Scientific confidence: High
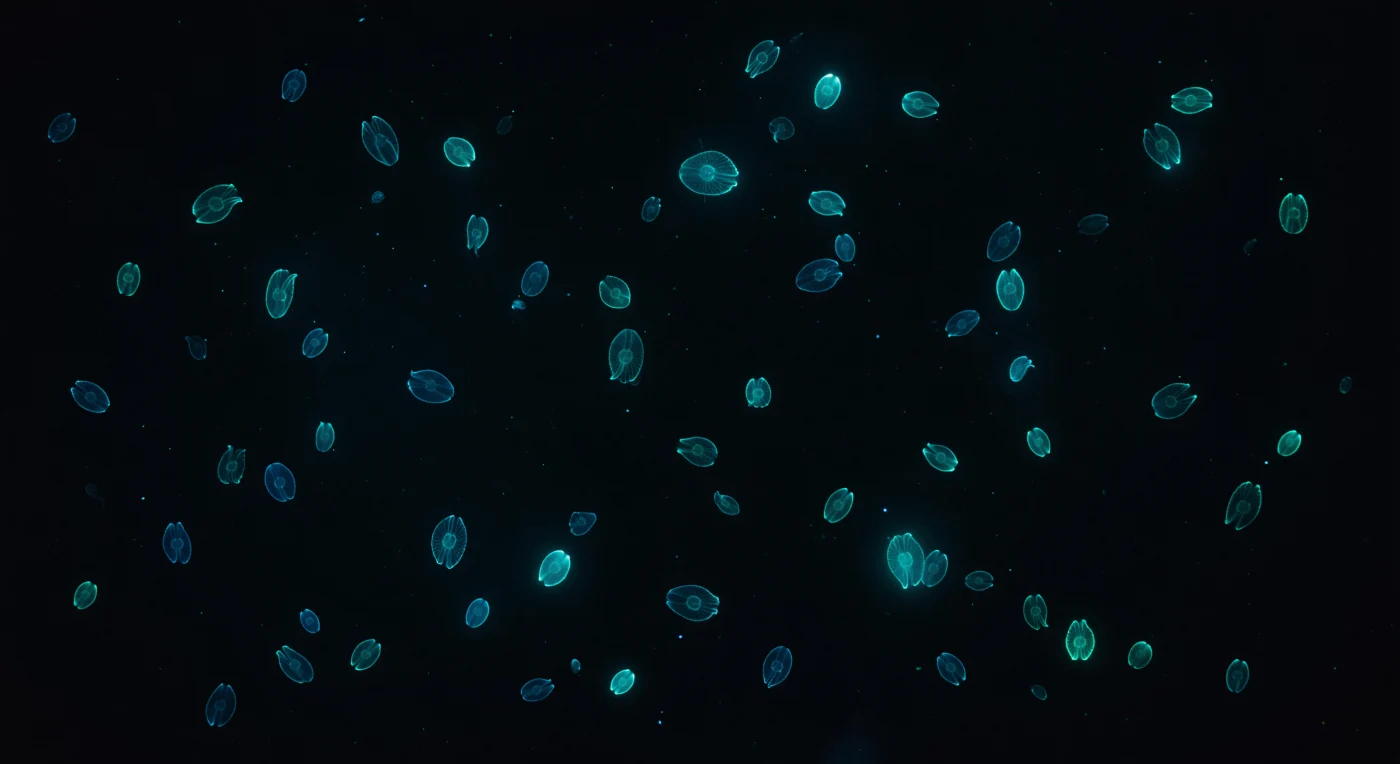
Suspended in the absolute darkness of a moonless ocean, you are surrounded by living geometry — dozens of *Mnemiopsis leidyi* drifting in every direction, their gelatinous bodies so perfectly transparent that they vanish entirely into the black water, leaving only eight ribbons of cold blue-green light to trace each animal's outline against the void. These are the comb rows, eight longitudinal bands of fused cilia whose underlying photocytes fire in a luciferin-luciferase cascade triggered by mechanical disturbance, emitting at precisely 490 nanometers — a teal that exists nowhere on land — as waves of bioluminescent activation travel slowly and repeatedly from the blunt aboral pole down toward the oral end, each traversal taking roughly a second before the row dims and reignites from the top. The mesoglea composing each body is a viscoelastic gel of collagen, glycoproteins, and water refracting light at nearly the same index as seawater itself, so that the animals register only as the faintest curved meniscus, a ghostly lensing distortion where living tissue meets ocean. Farther animals dissolve into soft cyan coronas as suspended marine snow scatters each photon outward before extinguishing it, giving the black water a luminous volumetric texture — shafts of blue-green light extending centimeters into open water, illuminating nothing but the sea itself. The aggregation breathes asynchronously around you, some comb rows mid-pulse, others just igniting, a field of cold living constellations drifting on imperceptible currents through water that is otherwise lightless and absolute.

