
You press forward through a winding corridor of ancient mineral architecture, its irregular walls built from stacked silver-grey smectite platelets whose razor-thin edges catch some sourceless diffracted glow and throw faint pewter and lilac iridescence across the pore ceiling, while massive translucent quartz boulders — each a looming crystalline monolith relative to your body — refract that dim luminescence into prismatic flecks across the floor below you, every surface draped in pooling amber-ochre organic films that resemble cooled resin, glossy and rippled where successive water menisci have dried and retreated. Dominating the center of the corridor ahead, curving gently away into the middle distance, is the hypha: a smooth glassy cylinder occupying roughly a quarter of the passageway, its nearly transparent wall carrying a faint green-refractive sheen through which you can just discern the slow interior drift of cream-white lipid globules and amber-grey granules suspended in faintly luminous cytoplasm, the whole structure immaculate and taut against the rough mineral chaos surrounding it, its outer surface bridged to adjacent clay platelets by curved water menisci that arc and concentrate what little light exists into bright liquid threads. Along the pore walls, tucked into sheltered angles between clay stacks and half-buried in biofilm patches, colonies of bacterial rods cluster in loosely ordered mats — each individual barely one-fifth your own diameter, their grey-beige forms pressing against the mineral substrate like a low fog of tiny boulders — while three branching side-tunnels open from the far wall and floor, their entrances framed by jutting clay platelets and dark organic crusts, their interiors receding into absolute lightless black, the kind of darkness that exists only where photons have never reached and all navigation is chemical, all sensation dissolved in the faint electrical hum of living soil.
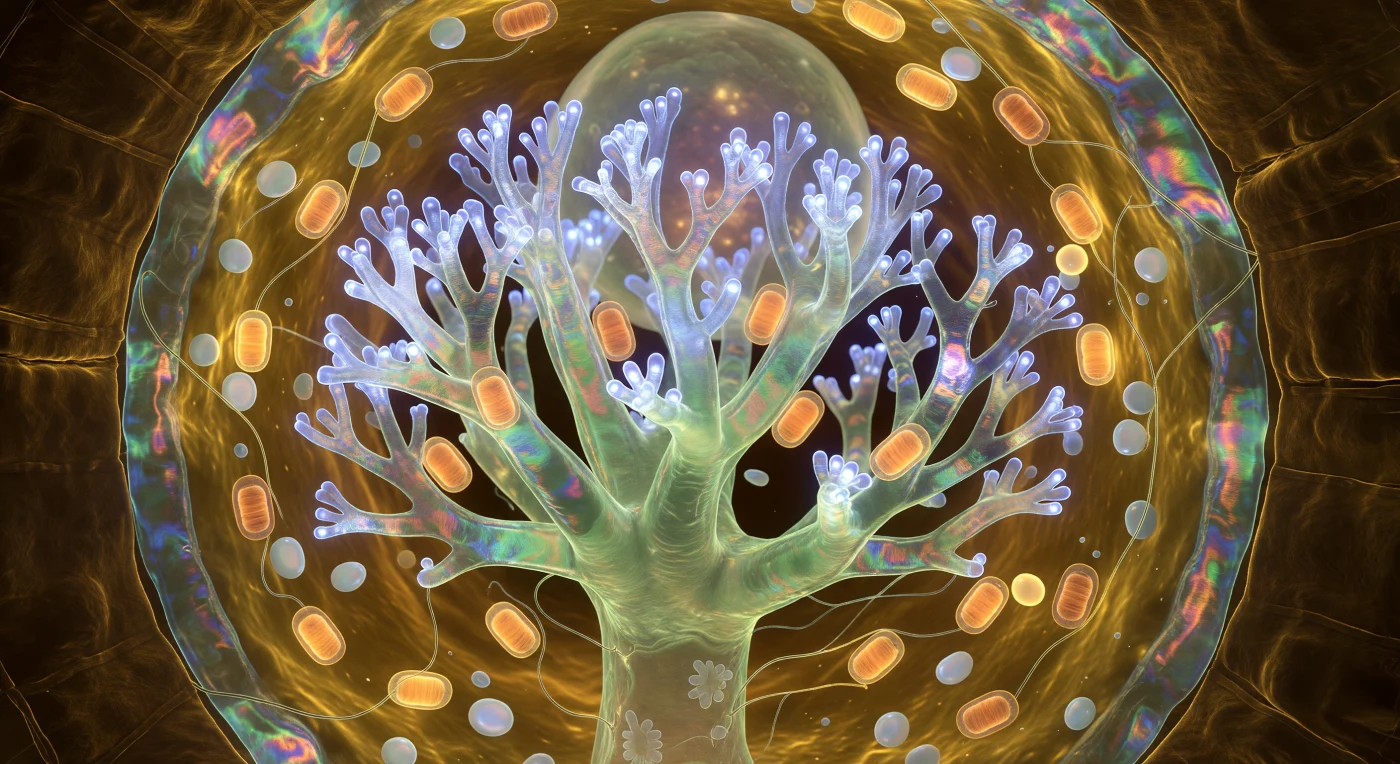
You float inside a living cell whose interior has become a cathedral of cold metabolic light, the arbuscule rising before you as an inverted coral forest of translucent jade-ivory, branching upward through recursive dichotomies until the terminal branchlets — each barely half a micron across — dissolve into a trembling haze of electric cobalt-white as phosphate transporter proteins cycle through their conformational states in massed, shoulder-to-shoulder unison across every square nanometer of the surrounding periarbuscular membrane. This double-membrane interface is the operational heart of the arbuscular mycorrhizal symbiosis: the fungal cell and the host root cortical cell never actually touch, remaining always separated by this taut amber-to-teal-iridescent film through which phosphate ions move outward into the plant and photosynthate flows inward to the fungus, an exchange so relentless that its energetic cost draws clusters of cigar-shaped mitochondria against every major branch like amber lanterns, their chemiosmotic respiration casting halos of warm orange-gold into the surrounding viscoelastic cytoplasm. Beyond the mitochondria, pearlescent amyloplasts drift through that golden cytoplasmic gel like opalescent river stones, refracting the bioluminescent blue into pale prismatic smears, while the cell wall curves at every horizon as a massive dark amber rampart — its crossed helical arrays of cellulose microfibrils catching the arbuscular glow at low angles and revealing a carved, vaulted topography. There is no external light source here; every photon you perceive is metabolic, emitted by the chemistry of an exchange that has been sustaining forests since before the first trees existed, and the entire glowing architecture around you — this single cell, this single arbuscule — is one node in a network that may span hectares of living soil above.

