Scientific confidence: High

You hover centimeters above what the eye first reads as a coastal plateau at dusk — a broad, gently undulating mesa of dark gray-brown integument stretching rightward, its surface alive with a pearlescent sheen where rhabdite secretions and subsurface guanine deposits catch the oblique amber beam raking in from the upper right, the dorsal epidermis resolving into a subtly quilted mosaic of thousands of individual cells, each flagstone-facet glinting differently, while along the lateral margins the tissue thins to a translucent amber where gut diverticula trace dark dendritic shadows beneath, like rivers glimpsed through ice. At the left margin, the world transforms: the blastema rises as a shallow opalescent dome, glassy with retained moisture, scattering the warm light back as cold blue-white luminescence — a freshly formed plateau of pale milky translucence, smoother than any mature epidermis, sitting proud above the older pigmented tissue along a boundary ridge that marks the exact biological shoreline between differentiation and possibility. Within it, barely resolved but unmistakably present, two minute black pinpoints sit like distant volcanic craters on a white plain — nascent ocelli, dark pigment just beginning to consolidate around the photoreceptor clusters of a nervous system still being built neuron by neuron. Five days ago this anterior quarter of a living animal did not exist; what the light now sculpts so dramatically into geological relief is nothing less than the reinvention of a body from pluripotent cells, a process ancient enough to predate the Cambrian and quiet enough to be mistaken, at this scale, for slow stone.

