direction like a sun-scorched badlands, its surface rising into polygonal ridges of packed cortical cells that cast long shadows under alpine ultraviolet arriving with almost physical weight. You cling to a hyphal strand as thick as a ship's rope, its translucent walls revealing slow drifts of cytoplasmic granules moving in the dim interior like silt suspended in still water, the elastic surface yielding fractionally under each curved claw before springing back. This is the living architecture of a Xanthoria parietina thallus — a composite organism in which fungal hyphae provide structural scaffolding and water retention while Trebouxia algal cells, clustered ahead like clusters of luminous green lanterns ten to fifteen micrometres across, drive photosynthesis deep within the cortex, their chloroplasts saturated with harvested light. The thin liquid film coating every surface here obeys surface tension more than gravity, drawing curved meniscus walls between adjacent hyphae that act as natural lenses, bending and amplifying the amber world behind them, while a tardigrade — an animal of perhaps three hundred micrometres, gripping with hooked claws and moving with the slow deliberateness of something for whom viscous drag outweighs inertia — navigates this architecture as a sailor moves through rigging. In the middle distance, the apothecium opens like a caldera, its rust-red walls dense with paraphyses and its interior choked with ivory ascospores tumbling in thermal microcurrents, the entire reproductive apparatus of the fungus exposed and operating at a scale where dust is architecture and stillness is never absolute.

Looking upward from the floor of this shallow interstitial world, the most commanding presence overhead is the water-air interface itself — an immense, gently bowed ceiling of mercury-silver perfection spanning the full upper hemisphere, its surface tension holding a boundary that, at this scale, feels as structural and imposing as vaulted stone. This meniscus is sustained by cohesive forces between water molecules that here dominate over gravity almost entirely, and where it pins against the cellulose walls of adjacent moss cells it climbs in flawless capillary arcs, trembling with an energy density of roughly 72 millinewtons per meter — a force that at human scale would be imperceptible, but here is sufficient to support, trap, or crush a microorganism. Caustic interference patterns — bright filaments born from refraction through the curved interface — sweep slowly across the amber-green mosaic of polygonal moss cells below, pooling into zones of intense illumination and deep shadow that give the flat substrate an unexpected, almost geological drama. At mid-column, a tardigrade drifts in arrested stillness, its translucent gold-amber body bisected by the depth gradient of the film, eight lobopodial legs loosely splayed, its dorsal cuticle catching the cold mirror-light from above while its ventral surface glows with warm transmitted light rising from the biofilm floor — a creature caught between two worlds, simultaneously bathed in silver and gold.

You hover suspended in warm saline water between two mineral masses that dwarf you completely — a rose-quartz boulder whose polished surface curves away like a planetary horizon, and a milky feldspar wall threaded with translucent grey veins, both so close that the viscous resistance of water pressing from every direction is a palpable physical presence. Ocean light has filtered down through centimetres of saturated sand to reach this interstitial chamber, arriving as a diffuse turquoise glow that renders the water itself a luminous blue-green medium, thick with drifting particulate and punctuated by amber biofilm mats where colonies of bacteria form rounded hillocks across the grain surfaces, their warm honey tones catching and redirecting the cool mineral light. Pressed flat against the nearest quartz face, a Batillipes heterotardigrade navigates the biofilm margin with deliberate, adhesive certainty — its dorsal cuticle divided into overlapping sclerotized plates like tectonic shields, its eight translucent disc-feet slightly deformed where they grip the grain surface, spreading visibly through the water-thin contact zone in a suction-pad intimacy that is the dominant logic of movement at this scale, where gravity is irrelevant and adhesion is everything. Pennate diatoms rest against the quartz like elongated stained-glass monuments, their silica frustules catching thin lines of specular silver, while beyond them the interstitial passage opens into deeper channels between receding grain silhouettes, a pale amber polychaete bristle — enormous here, a cable crossing the mid-ground — fading into the softly turbid oceanic blue where light diminishes and the boundaries between grain, water, and sediment dissolve into a single warm, living infinity.