Stretching before you like a dim desert plateau, the sandy soil cross-section opens into a vast mineral plain where three Glomus spores dominate the foreground like lacquered boulders—their concentric amber and honey-brown laminae catching diffuse overhead light with a rich, ceramics-like chiaroscuro, each wall layer a distinct chapter in a months-long developmental archive of fungal lipid reserves. Nearby, a cream-rose Gigaspora spore rises with barnacle-textured ornamentation and a faint inner luminescence, while a wine-red Scutellospora displays its rigid crescent germination shield, that curved maroon plate evolved specifically to orient the emerging germ tube through the soil column. Between them, quartz grains and rose feldspar crystals tower as translucent, caustic-refracting cliffs, and dark amber flakes of decomposed organic matter pave the sandy floor like slabs of petrified resin—the phosphorus, nitrogen, and carbon locked within them precisely the currency these spores exist to mobilize. Spanning the open gaps in shallow catenary curves, gossamer silver hyphae catch the diffuse light only as near-invisible bright lines, yet these ghost-smooth filaments are the operational core of the entire system: their walls selectively shuttle phosphate ions toward root surfaces while carbon flows the other direction, an exchange ancient enough to have enabled the first land plants to colonize bare mineral substrate some 450 million years ago. The scene reads as geological stillness, but each spore is a compressed biochemical potential waiting for the precise rhizosphere signal—root exudate chemistry, moisture, temperature—that will trigger germination and extend this delicate, nearly invisible architecture outward through the permanent dark.
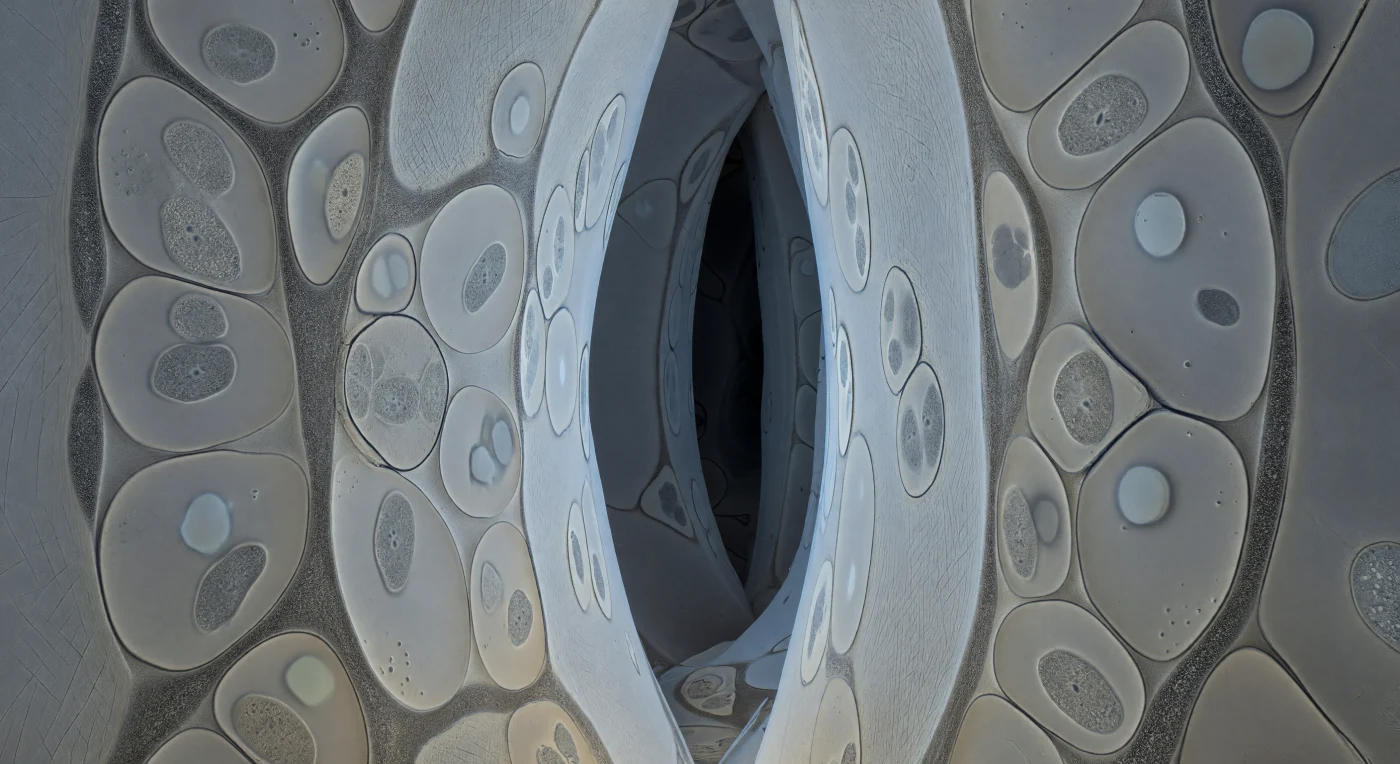
You are deep inside the root — not metaphorically but structurally, wedged into an intercellular corridor so compressed that the pale cream cellulose walls of adjacent epidermal cells press against you from every direction, their microfibril bundles running in crossed diagonals like the grain of ancient paper, cold and faintly translucent where metabolic chemistry leaks through the membranes behind them. The corridor is packed with fungal hyphae flattened into lens-shaped profiles by the same cellular pressure that shaped this passage, each one bounded by a razor-crisp graphite plasma membrane holding its curvature with palpable tension, enclosing dove-grey cytoplasm scattered with ovoid mitochondria and small lucent vacuoles that read as soft optical absences rather than resolved structures. Between every fungal wall and every root cell wall runs the interfacial matrix — a continuous 15–25 nm granular strip of slightly warmer mid-grey, heterogeneous in density, a glycoprotein meshwork secreted simultaneously from both sides, and it is here, in this near-invisible threshold, that phosphate ions leave fungal cytoplasm and photosynthetic sugars arrive from the plant, the entire metabolic contract of ectomycorrhizal symbiosis conducted across a boundary thinner than a ribosome is wide. Deeper corridors recede into flat planes of dark slate, losing fibrillar detail, the labyrinth extending beyond perception in every direction through lightless soil, navigated entirely by chemical gradient and membrane tension, without a photon to spare.