The view is pressed flat against the underside of a granite cobble, looking upward into a ceiling of living stone — feldspar and quartz territories interlocking at enormous apparent scale, their mineral edges catching refracted stream-light as faint prismatic glints, the entire surface filmed with a crust of periphyton in olive-drab, gold-green, and pale ochre where diatom colonies have colonized every facet. Three Dugesia planarians occupy this ceiling-world above, each a long dark ellipse of ash-gray and deep umber moving with the unhurried, muscular inevitability of something that has no skeleton to constrain it — bodies pressed intimately to the biofilm, flattened to near-perfect contact across their entire ventral surface by the continuous coordinated beating of cilia too small and too numerous to resolve, the locomotion appearing as pure, seamless glide. They are backlit by cold blue-green stream-light filtering down through the water column above the cobble and refracting into this sheltered underside chamber as a diffuse submarine luminescence, rimming each animal's flattened margins in smoky amber-green where tissue thins to translucency, the faint branching architecture of gut diverticulae just visible through the dorsal surface like weather systems seen from altitude. Behind each animal, the mucus ribbon it has laid down across the diatom mat catches the scattered upwelling light as a barely-there silver filament — refractive, slightly raised, a molecular adhesion highway still warm with the animal's passage — while deep in the mid-water beyond the cobble's sheltering lip, a mayfly nymph drifts as a soft volumetric amber ghost, warm against the aquamarine of the water column, its segmented body unresolved by distance into pure luminous suggestion.
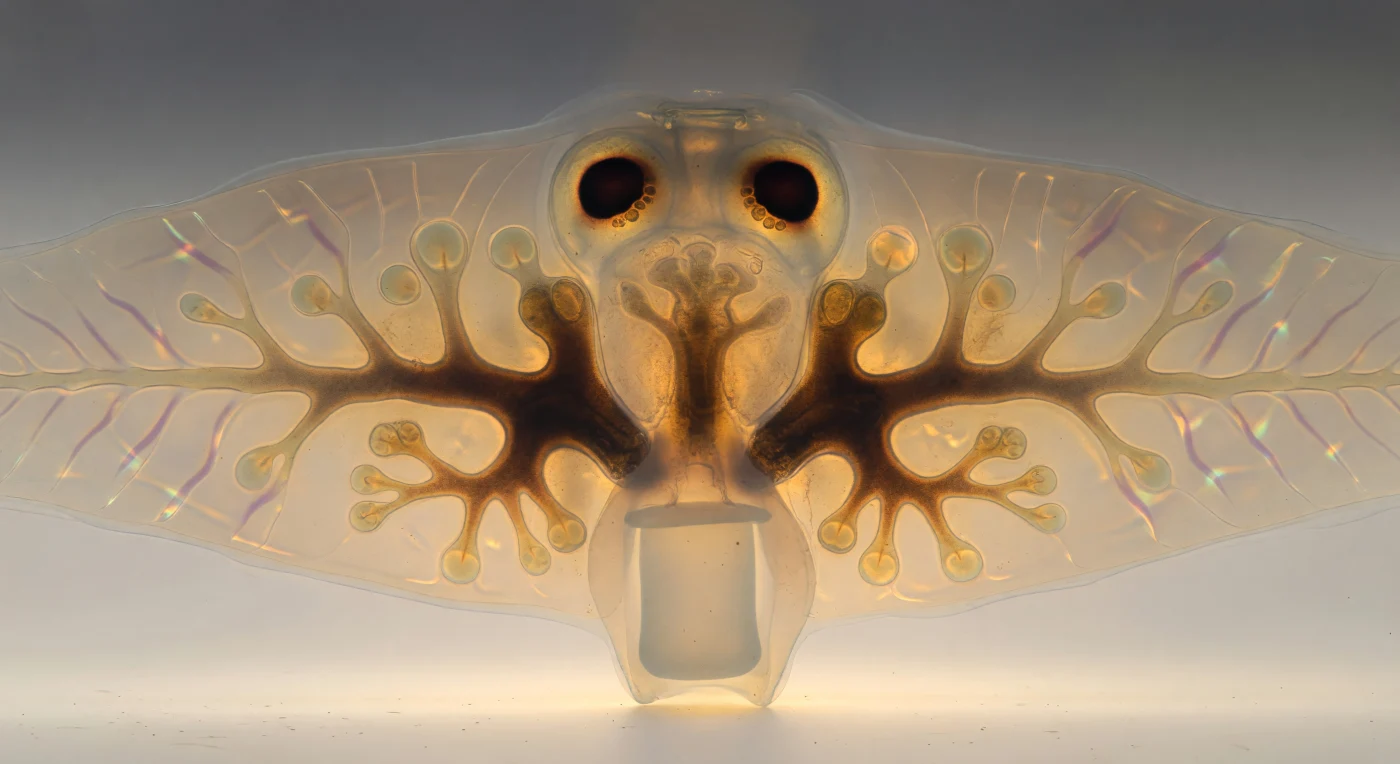
You are suspended just above the belly of a living *Dugesia tigrina*, hovering at the height of a single epidermal cilium, looking upward through the body as warm transmitted light floods the scene from below like sun through medieval glass. The creature stretches to every horizon overhead — a vaulted cathedral of translucent amber tissue, its ventral membrane threaded with diagonal muscle-fiber lattices that scatter the upwelling warmth into a honeyed haze, each cellular junction edged with the faintest ghost of copper-to-aquamarine iridescence where the surrounding glycoprotein mucus bends the light. Rising deep into the interior like the ribbed nave of an ancient basilica, the triclad gut ramifies in three great ochre-brown corridors — one driving straight toward the anterior, two sweeping symmetrically to either side and subdividing into tapering diverticula whose walls, dense with phagocytic cells and partially digested matter, bleed light sideways into the surrounding parenchyma and cast warm annular halos at every branching terminus, the whole structure less a shadow than a luminous drawing pressed into living flesh. Toward the anterior horizon, where the tissue thins to near-total translucency, the two ocelli loom as the scene's most dramatic punctuation — crescent masses of dense, light-absorbing pigment cupped around photoreceptor clusters that flare at their rims into burnt-sienna halos, two dark moons haloed by atmospheric scatter, serving as the animal's sole interface with the directionality of light in a world where vision means nothing more than knowing which way the brightness lies.

The viewer floats just above a calcium carbonate plateau of encrusting coralline algae, its surface cracked into polygonal cells of deep magenta and salmon pink that stretch in every direction like a vast mineral cityscape — and dominating this rocky terrain, the Pseudobiceros flatworm spreads itself like a luminous flag, its dorsal surface saturated electric fuchsia-violet shot through with the branching shadow-map of its gut diverticula showing faintly beneath translucent skin. This animal is among the most elaborate of the free-living polyclad turbellarians, an acoelomate whose dorsoventrally compressed body reaches near-transparency at its ruffled lateral margins, the white-cream frills thrown into perpetual complex folds by shallow surge — a consequence of tissue so thin and pliable that water movement alone sculpts it into standing waves of wet silk. The pool water above, only six centimeters deep in reality, reads here as an entire sky of liquid glass, its air-water interface refracting Pacific midday sun into caustic nets of hammered gold that slide continuously across the pink crust, veining every surface in shifting rivers of amber light and deep shadow. The mucus film secreted along the flatworm's ventral contact zone catches these same caustics as a mirror-bright meniscus, marking the animal's passage across living stone with a glittering molecular trail — the physical record of cilia-driven locomotion across a landscape that, at this scale, has the full presence and complexity of a continent.