You are standing inside a cyanobacterial mat, eye-level with the extracellular polymeric substance that forms amber towers and channeled ridges around you, each gelatinous column backlit by warm transmitted light until it glows like honey-stained glass. Dense bundles of Oscillatoria trichomes rope across open capillary channels, their blue-green mass occasionally erupting in flares of brick-red autofluorescence where chlorophyll-packed sheaths catch the light, while golden-brown diatom frustules jut from the EPS at oblique angles, their silica geometry refracting pale prismatic halos at every edge. This entire landscape — canyon walls, rope bridges, embedded monuments — is measured in microns, a world where surface tension forms curved walls of glass-like transparency and gravity matters far less than capillary adhesion. Into the foreground of this biofilm prairie, a tardigrade drives its anterior bluntly downward, buccal apparatus pressed against a filament bundle, stylet lancets working to pierce cyanobacterial cells while the pharyngeal bulb pulses visibly through the semi-translucent cuticle; the front legs are splayed and clawed deep into the EPS for purchase, the body arched in a taut muscular bow, rear legs dragging slow furrows through the gel — a creature of perhaps 400 microns commanding the entire scene with the unhurried, deliberate force of something that has survived five mass extinctions.

At the center of everything, a body is being born from itself — a tardigrade halfway extracted from the split anterior seam of its own shed cuticle, caught at the precise moment when two versions of the same animal coexist in the same frame. The discarded exuvia is the more immediately astonishing object: a complete hollow effigy, translucent as crinkled glass, its former claw sockets, leg tubes, and dorsal plate impressions all preserved in pale relief as though the cuticle were a mold made from an absence — this is ecdysis, the periodic molt that all ecdysozoans must perform to grow, the old trilaminar chitin-protein exoskeleton split and evacuated while the new cuticle beneath completes its sclerotization over the hours following emergence. The emerging animal reads differently from its ghost: its surface carries a faint warm amber iridescence where cross-linking of the new cuticle matrix has begun but not finished, the forward claws still pearl-soft stubs of unsclerotized cuticle that will harden over the coming hours into the precisely hooked tools used for gripping bryophyte cell walls, the hind legs still partially sleeved within the old leg sheaths so that limb and vacated lumen form a doubled image. Beneath them both, the moss leaf cell surface extends to every horizon — each hexagonal cell wall rising two or three body-lengths as a translucent green-amber rampart packed with chloroplast constellations, the water films pooled at cell junctions pulled into menisci that act as curved mirrors, and thin fungal hyphae crossing the mid-distance like slack rope bridges into a green-gold haze where the world quietly dissolves.

In the cold, cathedral stillness of a thin water film clinging to wet moss, two organisms the length of a dust mote are locked in a moment that will decide one of them. The luminous, lantern-bodied rotifer on the right — its ciliary corona mid-beat, its internal ovaries glowing jade through a transparent body wall — belongs to the phylum Rotifera, filter-feeders so ancient and so successful that bdelloid lineages abandoned sexual reproduction entirely, yet here the strategy of passive gathering meets its limit: *Milnesium tardigradum*, the apex predator of this microscopic ecosystem, presses its fully gaped circular mouth and extended stylet lancets — each one a biological hypodermic 15 to 50 micrometres long, driven by a muscular pharyngeal bulb lined with chitinous placoids — hard against the rotifer's pellicle, dimpling it inward in a crescent of compressed, intensified light. At this scale, the surrounding water is not empty space but a medium of genuine physical consequence: surface tension curves the capillary menisci at the frame's edges into slow glass walls, dissolved organics bend transmitted light in barely perceptible refractive waves, and viscous drag makes every movement a negotiation with the fluid itself rather than a passage through it. What the eye reads as a confrontation between two creatures is also a collision between two ecological strategies — one built on continuous filtration and near-eternal dormancy potential, the other on stylet-driven predation and the pitiless hydraulics of a pharyngeal pump that will, in moments, breach that glowing wall and begin to drink.

You grip the outer wall of a Spirogyra filament whose diameter dwarfs you thirty times over — a flawless crystalline cylinder curving away like the hull of a glass submarine, your curved claws finding just enough purchase on the faintly ridged cuticle as pond-surface light descends in broad diffused shafts from somewhere far above. Through the cell wall, the helical chloroplast ribbon spirals upward in continuous grass-green turns, its pyrenoid nodes catching the downwelling light as bright white-gold beads strung along the helix, the whole structure glowing as though generating its own illumination from within. Beyond the filament, the open water is a deep layered blue-green — a genuine liquid atmosphere whose viscous resistance registers against your legs whenever you shift your grip, because at this scale the pond is an ocean and you are pressed against a glass cliff by the gentlest of currents, while a symmetrical desmid hangs in mid-water like a small luminous building, its reticulate chloroplasts pulsing with photosynthetic emerald. Then, from the right, a paramecium sweeps through — the scale of a cargo vessel seen from a dock — its cilia-covered flanks shimmering silver-grey in the downwelling light, food vacuoles visible as dark inclusions through the barely-translucent pellicle, passing without acknowledgment and trailing a vortex that tugs lightly at your hold on the wall. The Spirogyra filament continues above and below into soft atmospheric distance, its spiral staircase of chlorophyll threading through still green water toward a surface neither of you will ever reach.