The ROV's blue LED cuts through four hundred meters of absolute darkness and the water column assembles itself in an instant: a slow constellation of marine snow drifts downward through the beam like white ash suspended in cold amber, and within it, materializing from nothing, the deep wine-red forms of *Bathocyroe fosteri* hang in loose proximity, their lobate bodies shaped like folded velvet lanterns burning crimson against a void so complete it feels less like the absence of light and more like the negation of space itself. These are lobate ctenophores — soft-bodied predators built almost entirely of mesoglea, a viscoelastic gel of collagen and water whose refractive index so closely matches seawater that the animals are optically invisible in ambient conditions, their deep pigmentation an adaptation that prevents bioluminescent flashes from silhouetting them against the faint downwelling light above. Under the 470-nanometer beam, the eight comb rows running longitudinally along each body surface act as living diffraction gratings — thousands of compound ciliary plates fragmenting the blue light into metachronal pulses of violet, teal, and amber that sweep the body in slow electric ripples, pure structural color with no chemistry involved, the physics of light encountering precisely spaced biological architecture. The marine snow filling the illuminated volume — loose aggregates of mucus, fecal pellets, and bacterial colonies, each fleck a millimeter or two across, sinking at roughly a meter per minute toward the sediment below — gives the scene its spatial depth and its strange patience, nearer particles sharp and white, distant ones dissolving into gray suggestion, everything moving with the unhurried certainty of a world accustomed to never being watched.