The spelunking beam finds them first — two white lozenges resting motionless on calcite sinter the color of old ivory, their bodies so pale and translucent that the branching gut diverticula glow through the tissue like river tributaries seen through frosted glass, warm beige-brown dendrites suspended inside a form that otherwise appears to be made of nothing but cold light and water. These are cave planarians of the genus *Dendrocoelum*, evolutionary refugees who have shed every pigment and both ocelli across countless generations in absolute darkness, navigating now by chemical gradients and vibration alone, their blunt anterior ends reading the subterranean stream through senses that have no use for the LED beam now illuminating them so clinically from above. Beneath their bodies, each animal floats above its own pale ghost-shadow cast by scattered upward light onto the sinter, while the calcite substrate itself — built in slow concentric ridges of deposited calcium carbonate, each layer a record of dissolved limestone precipitating from cold water over decades — fills the field of view like a frozen tundra of mineral, every ridge a soft lip of pale ivory frosted with microcrystalline texture. The water surface overhead is black glass, broken only by a single rigid stripe of reflected white light, and beyond the beam's edge the cave collapses into a darkness so complete that only isolated crystal facets betray their existence, igniting briefly as cold violet sparks before the void swallows them again.

You are suspended millimeters above an obsidian plain — the wet-glass substrate stretches in every direction like a frozen black lake, its shallow water film refracting overhead light into trembling caustic streaks — and directly before you, two living landmasses of *Dugesia dorotocephala* are in the final seconds of tearing themselves apart. The anterior body has moved forward, its chromatophore-dense dorsal surface rippling with slow muscular contraction waves, its flared auricles and crescent-moon ocelli facing away from the drama, while the posterior body anchors itself to the glass through locked adhesive glands, its isometric contractions visibly flattening and ridging the tissue in rhythmic resistance. Between them, the gravitational center of this entire world: a tissue thread no thicker than spun glass, translucent white verging on pale gold, backlit by the water film beneath so it glows with apparent luminescence against the surrounding darkness, and within it — if you resolve your gaze — parenchymal cells still connected, longitudinal muscle fibers drawn to their elastic limit, extracellular matrix holding suspension at the threshold of rupture. This is asexual fission in *Platyhelminthes*, a reproductive strategy requiring no partner and no gamete, only the coordinated muscular pull of a body dividing itself into two regeneration-competent fragments, each carrying the molecular instructions to rebuild what is lost — and the silver mucus roads crossing the black substrate in every direction are the calligraphic record of how this single animal became, in the time it takes to watch, irrevocably two.

You are pressed into cold, silty mud at the margin of a freshwater pond, your vantage point no higher than a grain of sand, and above you the pale underbelly of a *Dugesia* planarian stretches overhead like a living vault — its translucent tissue glowing amber-green in the diffuse surface light, the branching gut diverticula visible as dark dendritic shadows pressed against luminous flesh, biological stained glass suspended over a terrain of compacted clay platelets and drifting organic filaments. At the precise center of the scene, the everted pharynx descends as a fleshy muscular cylinder — pale pink-white and catching every available photon — pressing its circular suction lip against the brick-red body of a *Tubifex* worm partially buried in the silt like a segment of cable, a slow-expanding radial plume of displaced mud particles suspended around the contact point in perfect amber sparks. The pharynx is a muscular hydraulic structure, everted by coordinated hydrostatic pressure and retractor muscles, capable of delivering enzymatic secretions and generating suction sufficient to tear soft prey tissue within seconds of contact. What reads here as monumental architecture — the gripping ventral adhesive glands anchoring the predator to the substrate, the compression ripples radiating outward through the mud surface, the cathedral murk filtering down through millimeters of olive-green water — is a feeding event that, in real time, unfolds in under a minute, the entire biological violence of it compressed into this single luminous, suspended instant.

