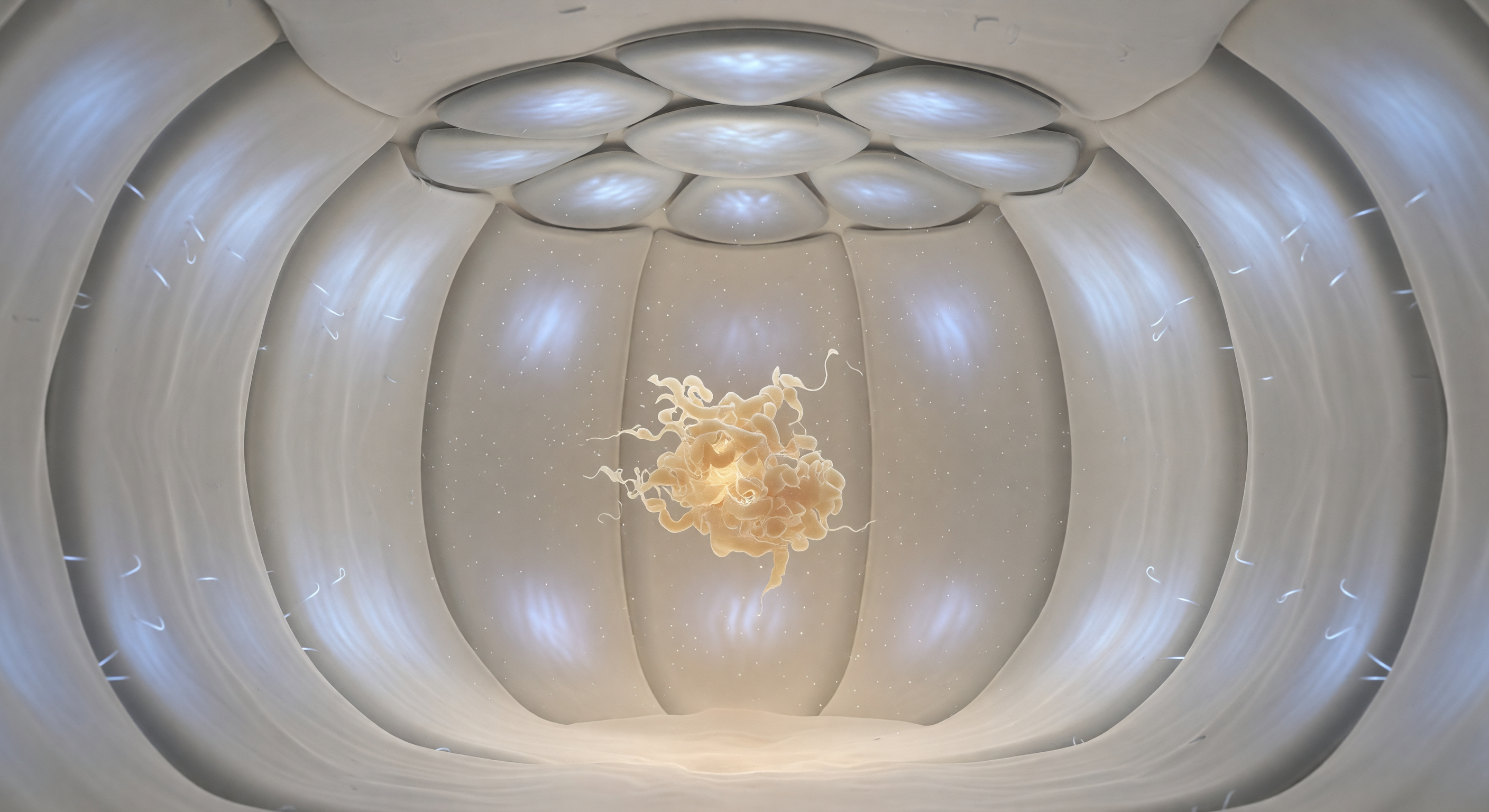
You find yourself suspended at the geometric center of a sealed molecular vault — a spherical enclosure roughly eight nanometers across whose pale, smoothly corrugated walls curve away from you in every direction with the silent completeness of the inside of a pearl, each segment of the GroEL barrel subtly articulated, its hydrophilic inner lining coated in outward-bristling side chains that catch a sourceless cool blue-white luminescence as though the protein itself radiates aqueous light. Overhead, seven rounded subunits of the GroES co-chaperonin lock together into a shallow dome, their fitted interfaces hairline seams of darker pewter, sealing this chamber against the cytoplasmic chaos outside — a molecular airlock designed by four billion years of selection pressure to give unfolded proteins a second chance. At the chamber's center, three subjective meters away, a misfolded substrate protein drifts in slow Brownian suspension, a crumpled mass of beige-amber polypeptide whose disordered loops fray probabilistically into the surrounding aqueous haze, glowing with a faint organic warmth entirely unlike the antiseptic radiance of the walls enclosing it. This is the GroEL-GroES chaperonin system in action: by sequestering the substrate within a chemically buffered, hydrophilic microenvironment and coupling ATP hydrolysis to cyclical enclosure and release, the complex suppresses off-pathway aggregation and grants the chain the isolation and time — milliseconds to seconds — needed to navigate its folding energy landscape toward a stable, functional conformation.
Other languages
- Français: Dans la Chambre Chaperonine
- Español: Dentro de la Bóveda Chaperonina
- Português: No Interior da Câmara Chaperonina
- Deutsch: Im Chaperonin-Faltungsgewölbe
- العربية: داخل قبة الشابيرونين
- हिन्दी: चैपेरोनिन कक्ष के भीतर
- 日本語: シャペロニン折畳み室の内部
- 한국어: 샤페로닌 접힘 공간 내부
- Italiano: Dentro la Volta Chaperonina
- Nederlands: Binnen de Chaperonine Vouwkamer