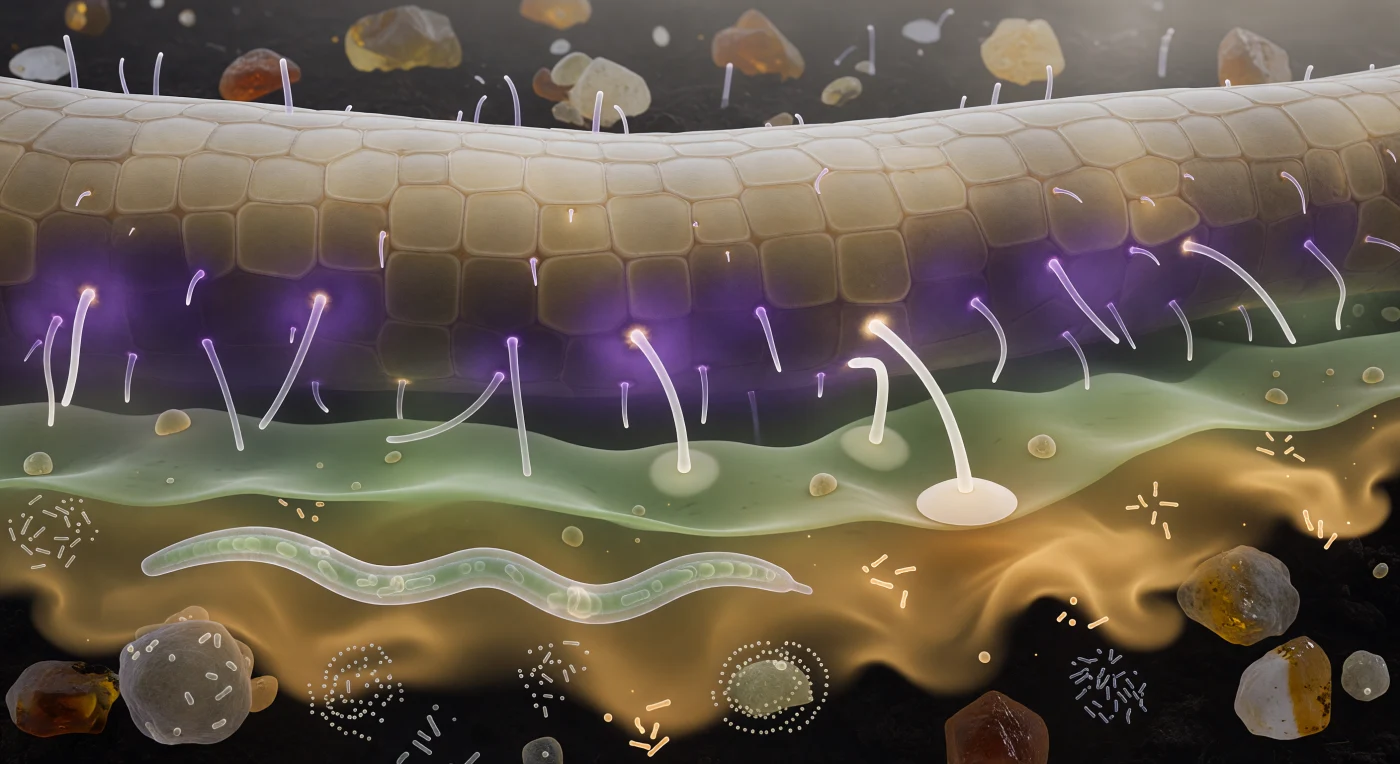
You are suspended just beyond arm's reach from a root wall that curves away like a pale sandstone escarpment, its epidermis a mosaic of subtly convex tan cells whose raised borders trace the geometry of ancient masonry, while glass-clear root hairs extend toward you as cold luminous filaments that refract the chemistry between you rather than any light source above. What you are seeing—the layered violet corona of strigolactones and flavonoids pressed against the epidermis, the cooler green-gray amino-acid haze diffusing outward through pore-water films, the slow amber-gold sugar plume dissolving in wisps toward the darkness behind you—is the rhizosphere's signaling landscape made visible, a chemical atmosphere that root and fungus have co-authored over 400 million years of coevolution to choreograph their encounter. Two hyphal tips bend measurably through the green layer, following the violet gradient as ships follow a coast, and one has already flattened its terminus into an appressorium disc flush against an epidermal tile, beginning the enzymatic negotiation that will, if the plant's immune system permits, thread the fungus into the cortex and establish a phosphate-for-carbon exchange worth more to both partners than anything visible at this moment. Below, a nematode crosses the amber fade in glassy sinusoidal progress while bacterial swarms pulse in loose spirals on aggregate faces around it, each rod or coccus barely a fifth the diameter of the hyphal walls overhead—a reminder that what looks like a bilateral drama between root and fungus is in fact an entire biome transacting simultaneously in the same few hundred micrometers of chemically saturated darkness.