You stand within a colonnade without end — silver-gray shafts rising all around you to heights that dwarf your own, their tapered crowns glazed in a blue-silver mucus film that catches the low, raking light like hoarfrost on winter grass, while several nearest shafts are caught mid-beat, their upper thirds angled sharply in the same oblique direction and trailing faint smears of arrested motion at their tips. Beneath your feet, the epithelial floor rolls gently in polygonal plateaus separated by low cellular ridges, the surface studded at irregular intervals by dark, perfectly circular gland-cell pores that drop away like volcanic calderas into pitch-black interiors rimmed with a wet overflow of freshly secreted mucus. This is the ventral surface of a living planarian, and the ciliary array that drives the animal's gliding locomotion — each shaft a precisely structured axonemal engine beating at dozens of cycles per second — surrounds you as an effectively infinite forest whose individual members you can reach out and touch. The mucus film pooled in lenses between the cell ridges refracts the background aquatic light into pale prismatic halos of blue and amber, and the distant cilia dissolve into a silver-gray atmospheric haze at what feels like a continental horizon but amounts to a distance no greater than the width of two human hairs. Above, the underside of the water-mucus interface trembles as ciliary wave fronts propagate outward in coordinated metachronal rhythm, a living ceiling rippling with slow caustic light that filters down in shifting ribbons of pale aquamarine through a medium that is simultaneously the animal's lubricant, its adhesive, and its interface with the world.

You are looking at the nervous system of a living flatworm rendered as pure light — a bilateral architecture of serotonin-labeled neurons blazing vivid green against absolute black, captured through confocal fluorescence microscopy of an eight-millimeter planarian whole-mount. At the anterior, two pear-shaped ganglia fuse into a bilobed brain, the organism's entire cognitive infrastructure compressed into paired jade masses from which twin ventral nerve cords extend the full length of the body like luminous rails, bridged at regular intervals by transverse commissures that form the ladder structure giving the image its organizing geometry. Peripheral neurons branch outward from these cords as fine fractal tendrils, dimming toward the body margins, while a diffuse blue haze of DAPI-stained nuclei suffuses the surrounding darkness — thousands of cell nuclei occupying the parenchymal tissue like a faint stellar field behind the green architecture. What makes this image extraordinary is its compression: the complete wiring diagram of a regeneration-capable, bilaterally symmetric animal — an organism capable of regrowing its own brain — rendered as a single luminous glyph, geometric and cathedral-like, its serotonergic pathways tracing the deep evolutionary logic of the very first nervous systems to organize around a head.

You are pressed flat against the underside of a decomposing tropical leaf, your entire ribbon body — twelve centimeters of chocolate-brown and cream-banded tissue — conforming without resistance to the microtopography of collapsed veins and white fungal hyphae, reading the substrate through a thin film of your own mucus while the only light in this absolute forest-floor darkness comes from bracket fungi thirty millimeters to your left, pulsing cold blue-green bioluminescence that catches every moisture droplet as a prismatic lens and throws the hyphal lattice into sharp low-relief shadow. *Bipalium kewense* is a land planarian — a free-living platyhelminth that abandoned aquatic life for the saturated chemistry of tropical leaf litter, moving by muscular undulation across surfaces it grips with adhesive glands, its hammer-shaped head sweeping in slow lateral arcs as chemosensory margins parse molecular gradients that its diffuse nervous system processes not as thought but as turning. The earthworm emerging at the frame's far edge — its annular segments each the width of your entire body, its skin surging in visible peristaltic rings — represents a prey item you will subdue by everting your pharynx directly through your ventral surface, secreting digestive enzymes externally before ingesting liquefied tissue, a feeding strategy unchanged in its fundamentals since the earliest bilaterians moved across Ediacaran seafloors. Behind you, a thin iridescent line of mucus catches the bioluminescent light — the only record that anything has moved here at all, already being colonized by the same microbial decomposers that are slowly returning this entire leaf to soil.

The underside of a lifted river cobble has become a sudden stage: twenty flatworm bodies ranging from a few millimeters to roughly two centimeters lie distributed across a biofilm plain of diatoms and bacterial mat, their dorsally dark forms now catching intrusive afternoon sunlight that pours in from the exposed stone margin as a violent gradient of white-gold fading into deep olive shadow. These are free-living turbellarian planarians — soft-bodied, bilaterally symmetrical animals navigating the world through ciliary gliding across mucus secretions, guided by primitive ocelli that register light intensity rather than images, and already the phototactic response is visible: animals at the illuminated margins are contracting and reversing within seconds, longitudinal muscles bunching the tissue into visible waves, while those deeper in shadow continue to extend forward in tapered ribbons, their translucent posterior margins faintly ghosting the branching gut diverticula beneath. The mucus trail network left by their nocturnal movements covers the biofilm floor in overlapping arcs of iridescent secretion — fresh trails raised slightly above the substrate surface and catching lateral light as bright silver ribbons, older trails already colonized by bacteria and matte with amber tone — a chemical and structural record of hours of movement suddenly made visible. At the lower frame, a caddisfly larval case rises as a rough cylinder of sand grains cemented in silk, each quartzite and feldspar grain glinting with its own mineral facet, a structure orders of magnitude more massive than the flatworms beside it, the entire scene suspended in that first, violent instant of a nocturnal world's exposure to daylight.