Suspended at eye level with a creature that barely registers as solid matter, you drift in the luminous blue-green water column of the Mediterranean shallows, watching eight arching comb rows ignite in traveling waves of pure spectral fire — carmine blooming into amber, then acid green, then violet — cascading along the flanks of a *Bolinopsis infundibulum* whose mesoglea is so perfectly refractive-index-matched to the surrounding sea that the animal announces itself only as a ghost of lens-distortion and living rainbow. Each discrete comb plate, a compound paddle of fused cilia beating at tens of cycles per second, acts as a biological diffraction grating: as its geometry tilts through the stroke cycle it paints a fresh band of the visible spectrum before the next plate ignites in antiplectic metachronal sequence, so the entire body shimmers in slow-burning, asymmetric aurora across eight staggered rivers of light simultaneously. The oral lobes drape open below, their mesoglea faintly flushed with the rose tracery of underlying gastrovascular canals — visible only where backlit by surface glow, like capillaries through pressed skin — while far beneath, sunlight broken by surface chop has written long sinuous caustic ropes of gold across the rippled sandy floor, a warm amber underworld that makes the intervening water column feel like cathedral nave space, vast and photon-saturated. This is an organism that is ninety-six percent seawater given form, a soft-bodied predator running a bioluminescent diffraction engine in open water, revealing through iridescence alone the improbable fact of its existence.
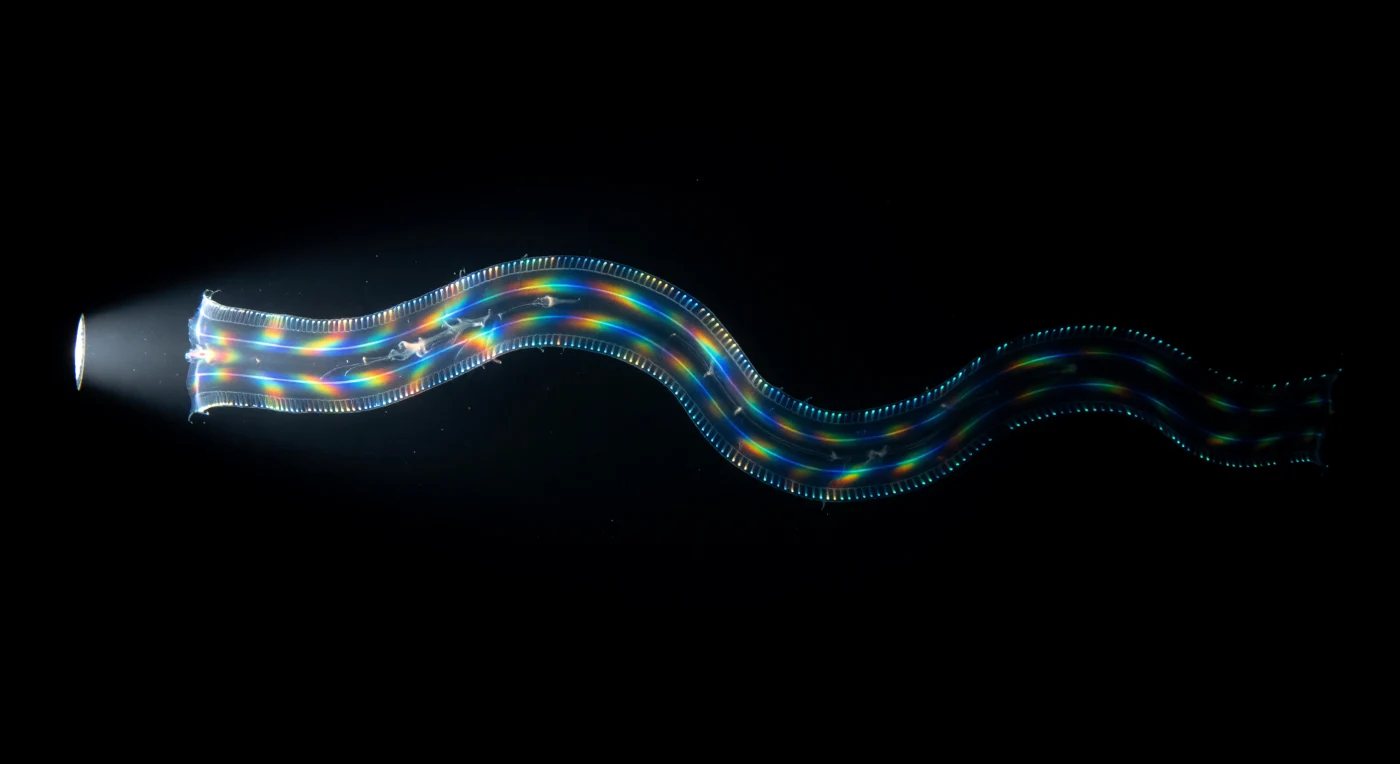
You hang suspended in absolute blackness, weightless in body-warm tropical water, and from the void a ribbon materializes — sixty centimeters of *Cestum veneris*, the Venus's girdle, flexing through your field of view in a single long sinusoidal standing wave, its gelatinous body so perfectly refractive-index-matched to seawater that you perceive it less as a solid object than as a slight lensing of the dark, a membrane that separates one emptiness from another. What betrays the animal entirely are its four comb rows, the defining architecture of all ctenophores — parallel seams of compound ciliary plates running the ribbon's full length, each plate a fused array of thousands of individual cilia beating in antiplectic metachronal waves at up to thirty-five cycles per second, and every pulse triggering the photocytes beneath to fire their cold luciferin light at 490 nanometers, so that the creature outlines itself in traveling blue-green foxfire against impenetrable black ocean, a slow-breathing waveform of bioluminescence tracing every sinuous flex. Then your flashlight strikes the ribbon broadside, and the comb plates transform instantly from bioluminescent seams into diffraction gratings in violent motion — the mechanical spacing of thousands of beating cilia refracting white light into its full spectral components simultaneously, red bleeding into gold into cobalt into violet across the satin-clear width of the body, the entire color sequence scrolling in a strobing cascade as the metachronal wave advances, a spectral banner unfurling and re-furling in real time against a substrate of mesoglea so water-clear it reads as structured void. The animal undulates back into darkness, the flashlight colors vanishing as it exits the beam, and those four cold blue-green lines re-emerge as the only evidence of continued existence — narrowing, slowing, fading — until the ocean reclaims them completely and the water presses back in around your face, warm and dark and unmarked.