You stand on a living floor of interlocking fungal cells woven as tightly as hand-laid mosaic tile, each brick-shaped element five to fifteen micrometers across, their cream-white walls edged with faint sulphur-yellow where adjacent membranes press together, the whole surface carrying a cold pearlescent luminosity that reveals every seam and ridge in shallow relief like moonlit cobblestones. Behind you, the root column of a *Suillus*-colonized pine feeder root rises as an immense curved wall of tan-brown tissue, its surface striated with longitudinal cell boundaries, so vast in its curvature that it dissolves into haze before bending fully out of sight — and at the zone where mantle meets root epidermis, a subtle amber glow signals the Hartig net beginning its intercellular labyrinth within, visible only as lamplight through a paper screen. This plectenchymatous mantle is no passive sheath but an active interface: a tightly regulated symbiotic tissue through which photosynthetic carbon moves inward from tree to fungus while phosphorus, nitrogen, and water move outward from soil to root, all mediated by transporter proteins embedded in membranes pressed in molecular intimacy. At the fabric's fringe edge, the weave unravels into individual extraradical hyphae, each a single-cell-wide transparent tube three to six micrometers across, streaming outward into the absolute darkness of uncolonized soil pore space like fiber-optic filaments launched into a void, their glassy refractive walls and faint internal cytoplasmic movement the only light in a lightless world they are actively, incrementally, chemosensorily mapping.
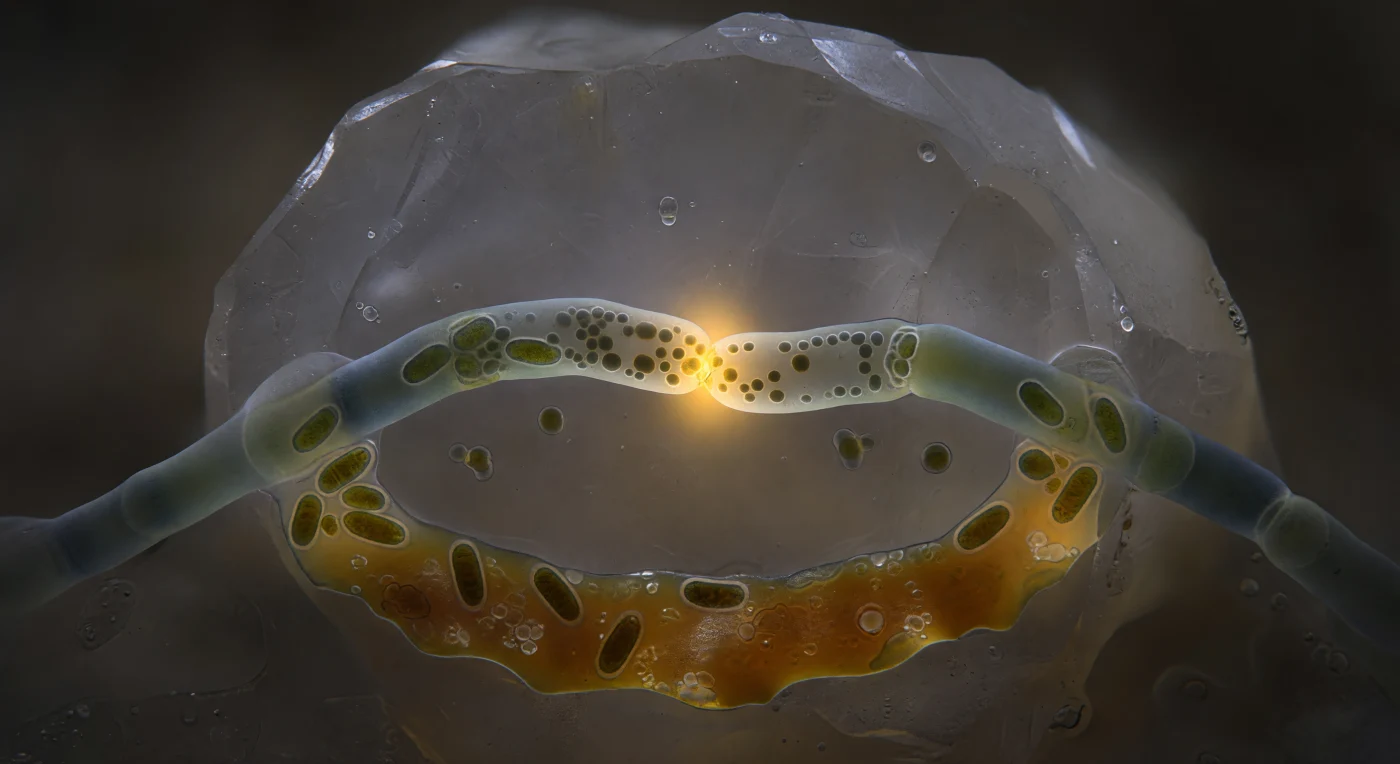
You are suspended in absolute darkness inside a soil pore no wider than a human hair's cross-section, pressed against the face of a feldspar grain that rises beside you like a glaciated cliff, its crystal planes emitting a cold diffuse gleam from within while a saturated film of soil water holds you in place. Before you, two arbuscular mycorrhizal fungal hyphae arc toward each other through this mineral-walled chamber, each tube five to six micrometres in diameter, their chitin-glucan walls the translucent blue-green of clouded sea glass — and at the single point where their walls have dissolved into one another, a fusion pore barely one and a half micrometres across blazes in concentrated amber-gold, the only warm light in this otherwise lightless world. This is hyphal anastomosis: the deliberate erasure of a boundary between two fungal individuals, a process that allows cytoplasm, organelles, nuclei, and signaling molecules to flow between formerly separate hyphal branches, enabling genetic exchange, resource redistribution, and network repair across the mycelium. Through the luminous throat of the pore, mitochondria cluster in dense amber-copper masses at the margin while lipid granules and cytoplasmic ground substance stream in a viscous rope from left to right, the metabolic traffic so intense it generates a visible thermal halo that scatters through the surrounding water film into the grey-brown obscurity of the soil pore — a pulse of biochemical energy intimate in scale yet foundational to the carbon and phosphorus economies of entire forest ecosystems.

