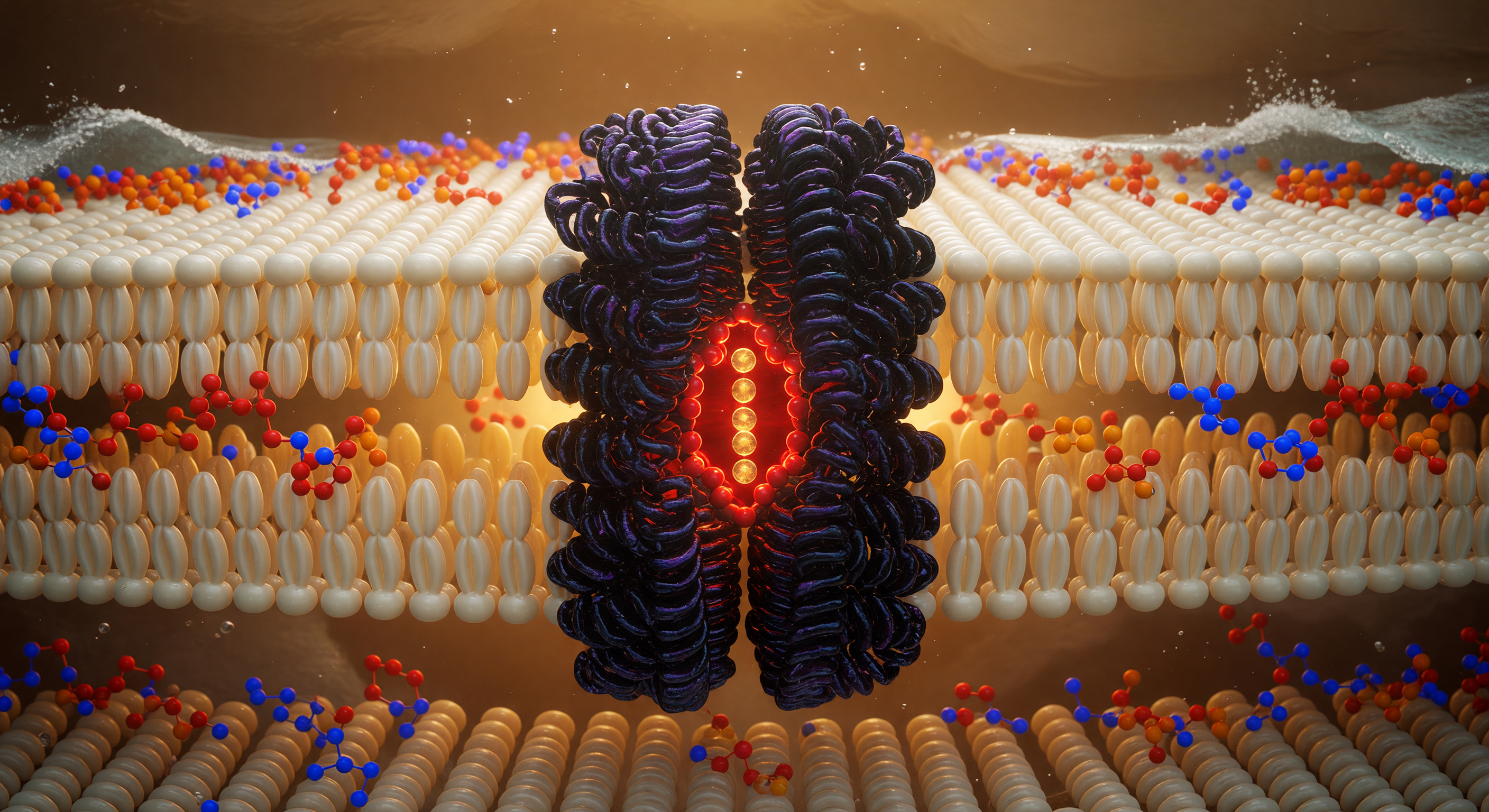
You are suspended at the geometric heart of a living membrane, hovering in the hydrocarbon midplane where phospholipid tails from opposing leaflets interdigitate in a dense, shimmering forest of ivory-gold carbon chains — each one a softly luminous cylinder roughly 0.4 nanometers across, packed so tightly that the interstitial spaces between them read as barely-there shadows in the ambient electrostatic glow. This hydrophobic core, spanning roughly 3–4 nanometers in total thickness, is the thermodynamic engine of membrane integrity: water is excluded not by any hard barrier but by the collective entropic cost of disrupting the hydrogen-bond network outside, making this oily interior a sanctuary of van der Waals stillness punctuated only by the ceaseless thermal writhing of unsaturated chain kinks. Rising through that fog directly ahead, the potassium channel — a tetrameric integral membrane protein whose transmembrane helices are stitched into the bilayer by hydrophobic matching, their nonpolar residues interdigitating seamlessly with the surrounding lipid tails — presents itself as a cathedral column of deep purple-black density, its fourfold symmetry resolving at the pore axis into the iconic selectivity filter: a precise angstrom-scale cage of backbone carbonyl oxygens that strips hydration shells from K⁺ ions and passes them in single file at rates approaching 10⁸ ions per second, burning red-orange with concentrated electron density. Above and below, two luminous headgroup shorelines mark the polar-nonpolar interfaces — phosphorus nodes flaring cadmium-orange, choline nitrogens pulsing cobalt-blue — while beyond them, invisible from here, the aqueous world hammers the charged surface with water molecules at hundreds of meters per second, the turbulent exterior pressing against this amber-lit interior like an ocean held at bay by chemistry alone.
Other languages
- Français: Canal membranaire bicouche central
- Español: Canal de membrana bicapa central
- Português: Canal de membrana bicamada central
- Deutsch: Bilayer-Mittelebene Membrankanal
- العربية: قناة غشاء ثنائي الطبقة
- हिन्दी: द्विपरत झिल्ली चैनल मध्यतल
- 日本語: 二重層中間面膜チャネル
- 한국어: 이중층 중간면 막 채널
- Italiano: Canale di membrana bistrato centrale
- Nederlands: Dubbellaags midvlak membraankanaal