Below fifteen meters in a February sea loch, the peat-stained water filters what little light reaches this depth into a cold, directionless jade, and within that dim suspension a dozen Pleurobrachia pileus hang motionless in the mid-water column — spherical bodies no larger than glass marbles, each one so nearly transparent that they register less as objects than as subtle displacements of the green-grey murk, their curved mesoglea walls refracting a thin lens-flare of ambient light along each equator while soft celery-green gut contents glow faintly at their cores. Each individual belongs to the phylum Ctenophora, animals constructed almost entirely of viscoelastic mesoglea — a collagen-threaded, water-saturated gel whose refractive index is so closely matched to seawater that the body verges on optical erasure. What betrays them is movement: eight comb rows arc over each sphere like the meridians of a tiny world, their fused ciliary plates beating in antiplectic metachronal waves that sweep muted rose and amber iridescence along each band in a rhythm that pulses like slow breathing, a structural color produced not by pigment but by the diffraction of whatever faint light filters down through the plankton-hazed column above. Behind the nearest individual, a pair of gossamer tentacles extend fifteen centimeters into the particulate haze, their colloblast-studded tentilla vanishing entirely into the suspension — invisible fishing lines set inside a cold, pressured world where gelatinous life drifts in loose congregation through water that is itself more biology than emptiness.

In the warm, glass-clear upper meter of the Gulf Stream, two nearly invisible animals have locked together in a transaction so slow and complete it reads more as merger than predation: a *Beroe cucumis* has stretched its entire macrostome mouth around more than half the body of a *Mnemiopsis leidyi*, and the combined form — perhaps fifteen centimeters from the *Beroe*'s aboral pole to the *Mnemiopsis*'s still-protruding posterior — hangs suspended in a column of luminous cobalt water while late-afternoon caustics sweep golden interference patterns continuously across both translucent bodies. The *Beroe*'s mesoglea, a viscoelastic matrix of collagen and water matched so precisely to the refractive index of the surrounding sea that it reads as tinted air rather than tissue, glows rose-apricot from within, its branching meridional canals mapping a bilateral river delta of digestion in real time, faint pulsations visible through walls only a few cells thick. Where the prey still emerges into open water, its four exposed comb rows continue their metachronal programs at fifteen to thirty beats per second — each ctene plate a fused paddle of ten thousand cilia sweeping structural color from violet through amber to green in rolling cascade, an involuntary rainbow that has not yet received the signal of its own ending. One by one, beginning at the oral end already dissolving inside the predator's pink interior glow, the iridescent flickers lose their rhythm, the last posterior plates still cycling their light into the darkening blue column below as the two gelatinous architectures, almost indistinguishable from the water that composes them, complete their slow exchange.

We drift motionless in the black water column of a warm summer estuary, held in place by nothing, surrounded by three *Mnemiopsis leidyi* bodies that loom through the darkness as enormous cathedral vaults of near-invisible gel — their mesoglea walls so refractive-index-matched to the surrounding seawater that we perceive them less as solid organisms and more as slow-breathing distortions in space, betrayed only by the eight comb rows pulsing along each flank in staggered metachronal sequence, each ctene plate igniting in turn at 490 nanometers and cascading a cold blue-green ripple from pole to lobe like light drawn along living fiber optic cable. From gonopore slits running beneath each comb row, slow seeping plumes of sperm billow outward into the water column — too viscous to be called jets, too pale and pearlescent to be called clouds — each one catching a neighbor's bioluminescent emission so that its interior briefly becomes silver-white edged in cold blue before sliding back into milky opacity against infinite black, the individual spermatozoa far below our resolution but their collective density forming a reproductive fog that hangs in the mid-water, laced with chemical signals that orient the gametes invisibly. Tumbling freely through this fog are the eggs — near-perfect refractive spheres roughly 120 micrometers across that read at our scale as small glass marbles, each acting as a micro-lens that gathers the surrounding bioluminescent light and compresses it into a brilliant internal caustic, a cold star ringed by a faint prismatic halo of spectral color, dozens of them glinting simultaneously at different depth planes so that the nearest burn sharply and the farthest dissolve into mere luminous suggestions swallowed by the dark. The whole scene — glowing gelatinous walls, drifting pearlescent gamete plumes, a three-dimensional constellation of tumbling refractive eggs — composes a biological aurora, entirely self-generated, suspended in warm black saline silence thirty feet below the surface of the Chesapeake at two in the morning, urgency rendered in the softest possible light.