Filling the frame like the face of a dark volcanic moon, a single soil macroaggregate of compressed humus, fungal melanin, and silicate minerals forms a world entire unto itself—its espresso-black surface mottled with ochre and grey, the crystalline faces of quartz and feldspar grains catching raking amber light like pale boulders half-submerged in a dark sea. Across every exposed face, a thin lacquer of glomalin—a glycoprotein secreted by arbuscular mycorrhizal fungi and one of the largest repositories of stable carbon in terrestrial ecosystems—seals the aggregate in a hydrophobic amber-gold film, a single water droplet beading against it with a steep contact angle that speaks to the structural chemistry locked into this three-millimeter body. From stress fractures splitting the aggregate's equatorial belt, white hyphal threads emerge in loose parallel arcs, biological sutures that actively bind mineral and organic particles together through both physical enmeshment and the continued secretion of that same glomalin cement. In the ultraviolet inset, the ambient world drops to near-darkness and the glomalin coating erupts in brilliant yellow-green fluorescence, tracing the invisible architecture of carbon sequestration in incandescent relief—making visible a soil engineering project that arbuscular mycorrhizal fungi have been executing, largely unchanged, for four hundred million years.
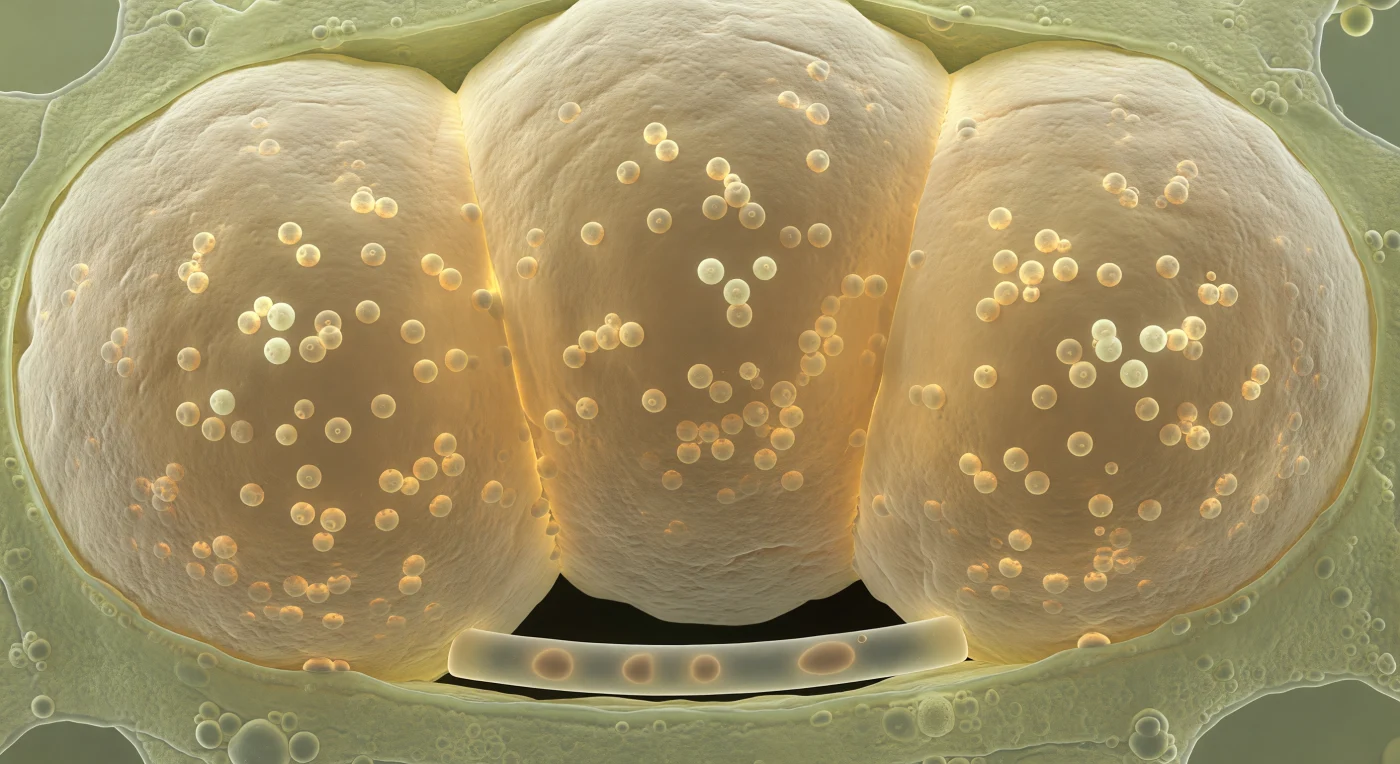
You are suspended inside a root cortical cell whose entire volume has been commandeered by three arbuscular mycorrhizal fungal vesicles, pressing against each other and the surrounding walls like amber hot-air balloons inflated well past capacity, their mutual contact zones flattened into broad planes of soft geometry wherever they collide. Each vesicle wall presents itself in layers — a rough, striated outer rind of chitin-glucan lamellae the texture of weathered ivory giving way inward to a glassy amber smoothness that seems to generate its own dim luminescence, as though the chemistry of lipid accumulation is itself warm enough to glow. Inside each amber mass, dozens of spherical lipid globules ranging from five to ten micrometers across hang in arrested suspension, refracting ambient light into pale-gold internal highlights: these are the fungus's primary carbon and energy reserves, triglyceride-rich droplets synthesized from sugars delivered by the host plant and destined for export outward along the extraradical mycelium to fuel soil exploration far beyond this root. The host cell has been reduced to a pressurized film of pale-green cytoplasm squeezed to the margins — ribosomes, organelles, and cellulose microfibrils still present but compressed under a load they were never designed to bear — while a single intraradical hypha no wider than five micrometers threads silently between two vesicles at the lower periphery, its colorless taut wall faintly glistening, the sole filament connecting this oil-packed interior vault to the invisible external network and everything it sustains.