You float at the surface of something that reads, at this scale, exactly like a world — a barrel-shaped moon of amber and sienna, its entire surface an undulating topography of compressed cuticle ridges and valleys, each fold a record of the living animal that once inhabited this contracted shell. This is a tardigrade tun: a state of cryptobiosis in which the animal has retracted its legs, expelled nearly all its water, and reorganized its cellular chemistry around trehalose glass, suspending metabolism so completely that the boundary between dormancy and death becomes philosophically ambiguous. Unfiltered solar radiation — the full spectrum with no atmosphere to edit it — strikes the near hemisphere with surgical precision, illuminating the interlocking polygonal cuticular plates along their crests in warm ochre-gold while the troughs plunge into lightless black, the raking angle transforming the surface into a landscape of simultaneous fire and void that no earthly environment, with its scattering air and diffuse shadows, could ever produce. The terminator line dividing sunlit amber from absolute dark is sharp as a material edge, because in vacuum there is no limb-glow, no gradient, nothing to negotiate the transition between a small, intricate biological structure holding its molecular architecture intact and the indifferent interstellar dark surrounding it in every direction. What makes this scene extraordinary is the equation it makes visible: a few hundred micrometers of compressed cuticle and vitrified cytoplasm, resting on a machined metal substrate somewhere in a laboratory, containing within its wrinkled geometry the complete suspended blueprint of an animal that has outlasted conditions that would reduce most life to chemistry.

A cold directional beam falls from somewhere impossibly far away — a cave entrance reduced to a pale bluish smear on the horizon — barely grazing the translucent celadon canopies of cave moss leaves overhead, their chloroplast-sparse cells transmitting more light than they hold, each marginal edge glowing like frosted glass. Calcium carbonate crystallites encrusting the moss stems flash brief hard white sparks where a single facet catches the beam at the right angle, then vanish back into grey stillness, while a thin tensioned water film coats every surface — limestone grain, leaf junction, hypha — unifying the entire subterranean landscape beneath curved slivers of specular light. The limestone floor extends outward like a weathered plateau, each calcite grain reading as a cliff face of striated buff and grey with shadow pools beneath every overhang where no light reaches at all, fungal hyphae crossing the middle distance as faintly luminous rope bridges, their walls thin enough to show granular internal structure suspended in the stillness. Emerging from deep shadow into the faint edge of the beam, a pale tardigrade resolves from the darkness, its cuticle reduced to near-translucency by cave life — vestigial eye-spots barely distinguishable from surrounding tissue — the faint shadows of midgut and body-wall musculature bleeding through the integument as the animal moves by touch alone, anterior low, curved claws finding purchase on calcite microcrystals, its glistening surface indistinguishable in texture and reflectivity from the wet crystalline world it reads through direct contact.

At moss-surface level, the viewer witnesses something that registers less as a biological event than as a geological one: a cream-yellow ovoid the size of a small boulder has cracked along its equatorial seam, and through the widening fracture an 80-micron first-instar Macrobiotus is pushing free, its amber-translucent body already fully articulated, eight stubby legs pressing against the shell edge with unmistakable intention. The egg belongs to a genus known for its elaborately ornamented shells — the conical mushroom-shaped processes studding its surface are species-diagnostic structures, each one a chitin-protein pillar tipped in warm amber-brown, arranged in radial ranks across a shell with the subsurface translucency of fine porcelain, their soft shadows mapping a landscape of extraordinary relief across the cream surface. The hatchling's interior is nearly as transparent as the shell it is abandoning: the midgut appears as a colorless loop, the hemocoel a fluid-filled cavity, nascent muscle bands barely resolved through the cuticle — all of it secondary to the two red-orange eye spots flanking the anterior brain mass, the only truly opaque pigment in the scene, glowing like embers in an otherwise diaphanous body. Behind the emerging animal, two sibling eggs rest against the moss leaf surface, backlit by chloroplast-packed cells glowing in cool jade-green, their identical process-studded forms slightly softened by the few hundred micrometers of depth that constitute the entire world visible here. The whole scene exists within what would be a single dewdrop at human scale, the water film betraying itself only as a faint meniscus gleam where egg meets leaf — gravity irrelevant, surface tension sovereign, and a new animal already fully alive inside a world most eyes will never find.
