Looking straight up from twelve meters below the surface of Hardangerfjord in high August, the water column above dissolves into a cathedral of drifting glass — hundreds of *Bolinopsis infundibulum* suspended at every depth between viewer and the rippled silver ceiling of the surface, each individual an oval lantern of near-perfect transparency, eight to twelve centimeters long, their mesoglea so optically matched to the surrounding seawater that they read less as solid matter than as refractive ghosts, betrayed only when a shaft of Nordic sunlight catches their geometry at precisely the right angle. The defining spectacle is the eight comb rows running meridionally along each body: longitudinal bands of fused compound cilia — each plate bearing roughly 100,000 individual axonemes beating in antiplectic metachronal waves — that act as living diffraction gratings, cycling continuously through red, gold, teal, and violet as ciliary plates open and close in rolling sequence, the phase offset between neighboring animals ensuring that at any frozen instant the entire chandelier above shimmers with asynchronous, polychromatic fire. Several of the nearest individuals carry a warm apricot blush through their gastrovascular canals, the translucent flush of recently ingested copepods visible through colorless mesoglea the way a yolk shows through an egg held to the light, while broad volumetric columns of blue-white sunlight descend from the surface at oblique angles and are spectrally dispersed by each comb row they intersect, smearing into short prismatic fans that fill the mid-water between bodies with a diffuse chromatic haze. The fjord's late-summer phytoplankton haze lends the water column a faint jade tint through which marine snow drifts in slow suspension, copepods scull in brief arcs of alarm, and the collective refractive and diffractive presence of hundreds of near-invisible animals transforms this cold Norwegian water column into something between a stained-glass nave and a field of slow-turning prisms — utterly silent, continuously luminous, and alive.

You hover in absolute darkness a kilometer beneath the Pacific surface, weightless in near-freezing water, as the ROV's blue LED beam cuts through the void and strikes something that should not exist at this depth — a ctenophore the color of fresh blood, *Lampocteis cruentiventer*, suspended motionless in the water column like a dropped jewel. The animal belongs to the lobate ctenophorans, gelatinous predators whose bodies are composed almost entirely of mesoglea, a viscoelastic collagen-glycoprotein gel that is ninety-six percent seawater by mass yet holds its architecture with surprising structural integrity, the broad oral lobes hanging open and relaxed like the draped petals of a dark tulip, their wet surfaces throwing sharp cyan highlights where the blue beam strikes the uppermost rims before the light vanishes entirely into tissue so saturated with red pigment that no wavelength below 600 nanometers survives to reflect back. Eight comb rows — the defining feature of all Ctenophora, each a longitudinal series of compound ciliary paddles fused from hundreds of thousands of individual axonemes — trace faint topographic seams across the scarlet body, their plates too small to resolve individually at this distance yet collectively imposing a ribbed texture that catches the raking illumination along the animal's flanks. Marine snow drifts continuously through the cone of light, particles of biogenic detritus on slightly diverging vectors in the ambient deep current, giving measurable depth and scale to water so dark it is otherwise indistinguishable from the inside of a closed eye, while the ctenophore blazes at the center of the beam as though generating its own red light — a body the size of a human fist, sovereign in a desert of cold pressure and absolute black.

