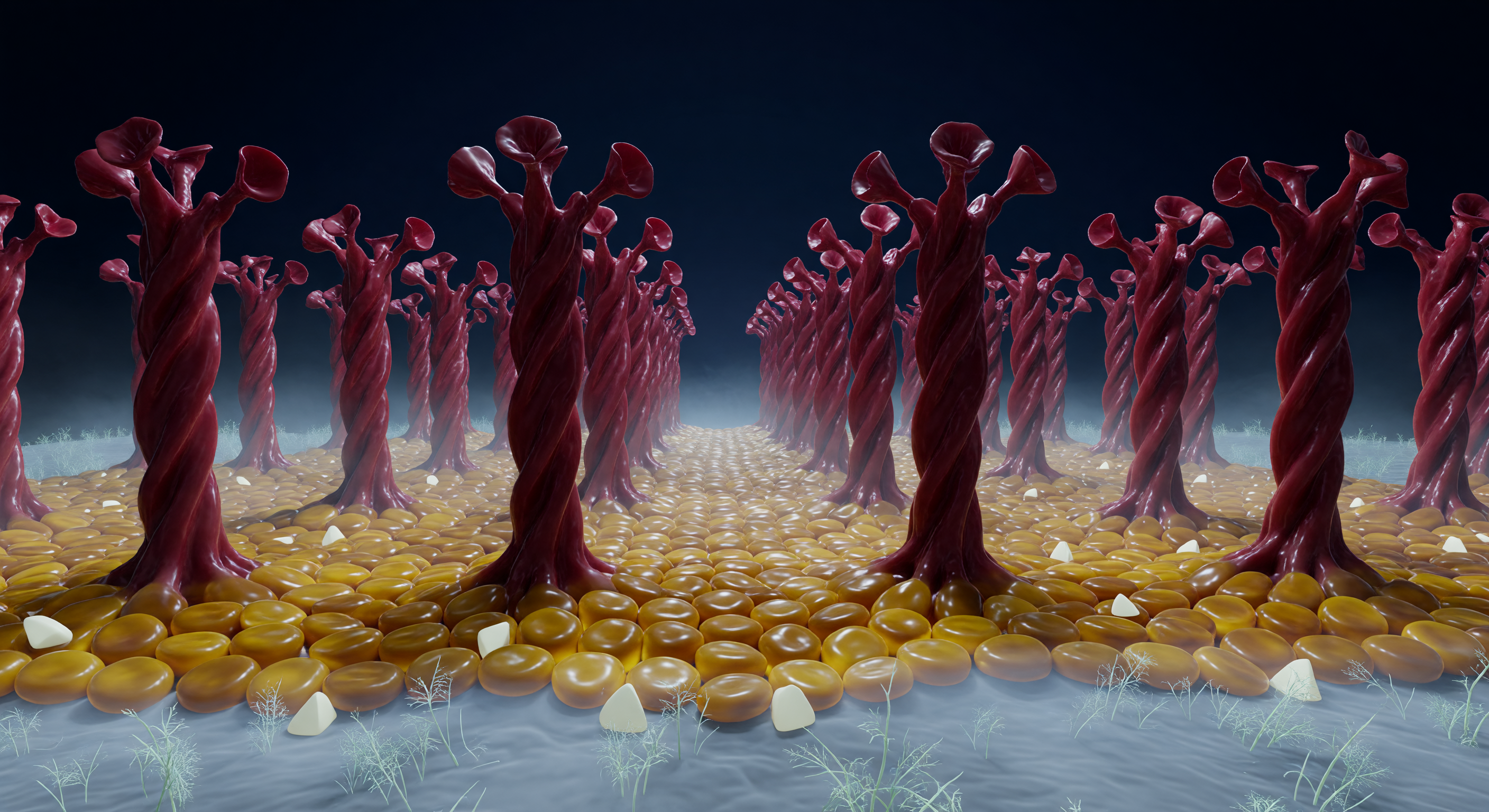
You are standing on the outer skin of one of the most consequential structures in modern biological history, and the ground beneath you is alive with motion. The surface rolls in slow thermal undulations — a fluid mosaic of tightly packed phospholipid head-groups glinting like amber river stones under diffuse bioluminescent light, stiffened in patches by pale cholesterol wedges, and crowned everywhere with a ghostly blue-white electrostatic haze rising from clustered phosphate charges. From this undulating plain, spike protein trimers erupt in every direction: massive oxblood columns twenty nanometers tall, each a triple-wound helix of extraordinary mechanical complexity, their transmembrane roots visibly distorting the lipid terrain, their upper receptor-binding domains splayed open like turbine blades reaching into void — the open conformation of a protein primed for cellular contact. The gaps between trimers feel like avenues through a forest of dark-red monoliths, the columns receding toward a curved horizon that betrays the spherical geometry of the entire virion, while far below, the host cell membrane stretches as a vast grey-blue glycan-frosted plain across the lower field of view. Nothing here is still: at this scale, thermal noise is a constant violent fact, every surface a potential binding event, and the entire architecture exists in a state of molecular urgency that the eye reads as beauty but the biology knows as predatory readiness.
Viruses