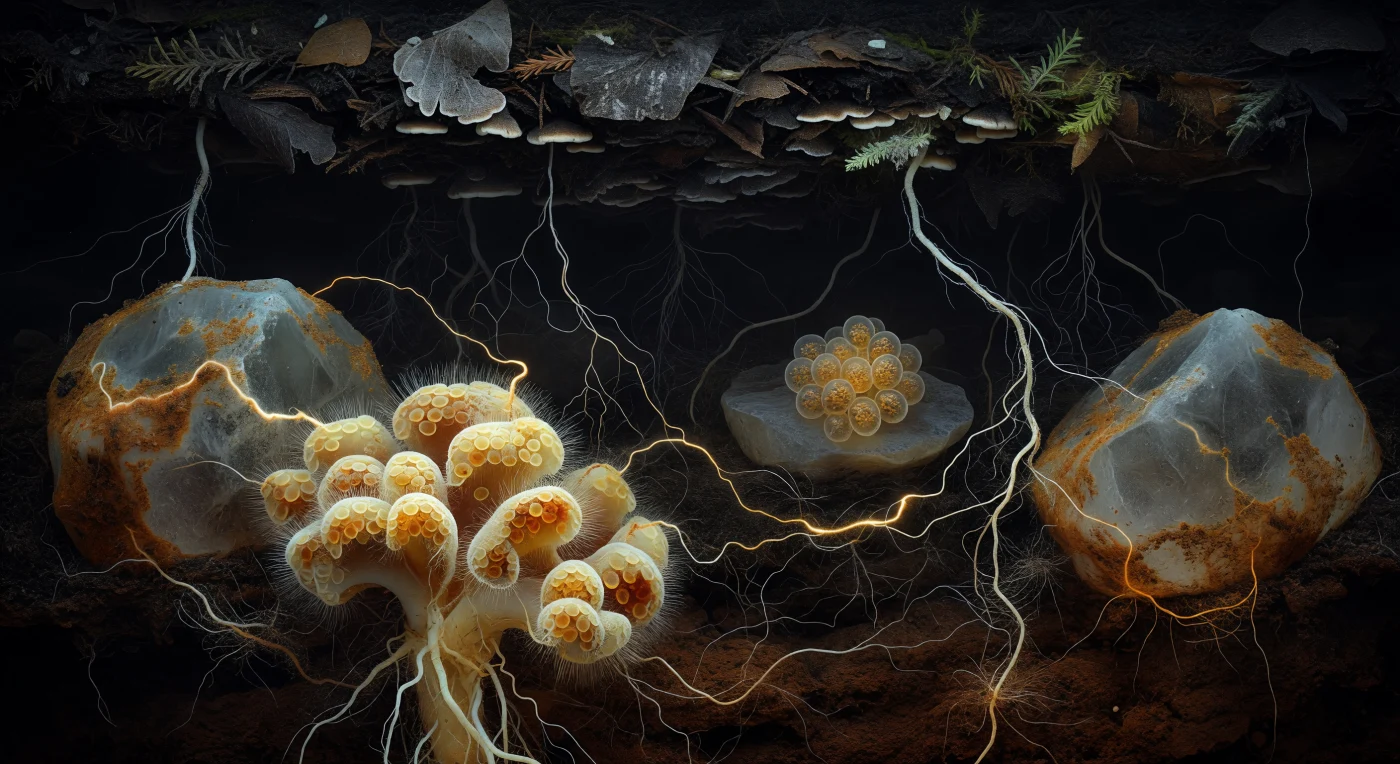
You are drifting through absolute darkness inside a pore channel no wider than a crack in old stone, suspended between quartz grains the scale of cathedral walls, their surfaces glazed in amber organic matter that seems to hold warmth rather than emit light. Directly ahead, an ectomycorrhizal root-tip cluster rises like a baroque reef structure: its mantle sheath layers cream, butter-yellow, and rust-ochre pseudoparenchyma around the root's pale cortex, outer hyphae radiating into the surrounding soil matrix like hair lifted in a slow current, the whole assembly functioning as a living interface where the tree surrenders photosynthate carbon and the fungus returns phosphorus and water scavenged from mineral surfaces no root hair could reach. Weaving outward from this cluster and threading across tens of centimetres of dark soil, the Common Mycorrhizal Network materializes as gossamer white hyphae — individually one to ten micrometres across — braiding near root surfaces then thinning to near-invisibility across bare feldspar faces, a recursive three-dimensional mesh that physically links trees of different species into a shared biochemical conversation. Mid-ground, an amber spore cluster rests against a flat mineral face, its stratified walls enclosing lipid globules that refract the scene's faint inferred luminosity into honeyed gold, each spore a dormant archive of fungal genetic potential awaiting the chemical signal of a nearby root. Above, fine roots descend from the near-black humus ceiling like silver and cream cables, some bare, others already cloaked in new mycorrhizal mantles, all of them dependent on this lightless web to survive.