You hover motionless above the summit of a living comb jelly, looking down into a structure that seems less biological than architectural — a hemisphere of near-perfect optical clarity, two hundred microns across, rising from the animal's aboral pole like a cupola of biological glass whose presence is announced only by the finest crescent of bent light tracing its equator. Inside this dome, a creamy aggregate of calcium carbonate granules — the statolith — hangs in suspended stillness, its chalky ivory surface casting a soft diffuse shadow into the transparent tissue beneath; this is the animal's entire gravitational sense organ, a statocyst from which the whole ctenophore reads orientation and coordinates the beating of its eight comb rows accordingly. Four fans of balancer cilia surround the statolith at cardinal positions, each rendered not as discrete filaments but as a frosted vibratory aureole — beating at tens of cycles per second, scattering transmitted oceanic light into pearlescent hazes that shift and shimmer like breath on cold glass. From the dome's base, four ciliated furrows radiate outward like the seams of a compass rose, their surfaces alive with finer motile cilia that produce a barely perceptible iridescent wash of silver and green at grazing angles, extending away into the middle distance where the first luminous ribbons of the comb rows begin their arcing paths along the body — the entire scene suspended within a vast medium of transparent mesoglea, the open ocean visible beyond the animal's curved wall as a featureless luminous cobalt void, simultaneously intimate and infinite.

Before you floats a two-millimeter sphere of living glass — a cydippid ctenophore larva so optically matched to the surrounding seawater that it registers less as a body than as a subtle warping of the dawn light filtering down through the surface microlayer, its presence betrayed only by faint prismatic sweeps of rose and violet as eight rudimentary comb rows beat their metachronal rhythms, each plate a fusion of thousands of cilia whose coordinated strokes will, in time, make this animal one of the ocean's most efficient gelatinous predators. The larva is built almost entirely of mesoglea — a viscoelastic extracellular matrix of collagen and glycoproteins whose refractive index so closely matches seawater that transparency is not camouflage but simply the consequence of being mostly structured water, while two hairlike tentacles already extend outward, their adhesive colloblast tips sampling a surrounding medium that is far from empty. Drifting past at what feels like chest height is a chain of Chaetoceros diatoms, each silica frustule a faceted amber lantern many times the larva's own diameter, their long hyaline spines scattering slanted dawn photons into prismatic needles; beyond them, an orange copepod nauplius strokes methodically through the suspension on three pairs of appendages, its single eyespot a bead of obsidian, its carapace burning copper in the diffuse surface glow. This is the ocean's dawn plankton world — a dense, three-dimensional cathedral of living particles, marine snow aggregates, and shifting caustic light where the larva hangs at the exact luminous boundary between the lit surface and the darkening deep, already alive and already hunting.

You are suspended at forty meters in open water, weightless in a column of blue that cools and greens perceptibly at eye level, where the thermocline asserts itself as a continuous trembling lens — a horizontal shimmer stretching wall to wall across the entire visible field, warping distant shapes the way summer heat warps asphalt, the density boundary between two distinct water masses rendered optically visible as a faint undulating glaze. Shafts of filtered afternoon sunlight angle down from above and diffuse into wide cones of pale gold that terminate just above this mirror-line, leaving the colder water below in a dim, greenish, particulate twilight dense with marine snow and scattered orange-red copepod specks — zooplankton concentrated precisely at the interface where mixing brings nutrients and the thermal step slows their descent. Three *Mnemiopsis leidyi* hang motionless at the boundary, each a flattened oval of near-perfect optical transparency — five to eight centimeters of mesoglea whose refractive index so closely matches seawater that the thermocline shimmer passes straight through their bodies and reappears on the far side, while their eight comb rows erupt in slow-rolling sweeps of structural color, ruby sliding to amber to acid green to indigo as each ctene plate acts as a diffraction grating and the metachronal wave travels mouth to aboral pole in under a second. Their oral lobes hang open and extended downward into the colder prey-rich zone, hairlike cilia drawing copepod-laden water upward across colloblast-studded surfaces, the animals straddling the thermocline with the precision of buoyancy masters — upper halves lit in warm filtered gold, lower halves bathed in cold green dusk — the refractive boundary line bisecting each transparent body like a ruled line drawn cleanly through living glass.