Between the grains, the world is immense. Quartz spheres rear up on every side as amber and glass boulders, their curved surfaces acting as natural lenses that project wavering aureoles of caustic light into the water-filled corridors between them — a shifting lantern-show of refracted brightness rippling with the micro-turbulence of the overlying water column. Through this luminous labyrinth thread two acoel turbellarians, their torpedo-shaped bodies nearly colorless, backlit by grain-lens light so that interior parenchyma and gut contents glow faintly through translucent body walls like candle flames encased in living tissue. Golden-brown pennate diatoms lie lacquered against the grain surfaces in warm amber highlights, while deeper in the passages, where diffuse greenish-blue light from the shallow coastal water above fails to penetrate, a matte biofilm fills the lowest crevices in abyssal darkness — the geological negative of every glowing grain surface above it. These animals navigate by cilia and chemical gradient, exquisitely at home in a world that reads, at their scale, as both cathedral and continent.
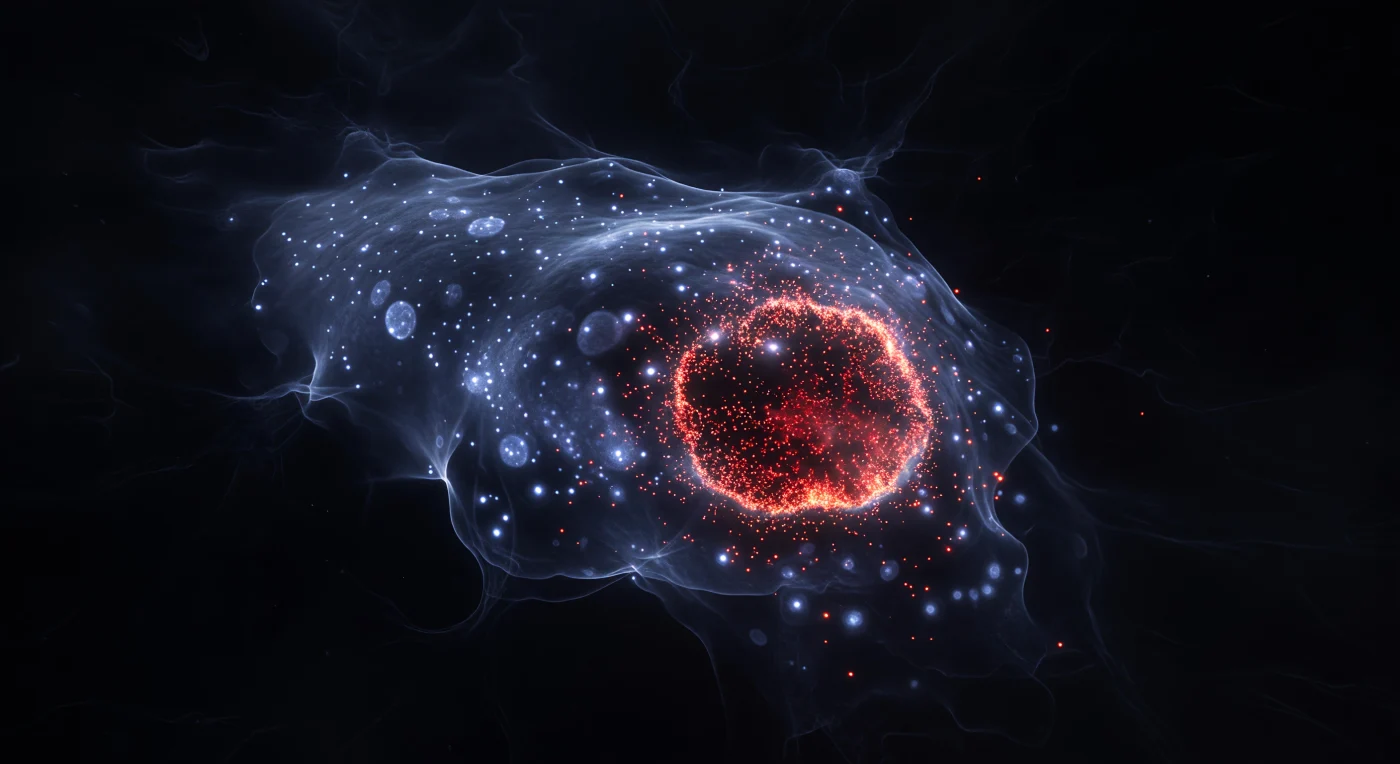
You are suspended in biological darkness, drifting at the scale of a single dividing cell, surrounded by a cosmos of cold nuclear light and volcanic crimson fire. The translucent body of a planarian — a freshwater flatworm imaged by fluorescence microscopy 48 hours after amputation — arches around you as a soft electric-blue shell: every nucleus stained with DAPI reads as a cold point of light, billions of cellular suns separated by dark parenchymal voids, the tissue between them a frozen gel of structural density. Moving toward the anterior wound margin, this quiet star field catastrophically thickens: neoblasts, the planarian's pluripotent stem cells, have migrated and proliferated in response to injury, their newly replicated DNA branded by EdU incorporation into brilliant crimson puncta that crowd, converge, and finally fuse into a continuous incandescent nebula — a solid wall of scarlet and deep red where individual dividing cells are no longer distinguishable, their signals merged into biological fire. This blaze represents one of animal biology's most dramatic regenerative events: neoblasts are the only dividing cells in the planarian body, and their rapid recruitment to wounds underlies the organism's legendary capacity to regenerate a complete head from a fragment of trunk within two weeks. The contrast between the sparse posterior ember-field and this anterior supernova encodes, in a single frozen image, the entire logic of regeneration — quiescence behind, urgency ahead, and the wound margin itself a luminous frontier between two states of biological time.