Before you, the severed face of the rhizomorph fills your entire field of perception like the exposed end of a buried foundation column, its concentric architecture as deliberate and layered as anything engineered: an outer ring of melanized cells fused into something approaching black glass, then the abrupt crossing into a cathedral of pale pipe cells whose thin walls glow with a cold, diffuse luminescence, and at the very center, a hollow axial canal running into the dark interior, its moist lining catching faint light like the inside of a wet pipe. This is an ectomycorrhizal rhizomorph — a multicellular, differentiated fungal organ, not a simple hypha, built to transport water and carbon across meters of lightless soil with a structural logic analogous to plant vasculature: the dense melanized rind provides mechanical protection and resistance to soil pathogens, while the wide-lumened medullary cells function as low-resistance conduits, their vacuoles and thin walls optimized for bulk flow. The melanin packed granularly into those outer walls is not incidental — it confers resistance to enzymatic degradation, desiccation, and ultraviolet exposure during any above-ground phase, a biological armor evolved across hundreds of millions of years of subterranean chemistry. Outside the rind's sharp perimeter, fine satellite hyphae thread outward between quartz grains pressed against the structure like cobblestones against a buried cable, each hypha a few microns across and nearly glassy, dissolving into the compressed labyrinth of clay platelets, organic films, and mineral aggregates that constitute the soil matrix — the whole system a living cable, ancient and humming with an invisible chemistry of carbon, phosphorus, and water moving perpetually between tree and earth.

Before you stretches a landscape that has no business existing at the scale of a sand grain, yet here it is in full geological grandeur: a branching fan of living filaments advances across a canyon floor of amber quartz monoliths and tar-black organic mortar, each hyphal tip pressing forward with a cold blue-white cytoplasmic glow that is the only light this world has ever known. The advancing front belongs to an arbuscular mycorrhizal fungus in the act of colonizing virgin soil, its extraradical mycelium extending outward from a central runner hypha in a river-delta formation — older segments behind the front strung like chains of clouded glass beads where large vacuoles interrupt the cytoplasm at intervals of twenty to forty microns, younger segments near the tips dense and opaque with organelles, rounded like blind worms testing the dark. Where hyphae thread through narrow soil pores between mineral grains they deform slightly into oval cross-sections, their glow concentrating at the constriction as cytoplasm squeezes forward, while others span open macropores freely like suspension cables between cliff faces, coated in moisture films only microns thick. Scattered across the midfield, pale cream-colored calcite grains are being chemically dismantled at points of hyphal contact — orange-yellow dissolution haloes of oxalic acid chemistry pit and frost the mineral surfaces, a warm sulfurous glow working against the cold biological luminescence in a transaction that transfers mineral phosphorus into living tissue. Far in the background, barely resolved through layers of soil particle and organic film, a vast curved wall of root-hair epidermis rises like a destination continent, its cellulose-fibril texture enormous and cliff-like, organizing every tip's trajectory into something that feels less like growth and more like biological urgency written in living architecture.
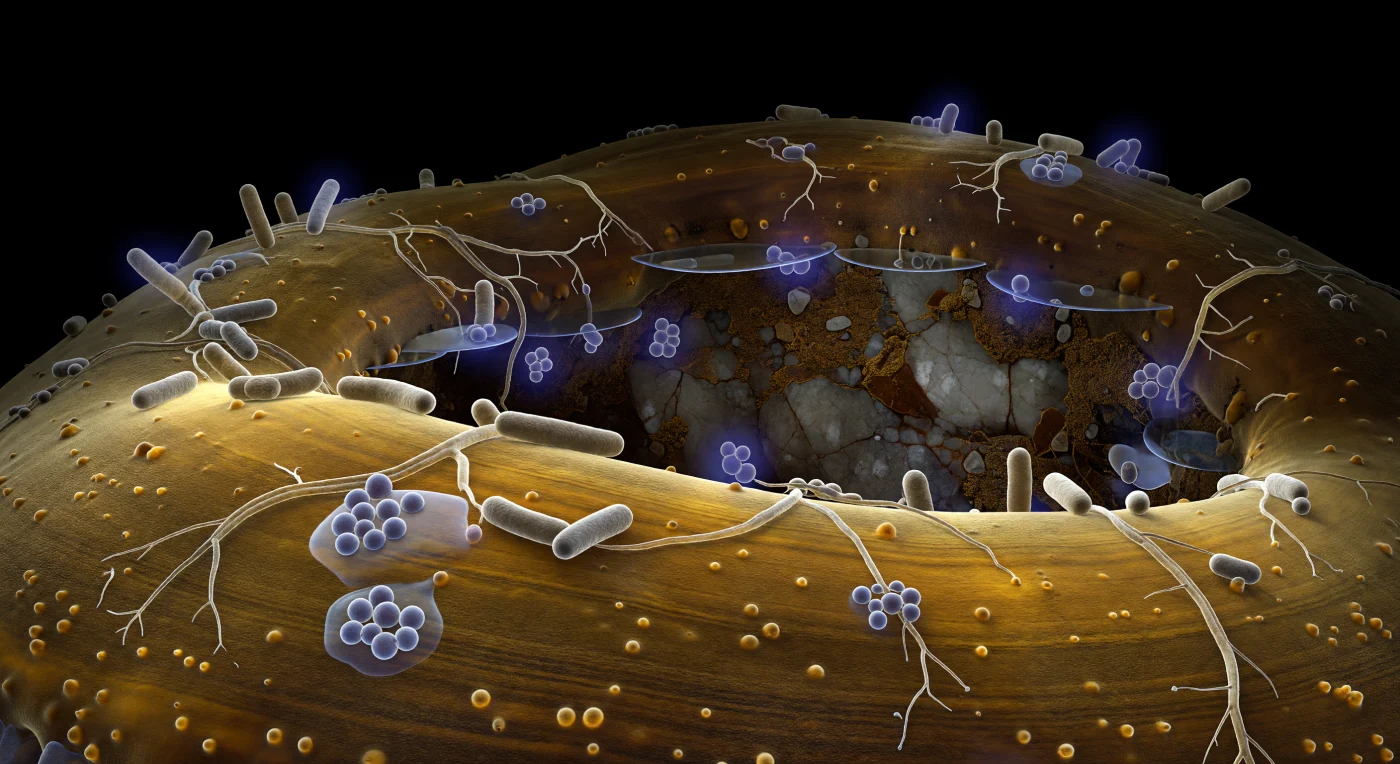
The curved amber wall rising before you is the outer surface of an ectomycorrhizal hypha — a single fungal filament perhaps seven micrometers wide, yet reading here as a glazed sandstone escarpment that fills your entire horizon, its chitinous laminations glowing from within as cold cytoplasmic light filters through semi-translucent layers of polysaccharide and glycoprotein. Across this hillside topography, rod-shaped bacteria of the genus *Bacillus* and branching *Streptomyces* filaments constitute a hyphosphere microbiome — mycorrhiza helper bacteria known to suppress pathogens, solubilize nutrients, and produce phytohormones that enhance fungal colonization of host roots, forming one of the most ecologically significant microbial guilds in forest soil. The bacteria cling and sprawl in small waxy colonies bridged by exopolysaccharide gels that pool between cells like spilled glycerin, anchoring the community to the hyphal wall through adhesive tethers visible only where the