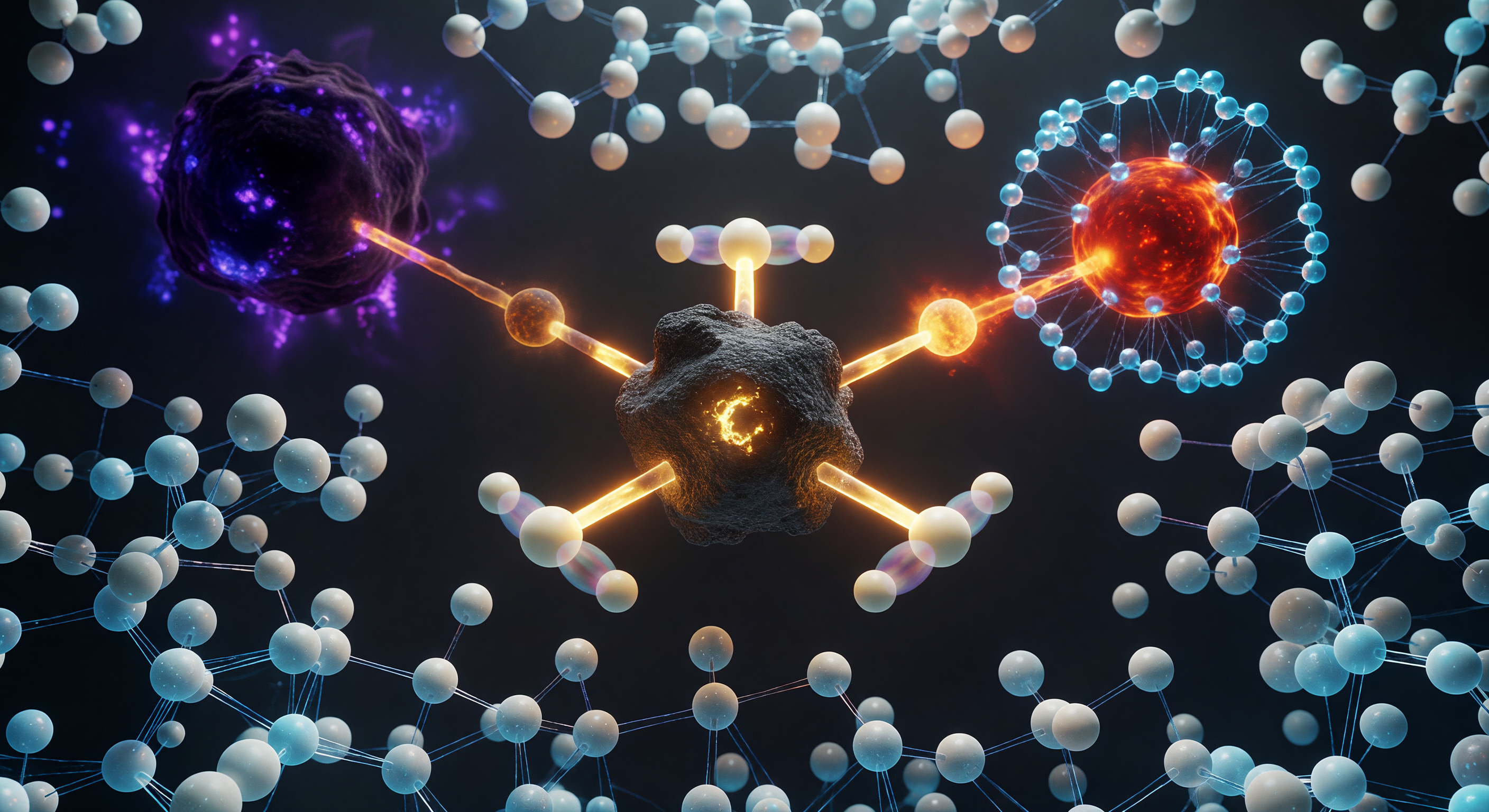
You hang suspended at the apex of a reaction that has not yet decided its own outcome — a moment so fleeting it exists only as a mathematical saddle point on the potential energy surface, lasting on the order of a single bond vibration, roughly ten femtoseconds. The central carbon atom, caught in trigonal bipyramidal transition-state geometry forbidden by equilibrium chemistry, holds three hydrogens locked in a perfect 120° equatorial plane while a nucleophilic oxygen closes from one pole at 2.0 Å and a bromine leaving group stretches away at 2.3 Å, both partial bonds at half-order, meaning neither fully formed nor broken — a condition quantum mechanics permits but classical intuition refuses. Around the carbon's equatorial hydrogens, ghostly translucent twins hover in near-coincident superposition, the signature of quantum tunneling probability distributions that allow hydrogen nuclei to sample classically forbidden positions even at this frozen instant. The surrounding solvent is not empty space but a pressing crowd of water molecules reorganizing their hydrogen-bond network in real time, their cyan halos of electron density straining and reforming to stabilize the emerging charge separation radiating outward from the reaction axis — because at this scale, solvation is not a background condition but an active participant in determining whether the reaction proceeds at all.
Other languages
- Français: Gel Quantique État de Transition SN2
- Español: Congelación Cuántica Estado SN2
- Português: Congelamento Quântico Estado SN2
- Deutsch: SN2 Übergangszustand Quanteneinfrierung
- العربية: تجمد كمي لحالة انتقال SN2
- हिन्दी: SN2 संक्रमण अवस्था क्वांटम फ्रीज
- 日本語: SN2遷移状態の量子凍結
- 한국어: SN2 전이 상태 양자 동결
- Italiano: Congelamento Quantistico Stato SN2
- Nederlands: SN2 Overgangstoestand Kwantumbevriezing