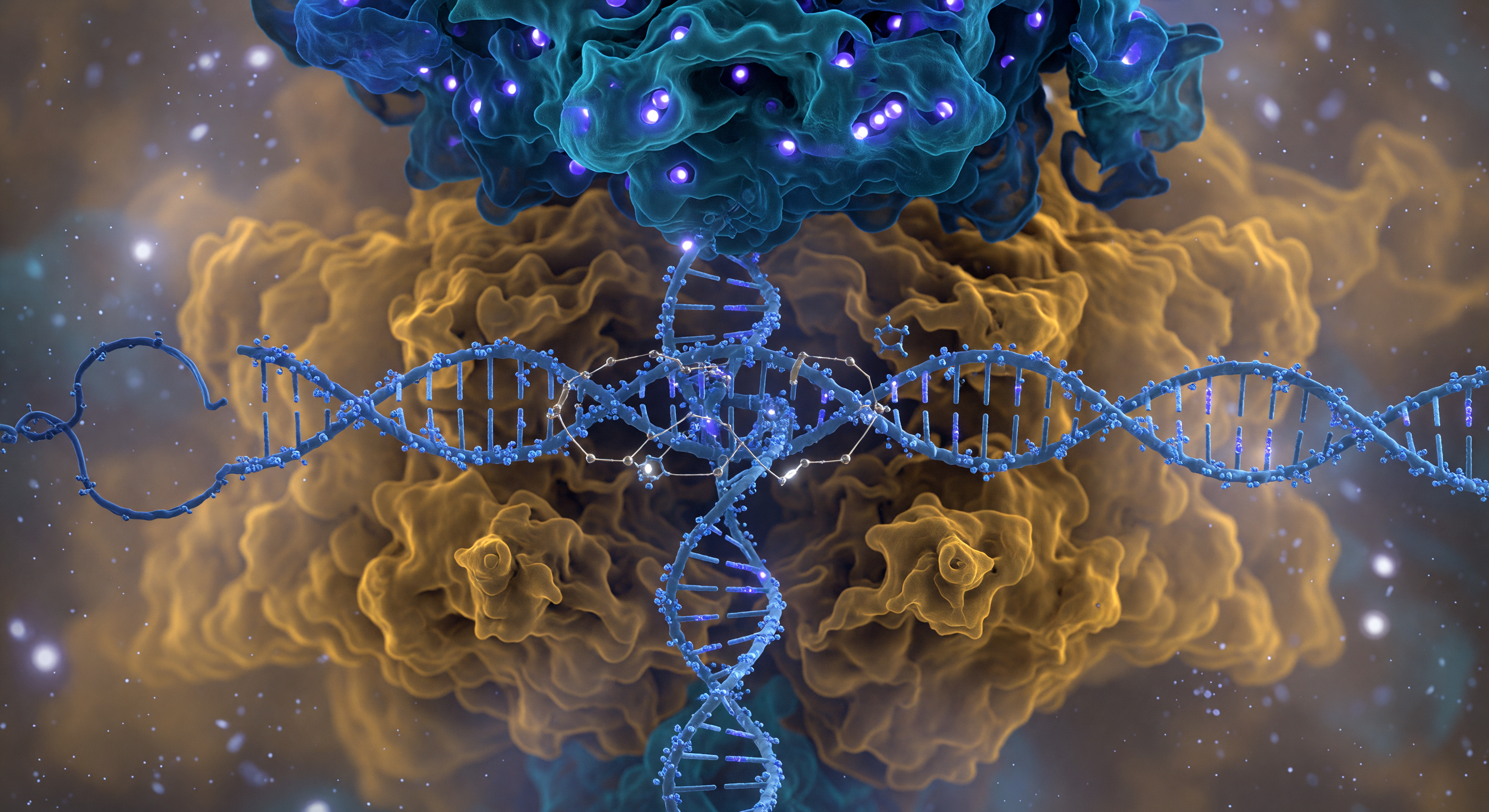
You are suspended five nanometers from the face of one of the most consequential molecular machines in biology, and the entire visual field is consumed by the Cas9 complex — a bilobed protein nearly ten nanometers across whose cobalt-blue recognition lobe overhangs you like a vaulted cathedral ceiling while the warm amber nuclease lobe swells outward below, both surfaces resolving not into hard edges but into probabilistic electron-density fog, the boundary between molecule and solvent perpetually negotiated rather than fixed. Threading between the two lobes, the ice-blue DNA double helix cuts across your view like a pillar of twisted rope under electrostatic tension, its phosphate backbone nodes glowing with cool violet charge while the displaced non-template strand loops away into the molecular haze — the major groove facing you directly, a shadowed canyon whose base chemistry is being read with extraordinary precision as the electric-cyan guide RNA spacer reaches inward to form R-loop base-pair bridges, each hydrogen bond rendered as a gossamer thread of gold-white light snapping two surfaces into complementary contact. On the rightward face, arginine fingers from the PAM-interacting domain press into the minor groove of the NGG trinucleotide like a key reading a lock, locally unwinding the helix geometry in a distortion you perceive as a visible narrowing of the groove. Deep in the catalytic cleft, paired magnesium ions occupy the HNH and RuvC active sites as brilliant white-hot pinpoints of divalent charge, each caged in octahedral coordination geometry — poised, once full R-loop formation is confirmed, to execute the precisely sequenced phosphodiester cleavages that will sever both strands of the target DNA with an accuracy that has reshaped the life sciences.
Other languages
- Français: Interrogation ADN par CRISPR
- Español: Interrogación del ADN con CRISPR
- Português: Interrogação do DNA pelo CRISPR
- Deutsch: CRISPR-Cas9 DNA-Erkundung
- العربية: استجواب الحمض النووي بكريسبر
- हिन्दी: CRISPR द्वारा DNA परीक्षण
- 日本語: クリスパーによるDNA探索
- 한국어: 크리스퍼의 DNA 탐색
- Italiano: Interrogazione del DNA con CRISPR
- Nederlands: CRISPR DNA-ondervraging