cytoplasmic glow catches their contact angle; individuals broadcasting chemical signals into the surrounding aqueous film appear wreathed in faint violet diffusion halos, their molecular messages dissolving within a few cell-lengths into the water that fills this subterranean pore. Beyond the hypha's curvature, a cavernous soil void opens into absolute darkness, its far wall a fractured feldspar face surfaced with organic films the color of dark toffee, bridged to the hypha by mirror-flat water menisci that catch the amber glow in single glinting reflections. There is no light from above — only chemistry, membrane boundaries, and the permanent collaborative negotiation of living surfaces pressed against one another in the lightless architecture beneath the forest floor.

Suspended in the absolute dark of a soil pore beneath a temperate meadow, you face a single *Dactylorhiza* orchid seed that fills your field of view like a translucent airship moored against cliffs of black basalt and grey quartz — its seed coat a single iridescent membrane no thicker than a soap film, refracting the cold chemistry of pore water into ghostly silver and pale blue-green interference colors, enclosing a loose cluster of colorless embryo cells that glow with the dim warmth of paper lanterns, metabolically suspended at the very edge of possibility. From the lower left advances a Rhizoctonia hypha reading at this scale as a massive lacquered cylinder of chitin, warm ochre shading to umber at its septate nodes, its cytoplasm visibly streaming with organelles, and where it has breached the seed coat the membrane gathers into a tight puckered annulus around the entry point, sealed by the living tissue itself. Inside one embryo cell the peloton occupies the entire cellular interior — a wound-spring coil of hyphae dozens of loops deep, amber and pale ivory where the turns compress against each other, pressing the cell wall into a slight ellipse as it burns with the slow metabolic negotiation of two organisms deciding whether this contact is invasion or alliance. The adjacent embryo cells are already answering: fractionally enlarged, their membranes drawn taut, their interior luminosity shifting from cool silver-white toward a warmer cream-gold as the earliest molecular steps of differentiation begin — the fungus delivering fixed carbon and mineral phosphate in exchange for photosynthate the seedling cannot yet make, a mutualism so obligate for this orchid that without it the seed, containing no endosperm and almost no stored energy, would simply remain paused here in the dark until it died. Beyond the seed, mineral grains press in like brutalist architecture coated in caramel films of humic acid, pore water curving into perfect menisci under surface tension, and at the far edge of focus pale bacterial rods cling to quartz surfaces like barnacles on a sea cliff, impossibly small even here.

You are suspended inside a lightless soil pore just beneath a root system that died perhaps days ago, and what surrounds you is nothing less than the controlled disassembly of a biological empire. Overhead, the dead root cortex has collapsed into dark chocolate ribbons and spongy cellular wreckage, from whose fissures AMF spores tumble free in slow motion — spherical bodies the size of boulders and small houses at your scale, their thick walls glowing amber, wine-red, and ochre, some already cracking to leak pale lipid fluid into the surrounding water film, each spore a dormant archive of genetic and lipid capital now released into open competition. Every mineral surface around you — quartz towers rising like office buildings on all sides — is lacquered in glomalin, the glycoprotein secreted by arbuscular mycorrhizal fungi that stabilizes soil aggregates and sequesters carbon for decades, its amber resin film catching the faint bioluminescent chemistry of decomposition and diffusing it into the only light this world possesses. Across the mid-distance, remnant common mycorrhizal network threads still run silver-white toward living neighboring trees, their cytoplasm barely flowing as the dead tree's hydraulic pressure slowly equalizes — while against this elegance, saprotrophic hyphae twice their diameter colonize the cortex above with blunt, matte urgency, their chemistry optimized not for mutualism but for enzymatic demolition of lignin and cellulose. From opposite sides of the pore, two mycorrhizal fronts — one pale yellow, one cream-white — converge on this nutrient-rich catastrophe, their approaching filaments already contacting the outermost rolling spores, the entire scene a silent negotiation over the phosphorus, nitrogen, and carbon wealth of a giant's last bequest.