You are suspended inside a living animal smaller than a fingernail, yet the space you occupy reads as vast — the interior of a two-millimeter cydippid larva rendered in light-sheet fluorescence, its absolute black background interrupted by cold blue DAPI-labeled nuclei drifting through the mesoglea like sparse stars at varying depths, each a small hard cerulean sphere marking a cell nucleus within the organism's water-rich extracellular matrix. From that darkness, a non-hierarchical web of magenta filaments — FMRFamide-immunoreactive fibers of the diffuse nerve net, a neural architecture so ancient it predates bilateral symmetry — radiates in every direction without center or hierarchy, branching and re-branching asymmetrically, nodes glowing where fibers converge, casting soft rose halos into the void around them. Eight arcs of acid green sweep overhead like the ribs of a vaulted cathedral, each one marking a serotonin-positive comb row band — the sensory and locomotor infrastructure that coordinates the metachronal ciliary waves driving this animal through open water — their clean lateral edges imposing a spatial grammar on the otherwise anarchic filament web below. At the zenith, the aboral apical organ blazes as a near-white circular halo where magenta and green signals co-localize in the statocyst's lithocyte crown, the gravity-sensing structure that tells the animal which way is up in a featureless ocean — and from here, drifting through overlapping focal planes where nearer filaments are razor-edged and distant ones dissolve into glowing threads, the felt scale of this two-millimeter creature becomes indistinguishable from a nebula.

You are floating at the scale of a copepod inside a kreisel tank — a specialized circular aquarium engineered to maintain laminar flow so that the animal before you never sinks, never touches a wall, simply hangs suspended as if gravity has been repealed. The *Mnemiopsis leidyi* fills the entire field of view like a slowly breathing airship of living glass, its body nearly 95% water by mass, its transparency arising not from absence of structure but from mesoglea — a viscoelastic extracellular gel whose refractive index so closely matches seawater that the organism bends light rather than blocking it, producing subtle internal caustics that shift with every pulse of the oral lobes. Eight comb rows arc along the body's meridians, each one a sequence of compound ciliary paddles beating in antiplectic metachronal waves at fifteen to thirty-five hertz, the massed cilia acting as a diffraction grating that splits the upwelling LED light into scrolling structural rainbows — red bleeding into amber, green cooling toward electric teal, violet fading back through blue — color generated by pure geometry rather than any pigment. Flanking the stomodeum, two pale coral-and-amber gonadal ribbons glow with granular warmth against the cold blue-white illumination, and at the far edge of focus a pipette tip hovers as an enormous out-of-register monolith, its mundane scale made planetary by the intimacy of this proximity to a single, suspended, quietly pulsing animal.

You are suspended beside a structure so fine it reads at first as a trick of the deep water's optics — a single tentillum of *Pleurobrachia*, a lateral filament no thicker than spun glass, arrayed along its length with dozens of colloblasts: adhesive cells unique to ctenophores, each one a domed hemisphere packed with refractile granules that throw back cold white-gold glints against the surrounding void of mesopelagic black-blue. This is a predatory surface unlike anything in vertebrate biology — colloblasts are not stinging cells but irreversibly adhesive ones, each dome tethered to the tentacle core by a spiral filament that functions as a coiled spring absorbing prey impact, and when a colloblast fires, its granular adhesive spreads biochemically across whatever surface it contacts, binding without venom, by pure molecular stickiness. In the upper right of your field of view, the prey has already arrived: a *Calanus* copepod's antenna segment, a beam of warm burnt-orange chitin sculpted in transverse ridges, has swept into the tentillum and several colloblast hemispheres are already crushed flat against its surface, deformed from perfect domes into adhesive discs of committed contact while spiral filaments unspooled behind them absorb the momentum of the strike. The tentillum itself curves infinitesimally under the copepod's weight, its pale ice-blue translucency beginning the muscular retraction that will haul this half-millimeter drama toward the ctenophore's mouth — a feeding event measured in seconds at organism scale, but here, at this proximity, unfolding as a slow catastrophe of cold light and irreversible chemistry.