At eye level with the glass floor, a razor-sharp frontier divides the world into two hemispheres — to the left, warm amber-white light bleaches every surface to overexposure, and to the right, a cool teal-gray shadow absorbs that warmth into something denser and more private, the boundary between them not a gradient but a surgical wall of photons ending with architectural precision. Pressed into that first millimeter of darkness, twelve gray-brown planarians rest against the light-dark edge like travelers who have found a border and stopped, their dorsoventrally flattened bodies matte and low against the crystal-clear glass, surfaces shifting between ash-gray and a faint iridescent silver where rhabdite cells in the epidermis catch the oblique light at steep angles. One animal straddles the boundary in frozen mid-reversal — its auricle-bearing head already stilled in shadow, the paired crescent ocelli just visible as dark brown dots near the dorsal margin, while its posterior half remains in warm illumination, the branching gastrovascular cavity glowing as an olive-brown tracery beneath translucent skin, backlit like stained glass. The illuminated half behind them is a forensic record of departure: a lacework of dried mucus trails refracts the warm light into thin silver filaments, hairline iridescent threads crisscrossing the glass like a map of panic, overlapping paths recording every decision made by bodies that now rest, invisible, in the dark — the entire scene a behavioral experiment written in mucus, shadow, and the simple calculus of an animal that reads light as danger and darkness as safety.

You are standing inside a living cathedral that has no stone, only water, cellulose, and light. In every direction, the cylindrical stems of Fontinalis antipyretica rise like columns from dark gravel, each one branching into overlapping whorls of narrow, keeled leaves whose cellular architecture — a visible grid of thin-walled, elongated cells — catches the caustic light thrown by the stream's surface far overhead and glows like backlit jade, warm shafts of blue-white illumination rippling rhythmically across the geometry as the current shifts above. Through this dappled, fractal nave moves Crenobia alpina, a cold-stenothermic planarian restricted to the oxygen-saturated waters of alpine and boreal streams, its cream-white, dorsoventrally flattened body gliding on a coordinated wave of ventral cilia embedded in a mucus layer that adheres to every substrate irregularity, its anterior auricles sweeping slowly through the water column as chemoreceptive organs sampling dissolved cues — prey, conspecifics, the faint metabolic signatures of the microworld. Sharing this same dappled microspace are a spherical water mite — a predatory deutonymph, its carmine cuticle specularly bright where the caustic shaft strikes it — and two copepods hovering on extended antennae in the small eddies that the moss architecture generates, their amber-translucent bodies and beating swimming legs visible in full detail at this scale, every appendage a working instrument in the physics of low-Reynolds-number flow. Beyond them, the interior of the moss clump dissolves into layer upon layer of overlapping fronds, their accumulated transparencies building a cool green haze that deepens toward blue-black at its furthest extent — the whole scene a living pressure vessel of biological complexity, where the interstitial water itself is structured by surfaces, flows, chemistry, and the soft bodies navigating between them.















